Effective Date: May 2, 2012
(third revision)
Subject
This directive contains the requirements for the production of Pre-Elite seed potatoes from material other than nuclear stock, i.e. disease free cuttings, plants, tubers, or selected clones (Seeds Regulations C.R.C. c. 1400, section 47(2)). In this directive, the term nuclear stock equivalent will be used for this material.
This directive has been revised to update the review date, as well as minor administrative changes. The content of this directive has not changed.
Table of Contents
- Review
- Endorsement
- Amendment Record
- Distribution
- Introduction
- Scope
- Definitions, Abbreviations and Acronyms
- 1.0 General Requirements
- 2.0 Specific Requirements
- 3.0 Appendices
- Appendix 1: Flowchart for the production of Pre-Elite seed potatoes from other sources than Nuclear Stock (nuclear stock equivalent)
- Appendix 2: Sampling procedure for the detection of Clavibacter michiganensis subsp. sepedonicus (Spieckermann & Kotthoff 1914) Davis, Gillaspies, Vidaver & Harris 1984, the causal agent of bacterial ring rot, in nuclear stock equivalent seed potatoes
- Appendix 3: Sampling procedure for viruses and viroid (PSTVd) testing of nuclear stock equivalent seed potatoes
Review
This directive will be reviewed every three years unless otherwise needed. For further information or clarification, please contact the Canadian Food Inspection Agency (CFIA).
Endorsement
Approved by: Chief Plant Health Officer
Amendment Record
Amendments to this directive will be dated and distributed as outlined in the distribution below.
Distribution
- Directive mail list (Regions, PHRA, USDA)
- Provincial Government, Industry (determined by Author)
- National Industry Organizations (determined by Author)
- Internet
Introduction
Although there has been a general movement toward a flush through system starting with disease tested plantlets grown in vitro (nuclear stock), there is still some interest in using other sources of seed to start new field production of seed potatoes. For comparison, Holland, the number one seed potato exporting country, is extensively using clonal selection (process by which field grown tubers are used to initiate a new line of seed potatoes) in its own seed certification system. Therefore, the need has been identified to maintain the possibility of doing clonal selection in Canada.
Scope
This directive specifies the criteria to be met in order for Pre-Elite class seed potatoes to be produced from material other than nuclear stock.
Definitions, Abbreviations and Acronyms
Definitions for terms used in the present document can be found in the Plant Health Glossary of Terms.
1.0 General Requirements
1.1 Legislative Authority
The Seeds Act, R.S. 1985, c. S-8
The Seeds Regulations, Part II, C.R.C., c. 1400
Canadian Food Inspection Agency Fees Notice, Canada Gazette: Part I (as amended from time to time)
1.2 Fees
The CFIA is charging fees in accordance with the Canadian Food Inspection Agency Fees Notice. For information regarding fees associated with imported product, please contact the Import Service Centre (ISC). Anyone requiring other information regarding fees may contact any local CFIA office or visit our Fees Notice website.
1.3 Regulated Pests
Numerous pests regulated by Canada could be associated with the commodities and/or articles regulated by this directive. Notable examples of such pests are enumerated below. A more comprehensive list of pests regulated by Canada can be found on the CFIA website.
The Pests Regulated by Canada list does not include all organisms that potentially represent a plant health risk to Canada. When a new pest is found on plants or plant products, it will be categorized and added to the list as needed.
Viruses: Potato Leaf Roll Virus (PLRV), Potato Virus A (PVA), Potato Virus M (PVM), Potato Virus S (PVS), Potato Virus X (PVX), Potato Virus Y (PVY), Potato Latent Virus (Red la Soda Virus-RLSV) and Potato Mop Top Virus (PMTV)
Viroid: Potato Spindle Tuber Viroid (PSTVd)
Bacteria: Clavibacter michiganensis subsp. sepedonicus (Bacterial Ring Rot-BRR)
2.0 Specific Requirements
Any grower intending to use Nuclear Stock class equivalent to produce Pre-Elite class seed potatoes must comply with the following requirements (a flowchart summary is provided in Appendix 1).
2.1 Selection and Harvest
Mother plants must be selected from a seed potato field certified by the CFIA under the Seed Regulations Part II. Selected clones are specifically chosen because they express characteristics such as freedom from visual symptoms of disease and physiological defects, uniform size and set of tubers, and varietal trueness to type.
At this point, field testing of the mother plants to eliminate material that could be infected by viruses is highly recommended but not required. This could potentially allow elimination of plants affected by secondary infection, but likely not all plants potentially affected by primary infection, thus a post-harvest test is required.
Growers are responsible for administering the selection, handling and maintenance of clones. Growers must retain documentation on seed source, clonal selection and clone identification to demonstrate to the inspector that the identity of the selected material has been maintained.
2.2 Testing
Post-harvest testing is mandatory to ensure the elimination of plants affected by secondary infection. Due to the uneven distribution of diseases over the tubers of a selected clone, and because low concentrations of a disease causing organism can be undetectable by laboratory testing, all tubers (or their progeny) from the selected clone(s) must be post-harvest tested, even if not all tubers are intended to be replanted to produce Pre-Elite class seed.
All required testing must be done at the grower's expense in a CFIA-approved laboratory. A current listing of approved laboratories can be found on the CFIA website.
2.2.1 Testing for Clavibacter michiganensis subsp. sepedonicus - Bacterial Ring Rot (BRR)
In the case of BRR, testing must be done directly on the tubers harvested from the selected clones. Stem testing in the greenhouse or in the field the next season, or tuber testing in the greenhouse are not considered acceptable alternatives.
Core samples must be taken from the stem end of each tuber harvested from the mother plant, and tested for Clavibacter michiganensis subsp. sepedonicus (Spieckermann & Kotthoff 1914) Davis, Gillaspies, Vidaver & Harris 1984, the causal agent of Bacterial Ring Rot (Appendix 2 provides sampling details).
If any core sample tests positive for BRR, all clones from the same farm unit will be rejected for the production of Pre-Elite class seed, and regulatory actions related to Bacterial Ring Rot detection on a seed potato farm unit will be taken.
2.2.2 Virus and Viroid Testing
Virus and viroid testing can be carried out in two ways:
- in the greenhouse during the winter (see section 2.2.2.1 for details), or
- in the field the spring following harvest (see section 2.2.2.2 for details).
If any sample tests positive for any viruses, the entire clone must be rejected for the production of Pre-Elite class seed. Positive clones may still be eligible for certification as lower class seed, provided all other regulatory requirements are met.
If any sample tests positive for the presence of PSTVd, the Growing Crop Certificate will be removed for the entire seed lot from which the clone originates as outlined in section 52.(5)(e) of the Seeds Regulations.
When testing is done in the field (as described in section 2.2.2.2), all positive clones must be removed from the field for certification at the Pre-Elite class. If not, the entire field will be downgraded to a lower class, provided all other regulatory requirements are met.
2.2.2.1 Virus and Viroid Testing in the Greenhouse During the Winter (alternative 1)
One eye from each tuber in the clone can be sprouted and/or grown out. When removing the eye appropriate care has to be taken to avoid any possibilities of cross contamination between clones. To reduce the risk of contamination it is recommended to disinfect knives and equipment between each tuber. Leaf or sprout tissues produced from each excised eye must be tested for PLRV, PSTVd, PVA, PVM, PVS, PVX, PVY, RLSV and PMTV (Appendix 3 contains further details). Clones of the same variety that test negative for all diseases can be planted in bulk, and do not need to be kept separate, as no further testing is required.
2.2.2.2 Virus and Viroid Testing in the Field, the Spring Following Harvest of Selected Clones (alternative 2)
After planting of the selected clones the spring following harvest, terminal three leaflets from fully expanded young compound leaves are taken from at least one plant from each tuber unit and tested for PLRV, PSTVd, PVA, PVM, PVS, PVX, PVY, RLSV and PMTV (Appendix 3 contains further sampling details). Each clone must be properly identified and planted separately in the field. Clones must be planted so that no contact is possible between them before positive ones can be removed. Cut seed must be planted as tuber units, and spacing between each unit is required.
2.3 Further Multiplication
When tubers are tested (as described in section 2.2.1) and are confirmed to be disease free, further multiplication is allowed before planting in the field. Cuttings must be produced in a protected environment. No further testing is required for these cuttings. If the process of multiplication started before all required testing is completed, care must be taken to avoid any possible cross contamination between clones and every clone must be kept separate and clearly identified.
Plants that are field-tested (as described in section 2.2.2) may only be planted as whole seed or be multiplied by cutting the tubers. Extra care must be taken, if tubers are being cut, to avoid cross contamination between clones. To reduce the risk of contamination it is recommended to disinfect knives and equipment between each tuber.
2.4 Application for Certification
The application process for seed potato crop inspection will be the same as for the production of Pre-Elite class from Nuclear Stock class seed potatoes. At the time of first inspection or earlier the applicant for crop inspection must show to the satisfaction of the inspector that all the laboratory testing has been completed. Documentation on clonal selection, clone identification, testing results etc., must be made available to the inspector anytime.
3.0 Appendices
Appendix 1: Flowchart: Production of Pre-Elite Seed Potatoes From Other Sources Than Nuclear Stock (Nuclear Stock Equivalent)
Click on image for larger view
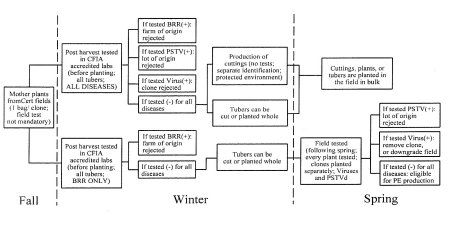
Description for Flowchart for the production of Pre-Elite seed potatoes from other sources than Nuclear Stock (nuclear stock equivalent)
Appendix one illustrates the timeline for laboratory testing of samples for the production of Pre-Elite seed potatoes from sources other than Nuclear Stock. Following harvest of samples in the fall, two alternatives for disease testing are illustrated. In one scenario all disease testing takes place in the winter months, and if results allow, planting may take place in the spring. In the second scenario, Bacterial Ring Rot testing takes place in the winter months, followed by testing for other diseases on individual plants in the field following spring planting. In the second scenario, if results allow, the field is eligible for Pre-elite status.
Appendix 2: Sampling Procedure for the Detection of Clavibacter michiganensis subsp. sepedonicus (Spieckermann & Kotthoff 1914) Davis, Gillaspies, Vidaver & Harris 1984, the Causal Agent of Bacterial Ring Rot (BRR), for Nuclear Stock Equivalent Seed Potatoes
Sampling
- Only tubers will be tested for BRR. All the tubers from the selected clones must be tested; therefore it is presumed that coring will be done by the grower and the cores sent directly to the lab. When cores are sent to the lab, cored tubers should be retained by the grower for further investigation.
- Cores must be taken at the stolon attachment site and must be cone or semi-spherical in shape, approximately 1 cm in diameter at the top and 1 cm deep (see Diagram 1). Each core should weigh between 0.5 - 1.0 g, and include as much of the vascular ring radiating from the stolon attachment as possible.
Diagram 1: How to Take a Core From a Tuber
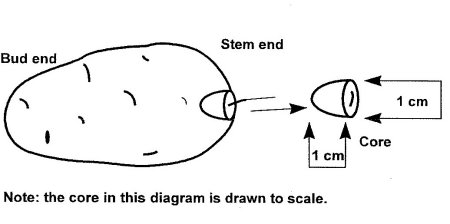
Description of Diagram 1: How to take a core from a tuber
Diagram one illustrates the procedure for removing a core sample from the stem end of the tuber. The core is one centimetre in length and diameter.
Combination of Samples
- Because follow-up measures on a positive sample (for BRR only) are taken on a lot of origin basis only, it is not necessary to bag and submit the samples to the lab by clone; clones from the same lot of origin can be submitted to the lab as one sample. However, clones should be identified on the bag.
- Each sample is logged in separately by the lab. However, according to official protocols, samples may be combined by the lab for testing purposes.
- The maximum number of cores that can be combined by the laboratory for testing is 200 (bags containing more than 200 cores will be subdivided). However, positive tests must be linked to a lot number for follow-up: backup material (tubers already cored and stored by the lab or by the grower) may be used to investigate positive composite samples. Samples from different farm units may not be combined into one laboratory sample.
- Many options being available for sample submission to the lab (some that could help reduce the cost), it is advisable to contact the lab to make arrangements.
Packaging and Shipping
- To assure sample continuity from the field to the lab it is crucial to follow proper packaging, shipping, and identification procedures. If sample integrity is in question, the sample will be discarded, and must be resubmitted.
- Because cores, dried and wrapped in paper towels, can be kept in cold storage (4°C) for up to a maximum of 14 days before processing by the laboratory, keeping them refrigerated at all times is important. Samples can be kept for such an extended period only if no decay is observed. Cores and stems that become decayed during storage will be discarded by the laboratory and a new sample requested.
- It should be ensured that cores or stems are as dry as possible before packing. Cores should be wrapped in paper towels and shipped in plastic bags. Cores should be bagged by lot of origin. Each bag should be properly identified (described in section on Identification below). Close and refrigerate the bags as soon as possible, but no more than two hours after coring. The bags should be kept in cold storage (4°C) long enough (e.g., overnight) and in such a way (well spread out) that the complete sample is brought down to 4°C before packing. Make sure that the sample size indicated on the bags is correct: the lab will only accept a 2% deviation from this number.
- Bags should then be packed loosely in insulated cardboard boxes. Ice-packs should be included on top of the samples (cold air is moving downward), but should be sufficiently insulated from the samples so that freezing is avoided.
- It is important to bear in mind that ice-packs are effective only for a short period: at most 24-48 hours depending on insulation. Shipment of samples must be postponed if the package is likely to be held in transit over a holiday or weekend, except if refrigerated storage is available.
- When more than one sample is packed in the same container, a complete content list of samples submitted must be placed on the top of each shipping container or with the bill of lading; this list should be signed by the collector.
- To ensure sample continuity, packages should be properly sealed so that nobody can open or alter them while in transit (transportation) without the laboratory being able to notice it upon arrival at the lab. If sample integrity is in question, the sample will be discarded and a new sample must be submitted.
- When outside temperatures can go below the freezing point (0°C), freezing of the samples must be avoided: the laboratory will reject any samples showing signs of freezing.
Identification
- Samples from each lot of origin must be submitted in a closed, separate bag, and individually labelled. The following information is required on each label:
- grower's name (exactly as per the current application for Seed Potato Crop Inspection.)
- variety name
- certification number of the lot of origin and clones
- identification of the clones present in the bag
- class (to be assigned/already assigned)
- number of cores submitted
- date collected
- grower's or representative's signature
- test for which sample is submitted
- Note: samples not properly identified will not be processed by the lab until correct identification is received.
Appendix 3: Sampling Procedure for Viruses and Viroid (PSTVd) Testing of Nuclear Stock Equivalent Seed Potatoes
Sampling
- In all situations (field or greenhouse) where sampling has to be done for nuclear stock equivalent seed potatoes every tuber unit must be sampled.
- Testing will be done on leaf or sprout material. The terminal three leaflets from a fully expanded young compound leaf are required from a plant from each tuber unit (Identified in Diagram 2). On arrival in the laboratory, acceptable samples must have at least 2 cm2 of intact leaf tissue per leaflet. Slight wilting or breakdown at the leaf margins is acceptable.
Leaves are collected from the upper third of the plant; this can be done as soon as the first compound leaves open, and always before plants begin senescing. When testing is done in the field sampling should be made early enough so that any removal pursuant to positive results can take place before cross contamination, by any means (insects, mechanical transmission by machinery, plants touching each other, etc.) can happen.
Sprouts of at least 1.5 cm have to collected from tubers that were maintain between 18-25°C.
Diagram 2: How to Sample Leaves for Virus Testing
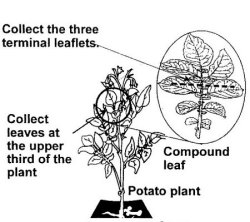
Description of Diagram 2: How to sample leaves for virus testing
Appendix three illustrates collection points for leaf samples on a diagram of a potato plant. A compound leaf is illustrated with the three terminal leaflets indicated, and instruction is noted to collect leaves at the upper third of the plant.
In most cases, according to official protocols, testing must be done within 72 hours from leaves or sprouts collection. The local lab can provide further details:
Packaging
- According to official protocols composite testing is possible. Options are available that can help reduce the cost: contact your local laboratory to make arrangements. However when a test is positive it must be linked to a clone. When composite samples of more than one clone are tested (to lower costs), backup material (only the terminal leaflet is used for the first test, leaving 2 leaflets for further analysis) may be used to investigate positive samples.
- Leaves/sprouts must be bagged by clone, and the clone properly identified. Loosely folding over the opening of the bag and stapling shut is a good way of sealing the bag. The bag should not be sealed airtight, particularly if it is warm or damp; if necessary make breathing holes.
- The leaf/sprout samples must be cooled (but not frozen) to 5°C as soon as possible. It must be done within the hour of picking the leaves or the sprouts (particularly on warm days). If "ice-packs" are used, they should be insulated with two or three layers of paper or other packing material and be placed in the middle or top of the cooler. Two 6" x 6" "ice-packs" per cooler are usually sufficient. Avoid packing the leaves too tightly.
Shipping
- If the leaves are to be shipped by courrier, the leaves should be held overnight in a refrigerated storage. For shipment, the bags should be packed loosely in the Styrofoam containers and place in cardboard boxes. "Ice-packs" should be included, but should be sufficiently insulated so as not to freeze the leaves.
- The 72 hours maximum period indicated above (sampling, paragraph 3) must be respected.
Identification (to assure sample continuity from the field to the lab)
- Every bag should contain the following information: grower or company name, the grower's signature, delivery date, variety, class and certification number. As indicated above, all clones must be identified separately.
- A complete content list must be placed on the top of the samples or with the bill of lading, and signed by the collector.
- Collection and submission dates of every sample must be provided.
- Packages should be properly sealed.
Notes:
- Samples not properly identified will not be processed until correct identification is received.
- If sample integrity is in question, the sample will be discarded, and must be re-submitted.
- Shipment of samples must be postponed if it is apparent that the package will be held in transit over a holiday or weekend.