Table of Contents
- Introduction
- Executive Summary
- 1.0 Purpose
- 2.0 Scope
- 3.0 Definitions
- 4.0 Background
- 5.0 Pest Risk Assessments Summaries
- 6.0 Risk Management Considerations
- 7.0 Pest Risk Management Options
- 8.0 Risk Management Decision
- 9.0 References
- Appendix 1A: Pest Risk Assessment Summary for Aegilops cylindrica (jointed goat grass)
- Appendix 1B: Risk Management Considerations for Aegilops cylindrica (jointed goat grass)
- Appendix 2A: Pest Risk Assessment Summary for Alopecurus myosuroides (slender foxtail)
- Appendix 2B: Risk Management Considerations for Alopecurus myosuroides (slender foxtail)
- Appendix 2C: Import data for wheat, rye, barley and other unmilled cereals
- Appendix 2D: Import data for grass seed
- Appendix 2E: Import data for hay and straw
- Appendix 3A: Pest Risk Assessment Summary for Centaurea iberica (Iberian starthistle)
- Appendix 3B: Risk Management Considerations for Centaurea iberica (Iberian starthistle)
- Appendix 3C: Import data for Centaurea iberica (Iberian star-thistle)
- Appendix 4A: Pest Risk Assessment Summary for Centaurea solstitialis (yellow star-thistle)
- Appendix 4B: Risk Management Considerations for Centaurea solstitialis (yellowstar-thistle)
- Appendix 4C: Import Data for Alfalfa seeds
- Appendix 4D: Import Data for Hay and Straw
- Appendix 5A: Pest Risk Assessment Summary for Crupina vulgaris (common crupina)
- Appendix 5B: Risk Management Considerations for Crupina vulgaris (common crupina)
- Appendix 5C: Import Data for Field Crops
- Appendix 5D: Import Data for Hay and Straw
- Appendix 6A: Pest Risk Assessment Summary for Dioscorea polystachya (Chinese yam)
- Appendix 6B: Risk Management Considerations for Dioscorea polystachya (Chinese yam)
- Appendix 7A: Pest Risk Assessment Summary for Echium plantagineum (Paterson's curse)
- Appendix 7B: Risk Management Considerations for Echium plantagineum (Paterson's curse)
- Appendix 8A: Pest Risk Assessment Summary for Microstegium vimineum (Japanese stiltgrass)
- Appendix 8B: Risk Management Considerations for Microstegium vimineum (Japanese stiltgrass)
- Appendix 8C: Import Data for field crops used as birdfeed
- Appendix 8D: Import Data for Hay and Straw
- Appendix 9A: Pest Risk Assessment Summary for Nassella trichotoma (serrated tussock)
- Appendix 9B: Risk Management Considerations for Nassella trichotoma (serrated tussock)
- Appendix 9C: Import Data forage seeds for sowing a originating
- Appendix 10A: Pest Risk Assessment Summary for Paspalum dilatatum (Dallis grass)
- Appendix 10B: Risk Management Considerations for Paspalum dilatatum (Dallis grass)
- Appendix 10C: Import Data forage seeds for sowing
- Appendix 11A: Pest Risk Assessment Summary for Persicaria perfoliata (mile-a-minute weed)
- Appendix 11B: Risk Management Considerations for Persicaria perfoliata (mile-a-minute weed)
- Appendix 12A: Pest Risk Assessment Summary for Senecio inaequidens (South African ragwort)
- Appendix 12B: Risk Management Considerations for Senecio inaequidens (South African ragwort)
- Appendix 12C: Assessment of Measures for Senecio inaequidens (South African ragwort)
- Appendix 13A: Pest Risk Assessment Summary for Senecio madagascariensis (Madacascar ragwort)
- Appendix 13B: Risk Management Considerations for Senecio madagascariensis (Madacascar ragwort)
- Appendix 13C: Assessment of Measures for Senecio madagascariensis (Madacascar ragwort)
- Appendix 14A: Pest Risk Assessment Summary for Solanum elaeagnifolium (silverleaf nightshade)
- Appendix 14B: Risk Management Considerations for Solanum elaeagnifolium (silverleaf nightshade)
- Appendix 14C: Import Data for selected cereal grains from countries where Solanum elaeagnifolium is present (2006-2008)
- Appendix 14D: Import Data for Maize and Forage seed from countries where Solanum elaeagnifolium is present (2006-2008)
- Appendix 14E: Import Data for Hay and straw from countries where Solanum elaeagnifolium is present (2006-2008)
- Appendix 15A: Pest Risk Assessment Summary for Zygophyllum fabago (Syrian bean-caper)
- Appendix 15B: Risk Management Considerations for Zygophyllum fabago (Syrian bean-caper)
- Appendix 16: Consultation
Introduction
Executive Summary
This risk management document (RMD) is part of a three step pest risk analysis process examining the risk to Canada associated with the importation, cultivation and trade of pest plants listed in Table 1: List of plant regulated as pest plants in Canada. The RMD provides a summary of the pest risk assessment for each pest plant, and potential risk mitigation measures. Mitigation measures may be applied to reduce the pest risk to acceptable levels and provide a cost-effective means of preventing the entry of pests into Canada. Although stakeholders were consulted on the individual RMDs in 2010, the general information and considerations for each pest were similar. As such the original RMD information has been combined into one decision document. Invasive plants are those plant species that spread when introduced outside of their natural past or present distribution and cause serious and often irreversible damage to Canada's ecosystems, economy and society.
The pest plants listed in Table 1 could establish in parts of Canada. Following stakeholder consultation in 2010, the Canadian Food Inspection Agency (CFIA) has decided to regulate these pest plants under the Plant Protection Act and the Seeds Act. As such, the plants listed in Table 1 will be regulated as pest under the Plant Protection Act, and placed on the List of Pests Regulated by Canada. The Plant Protection Act gives CFIA the authority to take action and respond to incursions of the pests in Canada. Furthermore, when a seed is a key pathway, the CFIA plans to designate some of these plants as prohibited noxious weed seeds under the Seeds Act's Weed Seeds Order.
| Species | Hardiness ZoneTable Note 1 and Distribution |
Sector(s)/Habitat Affected | Agricultural, Environmental and/or Societal Impacts | Potential Human Mediated Pathways of Entry |
|---|---|---|---|---|
1. Aegilops cylindrica(jointed goatgrass/Égilope cylindrique)  | Hardiness Zone 2 |
|
|
|
2. Alopecurus myosuroides (slender foxtail/Vulpin des champs) | Hardiness Zone 6 |
|
|
|
3. Centaurea iberica (Iberian starthistle/Centaurée ibérique ) | Hardiness Zone 6 |
|
|
|
4. Centaurea solstitialis (yellow starthistle/Centaurée du solstice) | Hardiness Zone 5 |
|
|
|
5. Crupina vulgaris (common crupina/Crupine vulgaire ) | Hardiness Zone 4 |
|
|
|
6. Dioscorea polystachya (Chinese yam/Igname de Chine) | Hardiness Zone 5 |
|
|
|
7. Echium plantagineum (Paterson's curse/Vipérine à feuilles de plantain) | Hardiness Zone 5 |
|
|
|
| 8. Microstegium vimineum (Japanese stiltgrass/Aucun nom commun français répertorié)
| Hardiness Zone 5 |
|
|
|
| 9. Nassella trichotoma (serrated tussock /Stipe à feuilles dentées)
| Hardiness Zone 7 |
|
|
|
10. Paspalum dilatatum (Dallis grass/Herbe de Dallis) | Hardiness Zone 6 |
|
|
|
| 11. Persicaria perfoliata (mile-a-minute weed/Renouée perfoliée)
| Hardiness Zone 6 |
|
|
|
| 12. Senecio inaequidens (South African ragwort/Séneçon du cap)
| Hardiness Zone 6 |
|
|
|
| 13. Senecio madagascariensis (Madagascar ragwort/Séneçon de Madagascar)
| Hardiness Zone 8 |
|
|
|
| 14. Solanum elaeagnifolium (silverleaf nightshade/Morelle jaune)
| Hardiness Zone 5 |
|
|
|
| 15. Zygophyllum fabago (Syrian bean-caper/Fabagelle)
| Hardiness Zone 5 |
|
|
|
Preface
As described by the International Plant Protection Convention (IPPC), Pest Risk Analysis (PRA) includes three stages: initiation, pest risk assessment and pest risk management. Initiating the PRA process involves identifying pests and pathways of concern and defining the PRA area. Pest risk assessment provides the scientific basis for the overall management of risk. Pest risk management is the process of identifying and evaluating potential mitigation measures which may be applied to reduce the identified pest risk to acceptable levels and selecting appropriate measures.
This Risk Management Document (RMD) includes a summary of the findings of a pest risk assessment and records the pest risk management process for the identified issue. It is consistent with the principles, terminology and guidelines provided in the IPPC standards for pest risk analysis.
1.0 Purpose
To record the risk management decision regarding the regulation of the following pest plants:
- Nom latin
- Aegilops cylindrica
- Alopecurus myosuroides
- Centaurea iberica
- Centaurea solstitialis
- Crupina vulgaris
- Dioscorea polystachya
- Echium plantagineum
- Microstegium vimineum
- Nassella trichotoma
- Paspalum dilatatum
- Persicaria perfoliata
- Senecio inaequidens
- Senecio madagascariensis
- Solanum elaeagnifolium
- Zygophyllum fabago
- Common name
- jointed goatgrass
- slender foxtail
- Iberian starthistle
- yellow starthistle
- common crupina
- Chinese yam
- Paterson's curse
- Japanese stiltgrass
- serrated tussock
- Dallis grass
- mile-a-minute weed
- South African ragwort
- Madacascar ragwort
- silverleaf nightshade
- Syrian bean-caper
Note: In the context of this document the term listed weeds refers to the plants listed above.
2.0 Scope
This risk management document (RMD) summarizes the Canadian Food Inspection Agency's (CFIA) decision to regulate the listed weeds as pests for Canada.
Information pertaining to current import requirements for specific plants or plant products may be obtained from the CFIA Automated Import Reference System (AIRS).
Additional points for consideration
- Prior to use as human food, new plants and/or derived products that fit the definition of a novel food require approval under the authority of the Food and Drugs Act from Health Canada.
- Prior to use as livestock feed, new plants and/or derived products must be assessed and approved by the Animal Feed Division, Canadian Food Inspection Agency (CFIA) under the authority of the Feeds Act and Regulations. A positive list of approved ingredients can be found in Schedules IV and V of the Feeds Regulations, 1983.
- Release (e.g. cultivation) of new plants into the Canadian environment may require prior approval under the authority of the Seeds Act and the Seeds Regulations from the Field Crops and Inputs Division, CFIA.
- The importation and sale of seed in Canada must meet the requirements of the Seeds Act, Seeds Regulations and Weed Seeds Order.
- Imported commodities likely to contain the listed weeds in this document must meet the phytosanitary requirements for other regulated organisms, as stated in CFIA's Plant Protection Policies and Directives and in AIRS.
3.0 Definitions
Definitions for terms used in this document can be found in the Plant Health Glossary of Terms and the IPPC Glossary of Phytosanitary Terms.
4.0 Background
- This RMD describes the risk associated with each invasive plant and the management options available to mitigate that risk and is an effort to prevent the introduction of invasive plants and weeds into Canada under the authority of the Plant Protection Act and the Seeds Act.
- The CFIA is evaluating and, where appropriate, restricting the importation and spread of invasive plants as part of its mandate to protect Canada's plant resource base and itscommitment to limit the introduction and spread of invasive plants under An Invasive Alien Species Strategy for Canada (Government of Canada 2004). The Strategy aims to reduce the risk of invasive species to the environment, economy, and society, and to protect environmental values such as biodiversity and sustainability.
- The CFIA prevents the introduction andspread of some invasive plants in Canada, those regulated as quarantine pestsunder the Plant Protection Act and designated as prohibited noxious weeds under the Seeds Act. (A quarantine pest is a pest of potential economicimportance to the endangered area and not yet present or present but notwidely distributed and being officially controlled.).
- Under the Plant Protection Regulations, Importation Requirements (subsection 29.1) "… no person shall import into Canada anything that is a pest, is or could be infested or constitutes or could constitute a biological obstacle to the control of a pest, unless the person has obtained and furnished to an inspector a valid permit number and, as applicable, a foreign Phytosanitary Certificate or a foreign Phytosanitary Certificate for Re-export."
- The CFIA requires that certain measures be taken both for import and domestic movement; to prevent the introduction or spread of plants considered to be pests in Canada. The D-12-01: Phytosanitary requirements to prevent the introduction of plants regulated as pests in Canada, outlines the general phytosanitary requirements to prevent the introduction or spread of these plants in Canada under the Plant Protection Act.
Note: It is well recognized that the risk of invasive species in Canada is a shared risk and therefore a shared responsibility among government jurisdictions and stakeholders. Once an invasive plant is determined to be a pest and is introduced into Canada, the CFIA develops and enforces domestic regulatory measures to mitigate the risks associated with the plant species. The CFIA's approach for domestic regulation is based on the principles of: most up-to-date science during the Pest Risk Assessment, shared responsibility and partnerships with provinces and industry stakeholders, consultation and regular review.
5.0 Pest Risk Assessments Summaries
The CFIA completed a weed risk assessment for each of the species listed in this document. The Table 2: Weed Risk Assessments by Species references the appendices for each risk assessment.
6.0 Risk Management Considerations
6.1 Introduction
The Risk Management Considerations summarizes the rationale in determining the regulatory status of the plant. It outlines the possible phytosanitary import and domestic measures for traded commodities. The commodities may be the plant itself (intentional introduction) or a product contaminated with the plant (unintentional introduction).
6.2 International Responsibilities, Government of Canada Priorities and CFIA Objectives
The CFIA plays an important role in protecting Canada's plant resource base from pests. The objectives of the Plant Protection Program within the CFIA are: (1) to prevent the introduction and spread within Canada of pest plants of quarantine significance, including invasive plants; (2) to detect and control or eradicate designated pest plants in Canada; and (3) to certify plant and plant products for domestic and export trade.
Canada is a contracting party to the International Plant Protection Convention (IPPC). Canada is a member of the World Trade Organization (WTO) and the IPPC is formally identified in the WTO Sanitary and Phytosanitary (SPS) Agreement as the international standard setting organization for plant health. The IPPC is an international treaty to secure action to prevent the spread and introduction of pests of plants and plant products (including plants as pests), and to promote appropriate measures for their control.
The CFIA is Canada's official National Plant Protection Organization (NPPO) responsible for implementing the provisions of the IPPC and administers the Plant Protection Act, Plant Protection Regulations, Seeds Act and Weed Seeds Order. The Plant Protection Act provides authority to prevent the importation, exportation and spread of pests injurious to plants, provides for control and eradication methods and for the issuance of certificates.
In 1996, as a party to the United Nations Convention on Biological Diversity (CBD), Canada developed its own Canadian Biodiversity Strategy, which recognized the need to conserve biological diversity and promote the sustainable use of biological resources through increased understanding, legislation, incentives and other means. As party to these international and national instruments, Canada has a strong commitment to addressing the deleterious impacts of invasive plants.
Additionally, in September 2004 Canada introduced An Invasive Alien Species Strategy for Canada, aimed to minimize the risk of invasive alien species (IAS) to the environment, economy, and society, and to protect environmental values such as biodiversity and sustainability. The CFIA provides leadership in the implementation of the national IAS strategy as it relates to invasive plants and pest plants.
7.0 Pest Risk Management Options
Table 4 summarizes the risk management options considered for each pest plant and Table 5 weighs the advantages and disadvantages for each risk management decision.
Table 4: Considered option for each pest plant
| Options | Add to the List of Pests Regulated by Canada and regulate it under the Plant Protection Act and Regulations | Add or continue to regulate as a prohibited weed under the Weed Seeds Order of the Seeds Act and Regulations. | Do not add to the List of Regulated Pests | Do not add to the Weed Seeds Order | Other alternative specific to this option |
|---|---|---|---|---|---|
| Option 1 (recommended) | X | X | |||
| Option 2 | X | X |
| Options | Add to the List of Pests Regulated by Canada and regulate it under the Plant Protection Act and Regulations | Add or continue to regulate as a prohibited weed under the Weed Seeds Order of the Seeds Act and Regulations. | Do not add to the List of Regulated Pests | Do not add to the Weed Seeds Order | Other alternative specific to this option |
|---|---|---|---|---|---|
| Option 1 (recommended) | X | X | |||
| Option 2 | X | X | |||
| Option 3 | X | X |
| Options | Add to the List of Pests Regulated by Canada and regulate it under the Plant Protection Act and Regulations | Add or continue to regulate as a prohibited weed under the Weed Seeds Order of the Seeds Act and Regulations. | Do not add to the List of Regulated Pests | Do not add to the Weed Seeds Order | Other alternative specific to this option |
|---|---|---|---|---|---|
| Option 1 (recommended) | X | X | |||
| Option 2 | X | X | |||
| Option 3 | X | X |
| Options | Add to the List of Pests Regulated by Canada and regulate it under the Plant Protection Act and Regulations | Add or continue to regulate as a prohibited weed under the Weed Seeds Order of the Seeds Act and Regulations. | Do not add to the List of Regulated Pests | Do not add to the Weed Seeds Order | Other alternative specific to this option |
|---|---|---|---|---|---|
| Option 1 (recommended) | X | X | |||
| Option 2 | X | X |
| Options | Add to the List of Pests Regulated by Canada and regulate it under the Plant Protection Act and Regulations | Add or continue to regulate as a prohibited weed under the Weed Seeds Order of the Seeds Act and Regulations. | Do not add to the List of Regulated Pests | Do not add to the Weed Seeds Order | Other alternative specific to this option |
|---|---|---|---|---|---|
| Option 1 (recommended) | X | X | |||
| Option 2 | X | X |
| Options | Add to the List of Pests Regulated by Canada and regulate it under the Plant Protection Act and Regulations | Add or continue to regulate as a prohibited weed under the Weed Seeds Order of the Seeds Act and Regulations. | Do not add to the List of Regulated Pests | Do not add to the Weed Seeds Order | Other alternative specific to this option |
|---|---|---|---|---|---|
| Option 1 (recommended) | X | X | Prohibit the importation of plants and plant parts BOTH for propagation AND for consumption or medicinal use | ||
| Option 2 | X | X | Prohibit the importation of plants and plant parts for propagation BUT NOT for consumption or medicinal use | ||
| Option 3 | X | X |
| Options | Add to the List of Pests Regulated by Canada and regulate it under the Plant Protection Act and Regulations | Add or continue to regulate as a prohibited weed under the Weed Seeds Order of the Seeds Act and Regulations. | Do not add to the List of Regulated Pests | Do not add to the Weed Seeds Order | Other alternative specific to this option |
|---|---|---|---|---|---|
| Option 1 (recommended) | X | X | |||
| Option 2 | X | Prohibition except for Research Field Trials | |||
| Option 3 | Provisionally Allowed for Industrial Crop Use | ||||
| Option 4 | Provisionally Allowed for Industrial Crop Use with Oversight from a Provincial/Territorial Agency and CFIA |
| Options | Add to the List of Pests Regulated by Canada and regulate it under the Plant Protection Act and Regulations | Add or continue to regulate as a prohibited weed under the Weed Seeds Order of the Seeds Act and Regulations. | Do not add to the List of Regulated Pests | Do not add to the Weed Seeds Order | Other alternative specific to this option |
|---|---|---|---|---|---|
| Option 1 (recommended) | X | X | |||
| Option 2 | X | X | |||
| Option 3 | X | X |
| Options | Add to the List of Pests Regulated by Canada and regulate it under the Plant Protection Act and Regulations | Add or continue to regulate as a prohibited weed under the Weed Seeds Order of the Seeds Act and Regulations. | Do not add to the List of Regulated Pests | Do not add to the Weed Seeds Order | Other alternative specific to this option |
|---|---|---|---|---|---|
| Option 1 (recommended) | X | X | |||
| Option 2 | X | X |
| Options | Add to the List of Pests Regulated by Canada and regulate it under the Plant Protection Act and Regulations | Add or continue to regulate as a prohibited weed under the Weed Seeds Order of the Seeds Act and Regulations. | Do not add to the List of Regulated Pests | Do not add to the Weed Seeds Order | Other alternative specific to this option |
|---|---|---|---|---|---|
| Option 1 (recommended) | X | X | |||
| Option 2 | X | X | |||
| Option 3 | X | X |
| Options | Add to the List of Pests Regulated by Canada and regulate it under the Plant Protection Act and Regulations | Add or continue to regulate as a prohibited weed under the Weed Seeds Order of the Seeds Act and Regulations. | Do not add to the List of Regulated Pests | Do not add to the Weed Seeds Order | Other alternative specific to this option |
|---|---|---|---|---|---|
| Option 1 (recommended) | X | X | |||
| Option 2 | X | X | |||
| Option 3 (recommended) | X | X | |||
| Option 4 | X | X |
| Options | Add to the List of Pests Regulated by Canada and regulate it under the Plant Protection Act and Regulations | Add or continue to regulate as a prohibited weed under the Weed Seeds Order of the Seeds Act and Regulations. | Do not add to the List of Regulated Pests | Do not add to the Weed Seeds Order | Other alternative specific to this option |
|---|---|---|---|---|---|
| Option 1 (recommended) | X | X | |||
| Option 2 | X | X | |||
| Option 3 | X | X |
| Options | Add to the List of Pests Regulated by Canada and regulate it under the Plant Protection Act and Regulations | Add or continue to regulate as a prohibited weed under the Weed Seeds Order of the Seeds Act and Regulations. | Do not add to the List of Regulated Pests | Do not add to the Weed Seeds Order | Other alternative specific to this option |
|---|---|---|---|---|---|
| Option 1 (recommended) | X | X | |||
| Option 2 | X | X | |||
| Option 3 | X | X |
| Options | Add to the List of Pests Regulated by Canada and regulate it under the Plant Protection Act and Regulations | Add or continue to regulate as a prohibited weed under the Weed Seeds Order of the Seeds Act and Regulations. | Do not add to the List of Regulated Pests | Do not add to the Weed Seeds Order | Other alternative specific to this option |
|---|---|---|---|---|---|
| Option 1 (recommended) | X | X | |||
| Option 2 | X | X | |||
| Option 3 | X | X |
| Options | Add to the List of Pests Regulated by Canada and regulate it under the Plant Protection Act and Regulations | Add or continue to regulate as a prohibited weed under the Weed Seeds Order of the Seeds Act and Regulations. | Do not add to the List of Regulated Pests | Do not add to the Weed Seeds Order | Other alternative specific to this option |
|---|---|---|---|---|---|
| Option 1 (recommended) | X | X | |||
| Option 2 | X | X | |||
| Option 3 | X | X |
| Risk Management Decisions | Advantages | Disadvantages |
|---|---|---|
Add to the List of Pests Regulated by Canada and regulate it under the Plant Protection Act and Regulations Add or continue to regulate as a prohibited weed under the Weed Seeds Order of the Seeds Act and Regulations |
|
|
Do not add to the List of Regulated Pests Do not add to the Weed Seeds Order |
|
|
Prohibit the importation of plants and plant parts both for propagation and for consumption or medicinal use Prohibit the importation of plants and plant parts both for propagation but not for consumption or medicinal use |
|
|
8.0 Risk Management Decision
After stakeholder consultation and review of information and comments, the CFIA has decided to regulate the listed weeds in this RMD under the Plant Protection Act. Under the Plant Protection Regulations, imported things must be free from species on the List of Pests Regulated by Canada.
9.0 References
- Agriculture and Agri-Food Canada (AAFC). 2008. Canadian Horticulture Sector - 2007 Crop Year. Agriculture and Agri-Food Canada report [October 2009].
- Agriculture and Agri-food Canada 2007. Forage.
- Agriculture Canada. 2008. Seed Industry Statistics [July 2009].
- Alabama Invasive Plant Council (AIPC), 2007. List of Alabama's Invasive Plants by Land-Use and Water-Use Sectors for 2007 (PDF, 51 kb) [cited 2009].
- Alberta Weed Control Act (2008, c. W-5.1).
- Aldrich-Markham, S. 1992. Blackgrass - Alopecurus myosuroides Huds; PNW 377 Edited by O.S. University. Pacific Northwest Extension (Oregon State University, Washington State University, University of Idaho), Corvallis, or.
- Allison, K. 2007. Nassella trichotoma (Nassella trichotoma (Nees) Hack. ex Arechav.) Pest risk assessment request No. 2006-06, Plant Health Risk Assessment Unit, Canadian Food Inspection Agency, Ottawa. Unpublished report submitted to CFIA's Plant Health and Biosecurity Directorate in April 2007.
- Allison, K. 2009. Alopecurus myosuroides Huds. (slender foxtail) Pest risk assessment request No. 2009-20, Plant and Biotechnology Risk Assessment Unit, Canadian Food Inspection Agency, Ottawa, ON. Unpublished report submitted to CFIA's Plant Health and Biosecurity Directorate in May, 2009.
- Allison, K. 2009. Paspalum dilatatum (Poir.) (Dallis Grass) pest risk assessment request No. 2009-20 padl, Plant Health Risk Assessment Unit, Canadian Food Inspection Agency, Ottawa, ON. Unpublished report submitted to CFIA's Plant Health and Biosecurity Directorate in May, 2009.
- Allison, K. 2009. Canadian Weed Risk Assessment (C-WRA) for Zygophyllum fabago. PRA REQUEST: 2009-20 zyfa. Canadian Food Inspection Agency. Plant and Biotechnology Risk Assessment Unit, Plant Health Science Division. Ottawa, ON.
- Allison, K. 2009. Centaurea iberica (Iberian star-thistle). Pest risk assessment request No. 2009-20 ceib. Plant and Biotechnology Risk Assessment Unit, Canadian Food Inspection Agency, Ottawa. Unpublished report submitted to CFIA's Plant Health and Biosecurity Directorate in April 2007.
- Allison, K. 2009. Centaurea solstitialis L. (yellow star-thistle). Pest risk assessment request No. 2009-20-ceso, Plant and Biotechnology Risk Assessment Unit, Canadian Food Inspection Agency, Ottawa, ON. Unpublished report submitted to CFIA's Plant Health and Biosecurity Directorate in October, 2009.
- Allison, Ken. 2009. Weed Risk Assessment 2009-20-dipo-Revised: Dioscorea polystachya Turcz. (Chinese Yam). Plant and Biotechnology Risk Assessment Unit, Canadian Food Inspection Agency. 2009.
- Anonymous. 2007. Syrian bean-caper (Zygophyllum fabago L.) [May, 2009].
- Anonymous. undated. Control of Knapweeds in Native Grasslands. ed. S.W. Authority. Saskatchewan Watershed Authority, Regina, SK. p. 4.
- Arizona Department of Agriculture (ADA). 2005. Prohibited, regulated and restricted noxious weeds. Plant Services Division, Arizona Department of Agriculture [August 2009].
- Australian Weeds Committee. 2008. Weeds Australia: An Australian Weeds Committee National Initiative [2008].
- B.C. Weed Control Act (RSBC 1996, c. 487).
- Bailey, L.G., and Bailey, E.Z. 1976. Hortus Third: A Concise Dictionary of Plants Cultivated in the United States and Canada. McMillan Publishing Co., New York, NY.
- Ball, D.A., Young, F.L. and Ogg, A.G. 1999. Selective control of jointed goatgrass (Aegilops cylindrica) with imazamox in herbicide-resistant wheat. Weed Technology 13:77–82.
- Ballast Water Control and Management Regulations (SOR/2006-129).
- Barbier, E. and D. Knowler. 2006. Commercialization decisions and the economics of introduction. Euphytica 148: 151–164.
- Barkworth, M. E., 2006. The Grass Manual on the Web.
- BC Ministry of Agriculture and Lands. 2002. Field Guide to Noxious and Other Selected Weeds of British Columbia [July 2009].
- BC Ministry of Forests. 1999. The Ecology of the Coastal Douglas-fir Zone. Victoria, BC [May 29, 2009].
- BC Ministry of Forests. 1999. The Ecology of the Coastal Western Hemlock Zone. Victoria, BC [May 29, 2009].
- Bega Valley Fireweed Association. no date. Impact on the Bega Valley [June 16, 2009].
- Beier, B.-A., Chase, M.W., and Thulin, M. 2003. Phylogenetic relationships and taxonomy of subfamily Zygophylloideae (Zygophyllaceae) based on molecular and morphological data.
- Plant Systematics and Evolution 240: 11-39.
- Berti, M., B.L. Johnson, S. Dash, S. Fischer, R. Wilckens, and F. Hevia. 2007. Echium: A Source of Stearidonic Acid Adapted to the Northern Great Plains in the US. In J. Janick and A. Whipkey (eds.). Issues in new crops and new uses. ASHS Press, Alexandria, VA.
- Bond, EJ. 1984. Manual of fumigation for insect control FAO Plant Production and Protection Papers- 54 Vol. 2008. FAO, Rome, Italy.
- Boyd, J. W., and Murray, D. S. 1982. Effects of shade on silverleaf nightshade (Solanum elaeagnifolium). Weed Science 30(3): 264-269.
- Boyd, J. W., and Murray, D. S. 1982. Growth and development of silverleaf nightshade (Solanum elaeagnifolium). Weed Science 30(3): 238-243.
- Boyd, J. W., Murray, D. S., and Tyrl, R. J. 1984. Silverleaf nightshade, Solanum elaeagnifolium, origin, distribution, and relation to man. Economic Botany 38(2): 210-217.
- Bravo, M. 2008. Mile-A-Minute Control Management Plan. Pennsylvania Department of Agriculture otany Weed Science Program. Harrisburg, PA. 12 pp. (PDF, 79 kb).
- Breitung, A.J. 1957. Annotated catalogue of the vascular flora of Saskatchewan. American Midland Naturalist 58: 1-72.
- British Columbia Ministry of Agriculture, Food and Fisheries (BCMAFF). 2004. An Overview of the British Columbia Grape Industry. 34 pp (PDF, 339 kb) [August 2009].
- Brouillet, L., Coursol, F., and Favreau, M. 2006. VASCAN. The database of Canadian vascular plants. Herbier Marie-Victorin, Institut de recherche en biologie végétale, Université de Montréal, Montreal, QC.
- CAB International. 2007. Commonwealth Agriculture Bureau - Crop Protection Compendium. CD-ROM - Internet application, Wallingford, U.K.
- California Department of Food and Agriculture (CDFA). 2003. Plant quarantine manual, California plant quarantine policy - weeds. California Department of Food and Agriculture (PDF, 131 kb) [August 2009].
- Callaway, J.C., T. Tennil, and D.W. Pate. 1996. Occurrence of “omega-3” stearidonic acid (cis-6, 9, 12, 15-octadecatetraenoic acid) in hemp (Cannabis sativa L.) seed. J. Int. Hemp Assoc. 3(2):61–63.
- Canada Shipping Act, 2001 (2001, c.26).
- Canada. 2000. Some Factors in Canada United States Wheat Trade. Canadian Embassy, Washington, D.C (PDF, 79 kb) [May 2009].
- Canadian Food Inspection Agency (CFIA). 2008 Plants of Canada Database. CFIA, Ottawa, ON.
- Canadian Food Inspection Agency (CFIA). 1990. Directive 90-03-AP-18: Importation of Hides, Skins, Furs, Stuffed Animals, Trophies, Wool and Feathers. CFIA, Ottawa, ON.
- Canadian Food Inspection Agency (CFIA). 1999. Directive D-98-06: Interim Import requirements of parasitic plants: Cuscuta, Striga, and Orobanche. CFIA, Ottawa, ON.
- Canadian Food Inspection Agency (CFIA). 2007. Weed Risk Assessment: Crupina vulgaris Cass., Common Crupina. CFIA, Ottawa, ON.
- Canadian Food Inspection Agency (CFIA). 2008. Directive D-97-04: Application, procedures, issuance and use of a Permit to Import under the Plant Protection Act. CFIA, Ottawa, ON.
- Canadian Food Inspection Agency (CFIA). 1990. Directive AHPD-DSAE-2001-1-1, Importation of Hides, Skins, Furs, Stuffed Animals, Trophies, Wool and Feathers. CFIA, Ottawa, ON.
- Canadian Food Inspection Agency (CFIA). 1999. Directive 98-06: Interim Import requirements of parasitic plants: Cuscuta, Striga, and Orobanche. CFIA, Ottawa, ON.
- Canadian Food Inspection Agency (CFIA). 2006. Directive 02-02: Plant Protection Import Requirements for Rooted, or Unrooted Plants, Plant Parts, and Tissue Cultured Plants for Propagation. CFIA, Ottawa, ON.
- Canadian Food Inspection Agency (CFIA). 2007. 2007 Plant Protection Survey Report, available online [July 2009].
- Canadian Food Inspection Agency (CFIA). 2007. DD2007-66 Determination of the Safety of BASF's Imidazolinone-Tolerant CLEARFIELD® Wheat Event BW.
- Canadian Food Inspection Agency (CFIA). 2008. Plants of Canada Database. CFIA, Ottawa, ON.
- Canadian Food Inspection Agency (CFIA). 2008. Directive APHD-DSAE-IE-2004-62: Inspection of imported live animals - (Internal access only), semen and embryos. CFIA, Ottawa, ON.
- Canadian Food Inspection Agency (CFIA). 2008. Directive 08-04: Plant Protection Import Requirements for Plants and Plant Parts for Planting: Preventing the Entry and Spread of Regulated Plant Pests Associated with the Plants for Planting Pathway. CFIA, Ottawa, ON.
- Canadian Food Inspection Agency (CFIA). 2008. Directive 95-26: Phytosanitary requirements of soil and related matter and for items contaminated with soil and related matter. CFIA, Ottawa, ON.
- Canadian Food Inspection Agency (CFIA). 2008. Directive 97-04: Application, procedures, issuance and use of a Permit to Import under the Plant Protection Act. CFIA, Ottawa, ON.
- Canadian Food Inspection Agency (CFIA) 2008. Directive D-95-26: Phytosanitary requirements for soil and related matter, alone or in association with plants. CFIA, Ottawa, ON.
- Canadian Food Inspection Agency (CFIA) 2008. Directive 98-08: Entry Requirements for Wood Packaging Materials Produced in All Areas Other Than the Continental United States. CFIA, Ottawa, ON.
- Canadian Food Inspection Agency (CFIA) 2008. Draft Directive 08-04: Plant Protection Import Requirements for Plants and Plant Parts for Planting: Preventing the Entry and Spread of Regulated Plant Pests Associated with the Plants for Planting Pathway. CFIA, Ottawa, ON. Unpublished.
- Canadian Food Inspection Agency (CFIA) 2009. Pests Regulated by Canada (under the Plant Protection Regulations 29 (2a)). CFIA, Ottawa, ON.
- Canadian Food Inspection Agency (CFIA) 2009. D-09-XX: Interim Plant Protection Requirements for Plant Quarantine Pests in Canada. CFIA, Ottawa, ON. Taxa that are Listed as Regulated.
- Canadian Food Inspection Agency (CFIA) 2009. Pests Regulated by Canada (under the Plant Protection Regulations 29 (2a)). CFIA, Ottawa, ON.
- Canadian Food Inspection Agency (CFIA) 2009. Prohibited Plants and Plant Material from Countries Other than the Continental United States. CFIA, Ottawa, ON.
- Canadian Food Inspection Agency (CFIA) 2009. The 2009 ABCs of Seed Importation into Canada [July 2009].
- Canadian Food Inspection Agency (CFIA) 2009. Directive D-99-01: Barley, Oats, Rye, Triticale and Wheat -- Phytosanitary Requirements on Import, Transshipped, In-Transit and Domestic Movement. CFIA, Ottawa, ON.
- Canadian Grain Commission, 2007. Varieties of wheat designated as the class Canada Western Red Spring (CWRS) [July 2009].
- Canadian Honey Council 2005. PA's in some Australian honey.
- Canadian Nursery Landscape Association (CNLA) 2009. F.I.N.D. – Search for Plants. Request date: June 19, 2009.
- Canadian Seed Trade Association 2008. Seed Trade Statistics [July 2009].
- Castro, K. 2007. Weed Risk Assessment – Echium plantagineum L. (Echium plantagineum). Plant Health Risk Assessment Unit, Science Advice Division, Canadian Food Inspection Agency, Ottawa, Ontario. 48 pp.
- Castro, K. 2008. Echium plantagineum L. - Additional information pertaining to the WRA and RMD Consultation. Plant Health Risk Assessment Unit, Science Advice Division, Canadian Food Inspection Agency, Ottawa, Ontario. 28 pp.
- Castro, K. 2008. Weed risk assessment for Persicaria perfoliata (L.) H. Gross (Persicaria perfoliata). Plant and Biotechnology Risk Assessment Unit, Plant Health Science Division, Canadian Food Inspection Agency. Ottawa, ON.
- Castro, K. 2009. Senecio madagascariensis Poir (fireweed). Pest risk assessment request No. 2009-20, Plant and Biotechnology Risk Assessment Unit, Canadian Food Inspection Agency, Ottawa, ON. Unpublished report submitted to CFIA's Plant Health and Biosecurity Directorate in June, 2009.
- Castro, K. Senecio inaequidens DC. (South African ragwort) Pest risk assessment request No. 2009-20sein, Plant and Biotechnology Risk Assessment Unit, Canadian Food Inspection Agency, Ottawa, ON. Unpublished report submitted to CFIA's Plant Health and Biosecurity Directorate in May, 2009.
- Centre for Agriculture and Biosciences International (CABI). 2005. Crop Protection Compendium, Global Module, 2005 edition. Wallingford, UK. CAB International [March, 2006].
- Centre for Invasive Plant Management (CIPM). No date. Invasive Plant Information – Worst Weeds in the West [February 24, 2010].
- Charlotte, N.C. 1990. Dictionary of Fiber & Textile Technology. Hoechst Celanese Co. 218 pp.
- Clapham, A. R., Tutin, T. G., and Warburg, E. F., 1962. Flora of the British Isles. University Press, Cambridge, Great Britain.
- Clough, P. 1993. Sources and production of specialty oils containing GLA and stearidonic acid. Lipid Technol. 5(3):9–12.
- Colpetzer, K., Hough-Goldstein, J., Harkins, K. and Smith, M. 2004. Feeding and oviposition behaviour of Rhinoncomimum latipes Korotyaev (Coleoptera: Curculionidae) and its predicted effectiveness as a biological control agent for Polygonum perfoliatum L. (Polygonales: Polygonaceae). Environmental Entomology 33(4): 990-996.
- Costanza, R., and C. Perrings. 1990. A flexible assurance bonding system for improved environmental management. Ecological Economics 2, 55–57.
- Cousens, R. and M. Mortimer, 1995. Dynamics of Weed Populations. Cambridge University Press, Cambridge, UK.
- Crooks, J.A. and M.E. Soulé. 1999. Lag times in population explosions of invasive species: Causes and implications. In: Invasive Species and Biodiversity Management. Ed. O.T. Sandlund et al. Kluwer Academic Publishers. 431 pp.
- Culvenor, CCJ. 1985. Echium plantagineum and toxic alkaloids. Search 16:219-223.
Daehler, C. no date. Weed Risk Assessments for Hawaii and Pacific Islands [June 16, 2009]. - Dairy Research and Development Corporation (DRDC). 1996. Little damage likely from fireweed control, Research note 45. Dairy Research and Development Corporation, Australia.
Darbyshire, S. J., 2003. Inventory of Canadian Agricultural Weeds. Agriculture and Agri-Food Canada, Research Branch, Ottawa, Canada. - Davison, J., and Wargo, M. 2001. Syrian Beancaper: Another New Noxious Weed Threatens Nevada; Fact Sheet FS-01-46. ed. C. Extension. University of Nevada, Reno, NV.
- Dimande, A.F.P., Botha, C.J., Prozesky, L., Bekker, L., Rosemann, G.M., Lasbuschagne, L. and Retief, E. 2007. The toxicity of Senecio inaequidens DC. Journal of the South African Veterinary Association 78: 121-129.
- Distribution and Biology. Krieger Publishing Company, Malabar, Florida. 609 pp.
Donald, W. and Ogg, A. 1991. Biology and Control of Jointed Goat grass (Aegilops cylindrica) a Review. Weed Technology 5:3-17. - Douglas, G.W., Straley, G.B., and Meidinger, D., eds. 1990. The Vascular Plants of British Columbia. Part 2 - Dicotyledons (Diapensiaceae through Portulacaceae). BC Ministry of Forests.
- Douglas, G.W., Straley, G.B., Meidinger, D., and Pojar, J., eds. 1998. Illustrated Flora of British Columbia. Province of British Columbia, Ministry of Environment, Lands and Parks, Ministry of Forests.
- Duncan, C.L. 2001. Knapweed management: another decade of change. In 1st international knapweed symposium of the 21st century. ed. L. Smith. U.S. Department of Agriculture, Agricultural Research Service, Coeur d'Alene, ID. pp. 1-7.
- EPPO. 2006. EPPO data sheet on Invasive Plants: Senecio inaequidens (draft doc 05-11836), PRA Documents. [October 7, 2008].
- EPPO. 2006. Eradication project of Senecio inaequidens in Corse, France. 2006/045. EPPO Reporting Service 2006(2): 19.
- EPPO. 2006. Pest risk analysis for Senecio inaequidens (Cav.) (PRA 06-12954), PRA Documents [October 7, 2008].
- EPPO. 2007. Data sheets on quarantine pests. Solanum elaeagnifolium. EPPO Bulletin 37 (2): 236-245.
- EPPO. 2007. Report of a Pest Risk Analysis. Polygonum perfoliatum L. (POLPF).
- EPPO. 2008. Distribution maps for Quarantine pests for Europe [October 7, 2008].
- EPPO. 2008. EPPO Alert: Microstegium vimineum (Poaceae).
- Ernst, W.H.O. 1998. Invasion, dispersal and ecology of the South African neophyte Senecio inaequidens in The Netherlands: from wool alien to railway and road alien. Act a Bot. Neer l. 47: 131-151
- European Plant Protection Organization (EPPO). 2009. EPPO Alert List [July 2009].
- Factsheet – Paspalum dilatatum. Tropical forages. Federal Noxious Weed Act. U.S. Public Law 93-629.
- Feeds Act R.S., 1985, c. F-9.
- Feeds Regulations, 1983 (SOR/83-593).
- Fernald, M.L. 1950. Gray's Manual of Botany. American Book Co. 1632 pp.
- Finnoff, D., J. F. Shogren, B. Leung, and D. Lodge. 2005. The importance of bioeconomic feedback in invasive species management. Ecological Economics 52:367–381.
- Finnoff, D., Shogren, J.F., Leung, B. and Lodge, D.M. 2007. Take a Risk-Preferring Prevention over Control of Biological Invaders, Ecological Economics In press.
- Flora of North America Editorial Committee (FNA Committee). 1993+. Flora of North America North of Mexico. 12+ vols. Vol. 1, 1993; vol. 2, 1993; vol. 3, 1997; vol. 4, 2003; vol. 5, 2005; vol. 19, 2006; vol. 20, 2006; vol. 21, 2006; vol. 22, 2000; vol. 23, 2002; vol. 25, 2003; vol. 26, 2002. New York and Oxford.
- FNA Editorial Committee (ed.) 1993+. Flora of North America North of Mexico, New York and Oxford. pp.
- Food and Drug Regulations (C.R.C., c. 870).
- Food and Drugs Act (R.S., 1985, c. F-27 ).
- Gaines, T., Preston, C., Byrne, P., Henry, W.B. and Westra, P. 2007. Adventitious Presence of Herbicide Resistant Wheat in Certified and Farm-Saved Seed Lots. Crop Science 47:751-754.
- Gaines, T.A., Henry, W. B., Byrne, P. F., Westra, P., Nissen, S. J. and Shaner, D. L. 2008. Jointed Goatgrass (Aegilops cylindrica) by Imidazolinone-Resistant Wheat Hybridization under Field Conditions. Weed Science 56:32–36.
- Garcia-Serrano, H., Escarré, J. and Sans, F.X. 2004. Factors that limit the emergence and establishment of the related aliens Senecio inaequidens and Senecio pterophorus and the native Senecio malacitanus in Mediterranean climate. Canadian Journal of Botany 82: 1346-1355.
- Garnatje, T., Vilatersana, R., Roché, C.T, Garcia-Jacas, N, Susanna, A., Thill, D.C. 2002. Multiple introductions from the Iberian Peninsula are responsible for invasion of Crupina vulgaris in western North America. New Phytologist 154(2): 419-428.
- Garry Oak Ecosystems Recovery Team (GOERT). 2009. Garry Oak Ecosystems Recovery Team Website, [July 2009].
- GBIF (Global Biodiversity Information Facility), 2008. Online biodiversity data.
- Georgia Exotic Pest Plant Council (GEPPC), 2006. List of Non-native Invasive Plants in Georgia: [cited 2009].
- Gerlach Okay, J.A., Hough-Goldstein, J. and Swearingen, J.M. 2007. Mile-a-Minute Weed.
- Gleason, H.A. 1968. The New Britton and Brown Illustrated Flora of the Northeastern United States and Adjacent Canada. Hafner Publishing Co., Inc., NY. 655 pp.
- Gleason, H.A., and Cronquist, A. 1963. Manual of Vascular Plants of Northeastern United States and Adjacent Canada. D. Van Nostrand Co. Inc., Princeton, NJ.
- Global Invasive Species Database(GISD), 2008. Invasive Species Specialist Group, [2008].
- Gmira, N., Douira, A., and Bouhacje, M. 1998. Ecological grouping of Solanum elaeagnifolium: A principal weed in the irrigated Tadla plain (central Morocco). Weed Research 38: 87.
- Gould, F. W. and Shaw, R. B. 1983. Grass Systematics. 2nd Edition. Texas A & M University Press, College Station, TX.
- Government of Alberta Agriculture and Rural Development (GAARD), 2007. Alberta Certified Weed Free Hay Program, available online [July 2009].
- Government of Canada. 2004. An Invasive Alien Species Strategy for Canada. Government of Canada. 46 pp.
- Government of Japan. 2006. List of Regulated Living Organisms Under the Invasive Alien Species Act (PDF, 71 kb) [2009].
- Graham, J., and Johnson, W.S. 2003. Managing Purple and Iberian Starthistles[April 21, 2009].
- Groves, R.H. 2006. Are some weeds sleeping? Some concepts and reasons. Euphytica 148: 111–120.
- Guadagnuolo, R., Savova-Bianchi, D., and Felber, F. 2001. Gene flow from wheat (Triticum aestivum L.) to jointed goatgrass (Aegilops cylindrica Host.), as revealed by RAPD and microsatellite markers. Theor. Appl. Genet. 103: 1–8.
- Guil-Guerrero, J. L., 2007. Stearidonic acid (18:4n-3): Metabolism, nutritional importance, medical uses and natural sources. European Journal of Lipid Science and Technology. 109: 1226-1236.
- Guil-Guerrero, J. L., García-Maroto, F., Vilches-Ferrón, M. A., and López-Alonso, D. 2003. Industrial Crops and Products. 18: 85-89.
- Güner, A. 1984. A new record for the Flora of Türkiye and a new subspecies from Anatolia. Candollea 39(1): 345-348.
- Haber, E., 2006. Jointed Goatgrass (Triticum cylindricum) in Canada: An Overview of its Occurrence and Potential Control. Prepared for the CFIA Plant Health Division, Invasive Alien Species Section, Ottawa, ON.
- Häfliger, E. and Scholtz, H. 1980. Grass Weeds 2: Weeds of the subfamilies Chloridoideae, Pooideae, Oryzoideae. CIBA-GEIGY, Basel, Switzerland.
- Hamill, A, Research Scientist, Sustainable Production Systems, Agriculture and Agri-Food Canada, 2005, pers. comm.
- Hammon, R., Armstrong, S., Meyer, W., Walker, D. and Peairs, F. 1989. Alternate hosts of the Russian wheat aphid in Colorado. Proc. Russian Wheat Aphid Conf. Alburquerque, NM p. 4-7.
- Hanf, M. 1983. The arable weeds of Europe with their seedlings and seeds. BASF Aktiengesellschaft, Ludwigshafen.
- Hanson, B., Mallory-Smith, C., Price, W., Shafii, B., Thill, D., and Zemetra, R. Interspecific hybridization: Potential for movement of herbicide resistance from wheat to jointed goat grass (Aegilops cylindrica). Weed Technology 19:674-682.
- Hardin, J. W., Doerksen, G., Herndon, D., Hobson, M., and Thomas, F. 1972. Pollination ecology and floral biology of four weedy genera in southern Oklahoma. The Southwestern Naturalist 16(3/4): 403-412.
- Health of Animals Act (1990, c. 21).
- Health of Animals Regulations, C.R.C., c.296.
- HEAR. 2003. Hawaiian Ecosystems at Risk Project.
- Noxious weeds - Hawaii. Images and online info. [2009].
- Heger, T., and Böhmer, H.J. 2006. NOBANIS - Invasive Alien Species Fact Sheet - Senecio inaequidens. Online database of the North European and Baltic Network on Invasive Alien Species [May 22, 2009].
- Hill, R.J., Springer, G. and Forer, L. 1981. Mile-a-minute, Polygonum perfoliatum L. (Polygonaceae), a new potential orchard and nursery weed. Regulatory Horticulture Weed Circular 7:25-28.
- Hinds, H.R., and Freeman, C.C. 1993. Persicaria (Linnaeus) Miller. In Flora of North America North of Mexico. Edited by Flora Of North America Editorial Committee, New York and Oxford.
- Holm, L., D. L. Plucknett, J. V. Pancho, and J. P. Herberger, 1991. The World's Worst Weeds,
Holm, L., Doll, J., Holm, E., Pancho, J. and Herberger, J. 1997. World Weeds, Natural Histories and Distribution. John Wiley & Sons Inc., New York, NY. 1129 pp. - Holm, L., Pancho, J. V., Herberger, J. P. and Plucknett, D. L. 1991. A Geographical Atlas of World Weeds. Krieger Publishing Co., Malabar, FL.
- Holm, L., Plucknett, D. L., Pancho, J. V. and Herberger, J. P. 1991. The World's Worst Weeds, Distribution and Biology. Krieger Publishing Company, Malabar, FL. 609 pp.
- Horan, R.D. and F. Lupi. 2005. Economic Incentives for Controlling Trade-Related Biological Invasions in the Great Lakes. Agricultural and Resource Economics Review 34(1): 75-89.
- Horan, R.D. and F. Lupi. 2005. Tradable Risk Permits to Prevent Future Introductions of Invasive Alien Species into the Great Lakes. Ecological Economics 52(3): 289-304.
- Horton, P.R. 1996. The Rangelands of British Columbia. British Columbia Ministry of Forests, Research Branch[October 2009].
- Hough-Goldstein, J. 2008. Biological control of mile-minute weed [August 28 2009].
- Hough-Goldstein, J. and Lake, E. 2008. New developments in biological control of mile-a-minute weed. Northern Journal of Applied Forestry 25(3): 164-165.
- Hough-Goldstein, J., Lake, E., Reardon, R., and Wu, Y. 2008. Biology and Biological control of Mile-a-Minute Weed. Forest Health Technology Enterprise Team. Report FHTET-2008-10. November 2008.
- Howard, J.L. 2005. Microstegium vimineum. In: Fire Effects Information System, [Online]. U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station, Fire Sciences Laboratory (Producer).
- Hubbard, C.E. 1968. Grasses, a guide to their structure, identification, uses, and distribution in the British Isles. Penguin Books Ltd., Bungay, Suffolk, England.
- Idaho State Department of Agriculture (ISDA). 2005. Idaho's strategic plan for managing noxious invasive weeds. ISDA, Meridian, ID.
- Idaho State Department of Agriculture (ISDA). 2009. Idaho's 57 Noxious Weeds [October 2009].
- IENICA, 2000. IENICA Summary Report for the European Union.
- IENICA, 2002. Viper's bugloss and purple viper's bugloss (Echium). Summary report for the European Union.
- Industry Canada. 2007. Trade Data Online (TDO). Data from Statistics Canada and the U.S. Census Bureau (U.S. Department of Commerce)[2007].
- Industry Canada. 2009. Trade Data Online (TDO). Data from Statistics Canada and the U.S. Census Bureau (U.S. Department of Commerce)[2009].
- Industry Canada. 2011. Trade Data Online (TDO). Data from Statistics Canada and the U.S. Census Bureau (U.S. Department of Commerce)[December 2011].
- International Plant Protection Convention (IPPC). 2006. International Standards for Phytosanitary Measures 1 to 27 (2006 Edition). Secretariat of the International Plant Protection Convention, Food and Agriculture Organization of the United Nations, Rome, Italy.
- International Plant Protection Convention (IPPC). 2007. Glossary of Phytosanitary Terms.
- International Standard for Phytosanitary Measures No. 5. IPPC, Rome, Italy.
- International Rice Research Institute, 2009. Rice Around the World – Rice in 54 Other Countries[June 18, 2009].
- Invasive Plant Council of BC (IPCBC). 2007. Invasive plant early detection and rapid response in British Columbia – draft version 7 (November 2007). IPC of BC, Williams Lake, BC.
- Isaacson, R.T., and Allen, K. 2007. Plant Information Online. University of Minnesota Libraries. Minnesota [2009].
- ISPM No. 2. 2007. Framework for Pest Risk Analysis. FAO, Rome.
- ISPM No. 5. 2009. Glossary of Phytosanitary Terms. FAO, Rome.
- ISSG, 2009. Global Invasive Species Database. [cited April 2009].
- Johnson Jr., C.F. 1996. Achene germination requirements, temporal viability and germination when stored under natural conditions, and abundance in the soil seed bank for the exotic invasive mile- a-minute (Polygonum perfoliatum L.), School of Forest Resources, Pennsylvania State University.
- Judge, C.A., et al. 2008. Japanese Stiltgrass (Microstegium vimineum) Management for Restoration of Native Plant Communities. Invasive Plant Science and Management 1: 111–119.
- Jung, M.J., Yang, S.Z. and Kuoh, C.S. 2005. Notes on two newly naturalized plants in Taiwan. Taiwania 50(3): 191-199.
- Karl, M.G., Leonard, S.G., Rice, P.M., and Rider, J. 1996. Noxious Weeds in the Interior Columbia Basin and Portions of the Klamath and Great Basin: Science Assessment of Selected Species. pp. 1-120.
- Kartesz, J. T. 1999. Synthesis of the North American Flora Version 1.0 - A synonomized checklist and atlas with biological attributes for the vascular flora of the United States, Canada, and Greenland. North Carolina Botanical Garden, Chapel Hill, NC.
- Kartesz, J.T. 1999. A Synonymized Checklist and Atlas with Biological Attributes for the Vascular Flora of the United States, Canada, and Greenland. First Edition. In: Kartesz, J.T., and C.A. Meacham. Synthesis of the North American Flora, Version 1.0. North Carolina Botanical Garden, Chapel Hill, NC.
- Kartesz, J.T., and Meacham, C.A. 1999. Synthesis of the North American Flora, Version 1.0.
- Kentucky Exotic Plant Pest Council (KY-EPPC), 2008.
- Klassen, D., 2006. Winter wheat acreage in W. Canada Jumps from Yr Ago Level. Resource News International, Trading charts.
- Klemow, K.M., D.R. Clements, P.F. Threadgill, and P.B. Cavers. 2002. The biology of Canadian weeds. 116. Echium vulgare L. Can. J. Plant Sci. 82: 235–248.
- Klinkenberg, B. 2008.E-flora BC: An electronic atlas of the plants of British Columbia. [Online database] Available: Lab for Advanced Spatial Analysis, Department of Geography, University of British Columbia, Vancouver.
- Knowler, D., and E. Barbier. 2005. Importing exotic plants and the risk of invasion: are market-based instruments adequate? Ecological Economics 52:341–354.
- Koike, F., Clout, M.N., Kawamichi, M., De Poorter, M. and Iwatsuki, K.. 2006. Alien species and wild flora. In Assessment and Control of Biological Invasion Risks. Edited by F. Koike, M.N. Clout, M. Kawamichi, M. De Poorter, and K. Iwatsuki.
- Shoukadoh Book Sellers, Kyoto, Japan and IUCN, Gland, Switzerland.
- Kourtev, P.S., Ehrenfeld, J.G., and Huang, W.Z. 1998. Effects of exotic plant species on soil properties in hardwood forests of New Jersey. Water, Air and Soil Pollution. 105 (1-2): 493-501.
- Kumar, V. and Ditommaso, A. 2005. Mile-a-Minute (Polygonum perfoliatum): An increasing problematic invasive species. Weed Technology 19: 1071-1077.
- Lafuma, L., Balkwill, K., Imbert, E., Verlaque, R. and Maurice, S. 2003. Ploidy level and origin of the European invasive weed Senecio inaequidens (Asteraceae). Plant Systematics and Evolution 243: 59-72.
- Lass, L., and Prather, T. 2007. A Scientific Evaluation for Noxious and Invasive Weeds of the Highway 95 Construction Project between the Uniontown Cutoff and Moscow. AquilaVision Inc., Missoula, MT.
- Lefèvre, I., Corréal, E., Lutts, S. 2006. Cadmium tolerance and accumulation in the noxious weed NRC Research Press Zygophyllum fabago [June 2009].
- Lehtonen, P. 1994. Pest risk assessment for mile-a-minute weed. U.S. Department of Agriculture, Animal and Plant Health Inspection Service, Plant Protection and Quarantine, Hyattsville, Maryland, USA.
- Looman, J., and Best, K.F. 1987. Budd's Flora of the Canadian Prairie Provinces. Minister of Supply and Services Canada. Research Branch Agriculture Canada. Publication 1662., Hull, QC.
- López, M.G., Wulff, A.F., Poggio, L. and Xifreda, C.C. 2008. South African fireweed Senecio madagascariensis (Asteraceae) in Argentina: relevance of chromosome studies to its systematics. Botanical Journal of the Linnean Society 158: 613-620.
- López-García, M.C., and Maillet, J. 2005. Biological characteristics of an invasive south African species. Biological Invasions 7: 181-194.
- Maiti, P. C., and Mathew, R. 1967. Rich sources of solasodine. Current Science 36(26).
- Mallory-Smith, C., 2001. Survey of Oregon Wheat Fields to Identify Hybrids Between Wheat and Jointed Goatgrass. Oregon Wheat March 2001: 8-9.
- McCartney, D. and Horton, P.R. 1997. Canada's Forage Resources. Report for the International Grasslands Congress, available online (PDF, 111 kb) [July 2009].
- McCormick, L.H. and Hartwig, N.L. 1995. Control of the noxious weed mile-a-minute (Polygonum perfoliatum) in reforestation. Northern Journal of Applied Forestry 12: 127-132.
- Mehrhoff, L. J., Silander, J.A. Jr., Leicht, S.A., Mosher, E.S., and Tabak, N.M. 2003. IPANE: Invasive Plant Atlas of New England. Department of Ecology & Evolutionary Biology, University of Connecticut, Storrs, CT, [2008].
- Mehrhoff, L.J. 2000. Perennial Microstegium vimineum (Poaceae): An Apparent Misidentification? Journal of the Torrey Botanical Society 127(3): 251-254.
- Mekki, M. 2007. Biology, distribution and impacts of silverleaf nightshade (Solanum elaeagnifolium Cav.). EPPO Bulletin 37(1): 114-118.
- Mendez, M. D., Rietcorrea, F., Schild, A. L. and Garcia, J. T. C., 1985. Poisoning by Echium plantagineum (Boraginaceae) in Cattle in Southern Brazil. Pesquisa Veterinaria Brasileira. 5(2): 57-64.
- Mondragón Pichardo, J., and Vibrans, H. 2005. Malezas de México. Senecio inaequidens DC. Manzanilla de llano (Spanish) [October 7 2008].
- Montana Department of Agriculture. 2009. Montana Noxious Weed Program – Noxious Weed List, available online Request date: October 2009.
- Morrison, L., Cremieux, L. and Mallory-Smith, C. 2002. Infestations of jointed goat grass (Aegilops cylindrica) and its hybrids in wheat in Oregon wheat fields. Weed Science 50:737-747.
- Moss, E.H. 1959. Flora of Alberta; a manual of flowering plants, conifers, ferns and fern allies found growing without cultivation in the province of Alberta, Canada. University of Toronto Press, Toronto, ON. 346 pp.
- Mountain. W.L. 1989. Mile-a-Minute (Polygonum perfoliatum L.) update – distribution, biology, and control suggestions.
- Regulatory Horticulture Circular No. 15. Pennsylvania Department of Agriculture Bureau of Plant Industry, Fall 1989.
- NAPPO (North American Plant Protection Organization), 2003. Aegilops cylindrica Host. PRA/Grains Panel Pest Facts Sheet. June 2003.
- Newmaster, S.G., Lehela, A., Uhlig, P.W.C., McMurray, S., and Oldham, M.J. 1998. Ontario Plant List; Forest Research Information Paper No. 123. Ontario Forest Research Institute, Ontario Ministry of Natural Resources, Sault Ste. Marie, ON. 550 + appendices pp.
- NJGRP (National Jointed Goatgrass Research Program). 2006.
- Noxious Weed Control Board – Washington State (NWCB). 2009. Washington State noxious weed list – Class B weeds [October 2009].
- NRC(Natural Resources Canada), 2000. Canada Plant Hardiness Zones (map) available online.
- ODA (Oregon Department of Agriculture), 2007. Echium plantagineum. Distribution in Oregon.
- ODA (Oregon Department of Agriculture). 2004. Pest Risk Assessment for Echium plantagineum. Prepared by G. Miller and T. Forney. Noxious Weed Control Program. February 2004.
- Okay, J.A.G. 1997. Polygonum perfoliatum: A Study of Biological and Ecological Features Leading to the Formation of a Management Policy. Ph.D. Thesis. George Mason University, Fairfax, VA. 132 pp.
- Oldham, M. and Brinker, S. 2009. Targeted Field Surveys for Jointed Goat grass (Aegilops cylindrica) in Niagara Region, Ontario, in 2008. Final Report for the Canadian Food Inspection Agency, Ottawa, ON.
- Oliver, J.D. 1996. Mile-a-Minute weed (Polygonum perfoliatum L.), an invasive vine in natural and disturbed sites. Castanea 61: 244-251.
- Ontario Ministry of Agriculture, Food and Rural Affairs (OMAFRA) 2006. Ontario Census Farms 2006. Statistics Unit, Agriculture and Economic Development Policy Branch[August 2009].
- Ontario Ministry of Agriculture, Food and Rural Affairs (OMAFRA), 2009. Field Crop Statistics [cited 2009].
- Ontario Ministry of Agriculture, Food and Rural Development. (OMAFRA). 2009. Grapes: Area, Production, and Farm Value by County and District, Ontario, 2007 [cited July 2009].
- Oregon Department of Agriculture (ODA). 2006. Quarantine; noxious weeds, Chapter 603-52-1200 in Oregon Administrative Rules. Oregon Department of Agriculture [August 2009].
- Oregon Department of Agriculture (ODA). 2007. Iberian starthistle [April 2009].
- Oregon Department of Agriculture (ODA). 2009. Noxious weed policy and classification system 2009. ODA, Salem, Oregon.
- Oswalt, C.M., Oswalt, S.N., and Clatterbuck, W.K. 2007. Effects of Microstegium vimineum (Trin.) A. Camus on native woody species density and diversity in a productive mixed-hardwood forest in Tennessee. Forest Ecology and Management 242: 727-732.
- Pankhurst, R. 1998. Flora Europaea. Royal Botanic Garden Edinburgh [May 2009].
- Park, C. 1986. Nomenclatural typifications in Polygonum section Echinocaulon (Polygonaceae). Brittonia 38: 394-406.
- Parsons, W. T., and Cuthbertson, E. G. 2001. Noxious Weeds of Australia, Second Edition. CSIRO Publishing, Collingwood, Victoria, Australia.
- Patterson, C.A. 2006. Gamma-Linolenic Acid: Healthy Canadian Ingredient. July 2006. AAFC No. 10057E.
- Peirce, J. 2006. How to control Echium plantagineum. Farmnote 169. The State of Western Australia.
- Peterson, J.E. 1985. The toxicity of Echium plantagineum (Echium plantagineum). Plant toxicology Proceedings of the Australia-USA poisonous plants symposium. Brisbane, Australia, May 14-18, 1984. pp. 191-199.
- Pheloung, P.C., Williams, P.A. and Halloy, S.R. 1999. A Weed Risk Assessment Model for Use as a Biosecurity Tool Evaluating Plant Introductions. Journal of Environmental Management 57: 239-251.
- Plant Conservation Alliance's Alien Plant Working Group, Weeds Gone Wild: Alien Plant Invaders of Natural Areas. 4 pp.
- Plant Protection Act (1990, c. 22).
- Plant Protection Regulations, SOR/95-212.
- Plant, R., and Robertson, S. 2007. Fireweed. Senecio madagascariensis. Victorian Alert Weed Fact Sheet. State of Victoria Department of Primary Industries [June 15, 2009].
- Potter, B. 2004. Managing Forage Supplies. Ministry of Agriculture, Food and Rural Affairs of Ontario. Last reviewed 09 June 2009 [cited August 2009].
- Preston, C. 2006. Resistance to acetolactate synthase-inhibiting herbicides in Echium plantagineum. Papers and Proceedings of the 15th Australian Weeds Conference: Managing Weeds in A Changing Climate, 24-28 September 2006, Adelaide, South Australia. pp. 530-533.
- Price, D.L, Hough-Goldstein, J. and Smith, M.T. 2003. Biology, rearing, and preliminary evaluation of host range of two potential biological control agents for mile-a-minute weed, Polygonum perfoliatum L. Environmental Entomology 32: 229-236.
- Queensland Government. 2007. Fact sheet: Invasive Plants and Animals. Fireweed. Senecio madagascariensis. Declared Class 2. Edited by Department of Primary Industries and Fisheries. Land Protection (Invasive Pland and Animals).
- Radford, I.J., Muller, P., Fiffer, S. and Michael, P.W. 2000. Genetic relationships between Australian fireweed and South African and Madagascan populations of Senecio madagascariensis Poir. and closely related Senecio species. Australian Systematic Botany 13(3): 409-423.
- Randall, J.M., Morse, L. E., Benton, N., Hiebert, R., Lu, S. and Killeffer, T. 2008. The Invasive Species Assessment Protocol: A Tool for Creating Regional and National Lists of Invasive Nonnative Plants that Negatively Impact Biodiversity. Invasive Plant Science and Management 1:36–49.
- Randall, R. P., 2002. A Global Compendium of Weeds. R.G. and F.J. Richardson, Meredith, Australia.
- Reinhardt, F., Herle, M., Bastiansen, F. and Streit, B. 2003. Economic impact of the spread of alien species in Germany. Texte des Umweltbundesamtes 2003(80).
- Retief, E. and Van Wyk, A. E., 1998. The genus Echium (Boraginaceae) in southern Africa. Bothalia. 28(2): 167-177.
- Rice, P. 1997-2008. Invaders Database System [cited 2008].
- Robbins, W.W., Bellue, M.K., and Ball, W.S. 1951. Weeds of California. California State Department of Agriculture, Sacramento, CA. 547 pp.
- Rotar, P.P. 1968. Grasses of Hawaii. University of Hawaii. Honolulu. 355 pp.
- Roy, B., Popay, I., Champion, P., James, T., Rahman, A. 1998. An illustrated guide to common weeds of New Zealand. New Zealand Plant Protection Society, Canterbury, New Zealand.
- Ruiz, G.M., and Carlton, J.T. 2003. Invasive Species Vectors and Management Strategies. Island Press, Washington, DC. 518 pp.
- Santos, J. C. A., Riet-Correa, F., Simoes, S. V. D., and Barros, C. S. L., 2008. Pathogenesis, clinical signs and pathology of diseases caused by hepatotoxic plants in ruminants and horses in Brazil. Pesquisa Veterinaria-Brasileira. 28(1): 1-14.
- Scherber, C., Crawley, M.J. and Porembski, S. 2003. The effects of herbivory and competition on the invasive alien plant Senecio inaequidens (Asteraceae). Diversity and Distributions 9(6): 415-426.
- Schmale, D., Lodgepole, Anderson, R., Lyon, D., and Klein, B. 2008. Jointed Goatgrass Best Management Practices Central Great Plains: a multi-practice guide for profitable wheat production. Washington State University, WA.
- Scoggan, H. 1979. Flora of Canada. National Museum of Natural Sciences, Ottawa, ON. Publications in Botany, No. 7(4).
- Seeds Act, (R.S., 1985, c. S-8).
- Seeds Regulations (C.R.C., c. 1400)
- Sexton, P. 2007. Recommendation regarding management of Echium plantagineum in Aroostook County, Maine. University of Maine Cooperative Extension. E-mail correspondence. 12 July 2007.
- Shafi, B., Price, W.J., Prather, T.S., Lass, L.W., and Thill, D.C. 2003. Predicting the likelihood of yellow starthistle (Centaurea solstitialis ) occurrence using landscape characteristics. Weed Science 51: 748-751.
- Sheppard, A. and M. Smyth. 2002. Predicting seedbank decay rates: the effects of field conditions on seed longevity and seedling recruitment in Echium plantagineum L. 13th Australian Weeds Conference. Perth, Australia. 8-13 September 2002. pp. 541-544.
- Sindel, B.M., and Michael, P.W. 1992. Spread and potential distribution of Senecio madagascariensis Poir (fireweed) in Australia. Australian Journal of Ecology 17: 21-26.
- Sindel, B.M., Radford, I.J., Holtkamp, R.H. and Michael, P.W. 1998. The biology of Australian weeds. 33. Senecio madagascariensis Poir. Plant Protection Quarterly 3: 22-28.
- Sissons, A. 2009. Microstegium vimineum (Trin) A. Camus (Japanese stiltgrass) Pest risk assessment request No. 2009-20mivi, Plant and Biotechnology Risk Assessment Unit, Canadian Food Inspection Agency, Ottawa, ON. Unpublished report submitted to CFIA's Plant Health and Biosecurity Directorate in May, 2009.
- South Carolina Exotic Plant Pest Council (SC-EPPC), 2008. Invasive Species List 2008[cited 2009].
- Starr, F., Martz, K. and Loope, L. 1999. Fireweed (Senecio madagascariensis): An Alien Plant Report. USGS Biological
- Resources Division, American Water Works Association Research Foundation, Maui County Board of Water Supply.
- State of Nevada. 2003. Chapter 555 - Control of insects, pests, and noxious weeds in Nevada Administrative Code. State of Nevada [August 2009].
- Statistics Canada. 1996. 1996 Census of Agriculture. Statistics Canada, Ottawa, ON. Request date: August 2009.
- Statistics Canada 2002. Farm Operator Data for the 2001 Census of Agriculture. Catalogue no. 95F0355XIE. Statistics Canada [August 2009].
- Statistics Canada, 2007. 2006 Census of Agriculture, Farm Data and Farm Operator Data, catalogue number 95-629-XWE[October 2009].
- Statistics Canada. 2008. The Daily: Characteristics of International Travellers. Statistics Canada, Ottawa, ON [June 2009].
- Statistics Canada. 2009. CATS Database. Unpublished Data. Statistics Canada, Ottawa, ON.
- Steward, A.N. 1930. Contributions from the Gray Herbarium of Harvard University. LXXXVIII. The Polygoneae of Eastern Asia. The Gray Herbarium of Harvard University, Cambridge, MA.
- Stone A. E. and Peeper, T. E. 2004. Characterizing jointed goatgrass (Aegilops cylindrica) x winter wheat hybrids in Oklahoma. Weed Science 52: 742-745.
- Stubbendieck, J., Friisoe, G., and Bolick, M. 1995. Weeds of Nebraska and the Great Plains. Nebraska Department of Agriculture. Lincoln, NB.
- Sutherst, R. W., Maywald, G. F., and Kriticos, D. J., 2006. CLIMEX Version 3 User's Guide. CSIRO, Australia.
- Swearingen, J.M. 2004. Weed US: Database of Plants Invading Natural Areas in the United States of America
- Synder, J., Mallory-Smith, C., Balter, S., Hansen, J. and Zemetra, R. 2000. Seed production on Triticum aestivum by Aegilops cylindrica hybrids in the field. Weed Science 48:588-593.
- Tan, S.Y., Evans, R.R., Dahmer, M.L., Singh, B.K. and Shaner, D.L. 2005. Imidazolinone-tolerant crops: History, current status, and future. Pest Manage. Science. 61:246–257.
- Tennessee Exotic Pest Plant Council (TN-EPPC), 2001. Tennessee Invasive Exotic Plant List [cited 2009].
- Thill, D.C., Roché, C.T., Zamora, D.L. 1999. Common Crupina. in: R. L. Sheley and J. K. Petroff, eds, Biology and Management of Noxious Rangeland Weeds. Oregon State University Press, Corvallis, OR. Pp. 189-201.
- Thomas, G. and Leeson, J. 2007. Tracking long-term changes in the arable weed flora of Canada. Pages 43-69 in D.R.
- Clements and S.J. Darbyshire, eds. Invasive Plants: Inventories, strategies and action. Topics in Canadian Weed Science, Vol. 5. Sainte Anne de Belluevue, Quebec: Canadian Weed Science Society.
- Thomas, K., Wilson, C. and Castro, K. 2007. Weed Risk Assessment – Jointed goat grass (Aegilops cylindrica Host). Plant Health Risk Assessment Unit, Science Advice Division, Canadian Food Inspection Agency, Ottawa, ON. 39 pp.
- Thompson, A.J., Jones, N.E. and Blair, A.M. 1997. The effect of temperature on viability of imbibed weed seeds. Annals of Applied Biology 130: 123 - 134.
- Tropicos. 2008. Tropicos.org [On-line database]. Missouri Botanical Garden, St. Louis, MO.
- Tsvelev, N. N., 1984. Grasses of the Soviet Union. Part I. A. A. Balkema, Rotterdam, The Netherlands.
- Tu, M. 2000. Elemental Stewardship Abstract for Microstegium vimineum. The Nature Conservancy, Arlington, VA.
- Tu, M. 2002. Element Stewardship Abstract for Dioscorea oppositifolia L. [cited May 2009].
- Tutin, T. G., Heywood, V. H., Burges, N. A., Moore, D. M., Valentine, D. H., Walters, S. M. and Webb, D. A. eds. 1980.
- Flora Europaea. Vol. 5. Alismataceae to Orchidaceae (Monocotyledons). Cambridge University Press, Cambridge, United Kingdom.
- UK Advisory Committee on Novel Foods and Processes. 2007. Initial Opinion on an Application under the Novel Foods Regulation for Refined Echium Oil Derived from Echium plantagineum as a Food Ingredient.
- United States Department of Agriculture – Agricultural Research Service (USDA-ARS), 2009. Germplasm Resources Information Network (GRIN) [2009].
- United States Department of Agriculture – Animal and Plant Health Inspection Service (USDA-APHIS), 2006. Federal Noxious Weed List, Pests and Diseases [2009].
- United States Department of Agriculture – Natural Resources Conservation Service (USDA-NRCS), 2009. The PLANTS database. National Plant Data Center, Baton Rouge, LA. [2009].
- United States Department of Agriculture (USDA) 2006. World Agricultural Production. United States Department of Agriculture, Foreign Agricultural Service. Circular Series WAP 10-08.
- United States Department of Agriculture, Agricultural Research Service (USDA-ARS) 2009. Germplasm Resources Information Network - (GRIN)[April, 2009].
- United States Department of Agriculture, Animal and Plant Health Inspection Service (USDA, APHIS). 2009. USDA Plant Import Plant Health Import Information [2009].
- United States Department of Agriculture, Foreign Agricultural Service (USDA), 2004. Map of Wheat Area Distribution in Canada. Production Estimates and Crop Assessment Division, Foreign Agricultural Service, USDA [March 2010].
- United States Department of Agriculture, Natural Resources Consercation Service. 2009. The PLANTS database. National Plant Data Center, Baton Rouge, LA [January 01, 2009].
- United States Department of Agriculture, Natural Resources Conservation Service. 2009. The PLANTS database. National Plant Data Center, Baton Rouge, LA [cited 2009].
- United States Department of Agriculture-Agriculture Research Service (USDA-ARS). 2009. Germplasm Resources Information Network - (GRIN) [January 01,2009].
- University of British Columbia Botanical Garden (UBCBG). 2006. Tender Plants available at 2008 UBC BG Perennial Plant Sale (PDF, 71 kb). University of British Columbia Botanical Garden. Vancouver (BC). 3 pages.
- USDA, NRCS. 2009. The PLANTS database. National Plant Data Center, Baton Rouge, LA 70874-4490 USA [cited 2009].
- USDA-ARS. 2009. Germplasm Resources Information Network - (GRIN) [cited 2009].
- Uygur, S. 2001. Importance and Distribution of Centaurea Species in Türkiye. in The First International Knapweed Symposium of the Twenty-First Century. L. Smith, ed. United States Department of Agriculture, Agricultural Research Service, Coeur d'Alene, ID.
- Vere, D. T. and Campbell, M. H. 1984. Economics of controlling serrated tussock in the southeastern Australian rangelands. Journal of Range Management 37(1):87-93.
- Walters C. G., Young, D. L. and Young, F. L. 2003. Economics of Alternate Control Practices for Jointed Goatgrass in Winter Wheat in the Pacific Northwest. Washington State University, Department of Crop and Soil Sciences, Technical Report 03-2.
- Wang, Z.N., Zemetra, R.S., Hansen, J. and Mallory-Smith, C.A. 2001. The fertility of wheat x jointed goatgrass hybrid and its backcross progenies. Weed Science 49:340–345.
- Washington State Noxious Weed Control Board (NWCB). Washington State noxious weed list – Class A weeds [Cited August 2009].
- Watson, R., Allan, H., Munnich, D., Launders, T. and Macadam, J. 1997. AG facts - Fireweed. p. 7.6.26. New South Wales Agriculture, Document 00758, electronic edition, Australia.
- Weber, E. 2003. Invasive plant species of the world: A reference guide to environmental weeds. CABI Publishing, Wallingford, UK.
- Weed Seeds Order, 2005 (SOR/2005-220).
- Westbrooks, R. and Cross, G. 1993. Serrated Tussock (Nasella trichotoma) in the United States. Weed Technology 7:525-529.
- White, T. 2004. Can Jointed Goat grass Invade Pastures? National Jointed Goatgrass Research Program, available online
- Wicks, G., Anderson, R., White, T., Stahlman, P. and Morishita, D. 2004. Jointed Goatgrass Control Tactics.
- Wilson, C. 2009. Solanum elaeagnifolium Cav. (Silverleaf nightshade) pest risk assessment request No. 2009-20, Plant Health Risk Assessment Unit, Canadian Food Inspection Agency, Ottawa. Unpublished report submitted to CFIA's Plant Health and Biosecurity Directorate in June, 2009.
- Wines of Ontario. 2009. The Wine Regions. Wine Council of Ontario [August 2009].
- Wood, H. and R. Degabriele. 1985. Genetic variation and phenotypic plasticity in populations of Paterson's curse (Echium plantagineum L.) in south-eastern Australia. Australian Journal of Botany, 33:677-685.
- Wooten, G. 2010. Floristic Survey for Okanogan County, Washington. Twisp, WA.
- WSU Extension. 2007. National Jointed Goatgrass Research Program [July 2009].
- Wu, Y., Reardon, C. and Jian-quig, D. 2002. Mile-a-minute weed. Pages 331-341 In Van Driesche, R., S. Lyon, B. Blossey, M. Hoddle and R. Reardon. 2002, Biological Control of Invasive Plants in the Eastern United States, USDA Forest Service Publication FHTET-2002-04. 413 pp.
- Wu, Z.-y., and P.H. Raven (eds). 2000. Flora of China; vol 24, Flagellariaceae through Marantaceae Science Press Beijing, China. 449 pp.
- Young, F.L., Gallandt, E.R. and All dredge, I.R. 2000. Predicting Winter Wheat (Tritiucum aestivum) yield loss based on Jointed Goatgrass (Aegilops cylindrica) populations from the previous seasons. Weed Technology: 423-427.
- Zamora, D.L., Thill, D.C., and Eplee, R.E. 1989. Eradication Manual for Common Crupina (Crupina vulgaris) Cass. Bulletin No. 701, University of Idaho, Moscow, ID, USA.
- Zouhar, K. 2002. Centaurea solstitialis. in Fire Effects Information System. U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station, Fire Sciences Laboratory.
Appendix 1A: Pest Risk Assessment Summary for Aegilops cylindrica (jointed goat grass)
- Identity of Organism
- Organism Status
- Regulatory Status
- Probability of Entry
- Probability of Establishment
- Probability of Spread
- Potential Economic Consequences
- Potential Environmental and Social Consequences
- Conclusion
Identity of Organism
Name: Aegilops cylindrica Host (1802), family Poaceae
Synonyms: Triticum cylindricum (Host) (1869), Cylindropyrum cylindricum (Host) (1982), Aegilops caudata
English Common Names: Jointed goat grass (Darbyshire, 2003), jointed goat grass, (Randall, 2002)
French Common Names: Égilope cylindrique (CFIA 2008)
Taxonomic Note: The taxonomy of the Triticeae tribe is notoriously difficult and unstable (Gould and Shaw, 1983). Due to its similarities with wheat, some authors have in the past renamed jointed goat grass Triticum cylindricum (Host) (Gould and Shaw 1983; Donald and Ogg 1991), although the currently accepted name is Aegilops cylindrica Host (e.g. Tutin et al. 1980; Tsvelev 1984; USDA ARS, 2006). Donald and Ogg (1991) describe the genus Aegilops as distributed worldwide and comprising at least 23 species. Holm et al. (1991) report Aegilops cylindrica as a weed in the United States and Türkiye, and other Aegilops species as weeds in Morocco, Portugal, Iran, Jordan and Israel. Donald and Ogg (1991) note that two varieties (or subspecies) of Aegilops cylindrica are recognized by some authors (Aegilops cylindrica var. cylindrica and var. rubiginosa) and Tsvelev (1984) lists three more (var. pauciaristata, var. aristulate and var. prokhanovii), all distinguished based on morphological differences of glumes and spikelets. Most studies fail to describe which varieties are studied, and it is not known whether they differ in their susceptibility to control mechanisms (Donald and Ogg 1991).
Description: Aegilops cylindrica is a winter annual grass that is very similar in appearance to winter wheat (Wicks et al. 2004). Individual plants consist of up to 50 erect flowering stalks (Donald and Ogg 1991). The root system of the plant is shallow and fibrous. Leaves are alternate and 2 to 5 mm wide, and vary from 3 to 15 cm in length (Barkworth 2006). The leaves are glabrous or sparsely hairy (Tutin et al. 1980) and the hairs are evenly spaced along the leaf blade margin (NJGRP 2006). The seed head, or spike, is a narrow cylinder 5-10 cm long with alternately arranged spikelets on opposite sides of the main axis of the spike. Spikelets are 8-10 mm long and contain 2-4 florets each. Glumes on the lower spikelets do not have awns, or have one awn. Glumes of apical spikelets have long awns (3.0 – 9.0 cm). Each spikelet contains on average 2 seeds (Hitchcock 1950; Donald and Ogg 1991; Lyon et al. 1995-2000; Barkworth 2006).
Seeds are reddish-brown caryopses, 6.5-9 mm long, 2 mm wide, and grooved. The lemma and palea adhere to the seed. Aegilops cylindrica plants produce an average of 130 seeds, and up to 3000 seeds under optimal conditions (Donald and Ogg 1991).
At maturity, some selections of Aegilops cylindrica may be distinguished from winter wheat by purple-coloured spikes (Donald and Ogg 1991). Spikes of Aegilops cylindrica are also much narrower and more cylindrical than those of wheat. In the field or in harvested seed lots, disarticulated Aegilops cylindrica spikelets are sometimes mistaken for small pieces of wheat straw (Donald and Ogg 1991).
Organism Status
The first occurrence of Aegilops cylindrica in Canada was likely a small non-persistent population in New Westminster, British Columbia in 1997 (Haber 2006). In 2002, there were four unconfirmed reports of Aegilops cylindrica present in shipments of Canada Western Red Spring wheat destined for Mexico.
In 2006, a small population of this species was discovered in the Regional Municipality of Niagara, Ontario. A small satellite population was discovered approximately 2.5 km away in 2007. In 2009 CFIA took some pre-emptive control measures to limit the further spread of these two populations.
As part of the CFIA import monitoring program, Aegilops cylindrica was found in eight samples of wheat and barley grain imported from the United States in 2008 and destined for British Columbia flour mills.
Regulatory Status
In 2005, the CFIA added Aegilops cylindrica to the Weed Seeds Order under the authority of the Seeds Actand Seeds Regulations, as a Class 1 Prohibited Noxious Weed Seed, prohibiting its movement as seed and as a contaminant in seed.
Provincially, Aegilops cylindrica is listed as a Provincial Noxious Weed in British Columbia, under the British Columbia Weed Control Act. The Province of Saskatchewan completed consultation on the revision of their Noxious Weed Act early in 2009, which includes the addition of Aegilops cylindrica to a prohibited noxious weeds list.
In the United States, Aegilops cylindrica is not included on the USDA's Federal Noxious Weed List, but it is regulated by Arizona, California, Colorado, Idaho, New Mexico, Oregon, and Washington (USDA, NRCS 2006).
Probability of Entry
The most likely pathway for the introduction of Aegilops cylindrica into Canada is general range expansion of the weed from infested states in the United States that share a border with Canada. General range expansion is a risk because Aegilops cylindrica is present in counties adjacent to the Canadian border in Washington, Idaho, Montana, and New York (Figure 1). Seeds of Aegilops cylindrica float and can be dispersed by runoff water in agricultural fields. However, the spiklets are large and unlikely to be moved great distances by wind. It has been suggested that Aegilops cylindrica seed can be dispersed by birds and mammals, after ingestion; however, it is not clear whether seeds remain viable after passing through the digestive tract in animals other than cattle (WSU Extension 2007).
The occurrence of Aegilops cylindrica in eight separate shipments of wheat and barley from the United States destined to Canada for milling and malting in 2008 provides evidence that the importation of grain for consumption is also a viable pathway for this species. If grains imported for consumption are contaminated with Aegilops cylindrica there is potential for some seeds to enter favourable environments and establish (Table 6). Examples include spillage along railway tracks, or dispersal of viable seeds by livestock that consume contaminated grain.
The listing of Aegilops cylindrica as a Class 1 Prohibited Noxious Weed in the Weed Seeds Order in 2005 provides a sufficient risk mitigation measure for preventing new incursions of this species through the seed pathway. Seed containing prohibited noxious weed seeds may not be sold, offered for sale, or imported into Canada.
Note: White areas indicate regions where the weed is not present
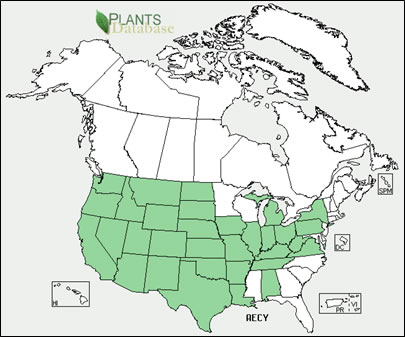
Description for Figure 1:
This image consists of a map of the United States and Canada demonstrating the distribution of Aegilops cylindrica through the use of the colour green. Approximately 4/5th of U.S. is covered in green with the majority located in the West half.
Source: (USDA, NRCS 2009) Footnote 1.
| Type of Potential Introduction | Specific Pathway | Description |
|---|---|---|
| Natural Means of Dispersal | Wind | Aegilops cylindrica reproduces only by seed, which have no obvious adaptations for long-range dispersal. |
| Water | Seeds float and will be dispersed by water runoff in agricultural fields | |
| Bird | Unclear if seeds can remain viable after passing through the digestive tracts of birds. | |
| Animal (external or internal) | Aegilops cylindrica has no obvious adaptations for this kind of dispersal. | |
| Intentional Introduction Pathways | Seed | Prohibited under Seeds Actand Seeds Regulations |
| Plants for planting (excluding seed) | None identified. | |
| Field crops not intended for propagation | None identified. | |
| Decorative arrangements and branches | None identified. | |
| Unintentional Introduction Pathways | Field crops not intendedfor propagation | Aegilops cylindrica could enter Canada as a contaminant in grain from areas where Aegilops cylindrica is established. Aegilops cylindrica is primarily a problem in cereal crops, particularly winter wheat. |
| Seed | Aegilops cylindrica is a Class 1 Prohibited Noxious Weed in the Weed Seeds Order, 2005 (Seeds Act) which provides sufficient risk mitigation. | |
| Hay and Straw | Straw originating from the U.S. and used for livestock bedding or packing material could contain seedsof Aegilops cylindrica. | |
| Manure | None Identified. | |
| Livestock | High viability and seedling emergence can be expected from seeds after passage through cattle. | |
| Raw wool and raw hides | Aegilops cylindrica has no obvious adaptations for this kind of dispersal. | |
| Motorized vehicles | Custom combine harvesters typically follow the winter wheat harvest northward, even across the Canada/U.S. border and is a potential mechanism of Aegilops cylindrica spred into Canada.
Farm trucks are commonlyused for multiple purposes , including hauling seed, fertilizer and harvested grain, and can lead to contamination and spread. |
|
| Nursery stock with soil | None identified. | |
| Used recreational equipment and clothing (excluding motorized vehicles) | None identified. |
The Seed Science & Technology Section of the CFIA Saskatoon laboratory analyze submitted samples of domestic and imported seed for contaminants, and have no record of Aegilops cylindrica being found in monitored samples of seed.
The level of risk associated with imports of hay and straw into Canada is relatively high when they originate from areas where Aegilops cylindrica is established and when Aegilops cylindrica has gone to seed in hay crop.
Other potential pathways considered to be of lesser importance include the movement of livestock with ingested seed across the border and used farm machinery potentially contaminated with soil which may carry seeds of Aegilops cylindrica.
Probability of Establishment
Aegilops cylindrica could potentially invade crops, pastures, and disturbed areas up to Canada Plant Hardiness Zone 2b, which includes the majority of Canada's agricultural land (Figure 2). Within this range, it is most likely to become a serious weed in winter wheat production areas; these include southern Ontario and in the southern Prairie Provinces where the majority of Canadian winter wheat is sown.
Probability of Spread
While seed production is moderate, Aegilops cylindrica could easily spread through both natural and man-made dispersal mechanisms if introduced into Canada.
Its biological and physical similarities to winter wheat make Aegilops cylindrica difficult to remove from dispersal pathways. Aegilops cylindrica spikelets measure approximately 8 to 10 mm long, and can be difficult to separate from wheat seed, which typically measure 5 to 7 mm. (Donald and Ogg 1991).
Note: Shaded area shows hardiness zones 2b and higher, based on the Canadian Plant Hardiness Zones map.
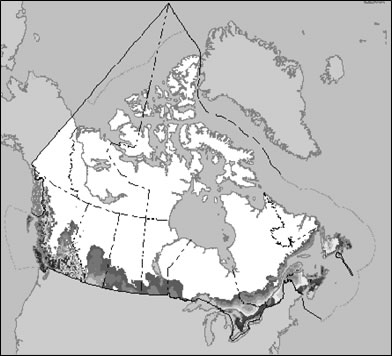
Description for Figure 2:
This image shows the potential range of Aegilops cyclindrica in Canada. In this image of a map of Canada, the lower region of Canada is coloured in various shades of black and grey showing hardiness zone 2b and higher, based on the Canadian Plant Hardiness Zones map.
Source: (NRC 2000)
There is also risk of hybridization of Aegilops cylindrica with wheat and other closely related species. Hybridization of Aegilops cylindrica with other species could result in increased weediness. The transfer of herbicide resistance traits from wheat into Aegilops cylindrica has already been documented in the United States (S. Darbyshire, 2006, pers. Comm.)
Potential Economic Consequences
In the United States, losses in winter wheat crop yield and quality due to Aegilops cylindrica are estimated at $150 million annually (Mallory-Smith 2001). Yield reductions are dependent on a variety of factors, notably rainfall, soil moisture and the severity of previous infestations. Field experiments in Washington State for instance, demonstrated that a first year infestation of one to five plants per square metre led to yield loss from 3% to 30 % in the following year (Young et al. 2000). This weed can also cause dockage losses in wheat. In non-herbicide resistant winter wheat varieties, costs of controlling Aegilops cylindrica are mostly indirect due to lack of selective control options. The presence of Aegilops cylindrica in Canadian seed could have negative trade impacts within Canada as well as with Mexico, certain U.S. states, Australia (although it is primarily a wheat-exporting nation), China, and possibly other countries.
Potential Environmental and Social Consequences
Aegilops cylindrica is primarily an agricultural weed, rather than an environmental weed, and its greatest environmental impacts are to sustainable agricultural practices. The presence of Aegilops cylindrica can discourage the cultivation of winter wheat, which is a relatively environmentally friendly crop, as it is an important component in reduced tillage systems and it provides nesting areas for waterfowl (Hamill 2005).
Conclusion
Additional phytosanitary measures may be necessary to prevent Aegilops cylindrica from further introduction into Canada, and to preventits spread. Eradication and subsequent monitoring of the Regional Municipality of Niagara populations should be a high priority, as should surveys in this and other potential high risk areas, particularly where winter wheat is grown, adjacent to railway lines, and along the Canada-United States border. Prevention of further introductions into Canada should be sought through activities such as public education, prevention of entry into Canada of contaminated farm equipment, vehicles, and straw, and possibly grain car inspections. Early control of Aegilops cylindrica, should it be introduced into Canada, would prevent negative economic and environmental consequences for Canada. Therefore, it is recommended that Aegilops cylindrica be considered for inclusion on the List of Pests Regulated by Canada.
Appendix 1B: Risk Management Considerations for Aegilops cylindrica (jointed goat grass)
- Existing Programs
- Detection Surveys in Canada
- Hybridization with Wheat
- Host for Pests that attack Winter Wheat
- Wheat Production Areas of Canada at Risk
- Previous imports
- Recommended Risk Mitigation Measures
- Trade Implications
- Cost effectiveness and feasibility
Existing Programs
In the United States the National Jointed Goatgrass Research Program was established in 1994 to guide research across the United States towards developing management systems based on cultural tactics and population dynamics.
The Government of Alberta, Agriculture and Rural Development, administers the Alberta Certified Weed Free Hay Program. The objective of the program is to provide a premium product that is recognized as marketable and transportable, to prevent the spread of restricted and noxious weeds, and to protect private and public lands from non-native, invasive plant species. Aegilops cylindrica is listed on their "Designated Weed List and Undesirable Plant Species List". Currently, this is the only existing weed free forage program in Canada.
Detection Surveys in Canada
Aegilops cylindrica is not known to occur in Canada outside of local incursions in the Regional Municipality of Niagara in Ontario (CFIA 2007). Field surveys for Aegilops cylindrica have focussed on potential points of introduction such as rail yards, ports, grain elevators, disturbed urban habitats and agricultural areas. Aegilops cylindrica was not found at any of the sites surveyed in Ontario or elsewhere in Canada.
Agriculture and Agri-Food Canada (AAFC) have been conducting weed surveys of the Prairie Provinces since the 1970s (Thomas and Leeson 2007). All weed species encountered in these surveys are identified and documented. Aegilops cylindrica is included in this general survey and has not been detected (J. Leeson per. comm. June 2009). An Inventory of Canadian Agricultural Weeds published by Darbyshire in 2002, also reported that Aegilops cylindrica was absent in Canada.
Hybridization with Wheat
The grass tribe Triticeae is known for inter-specific and inter-generic hybridization (S. Darbyshire, 2006, pers. comm). The genus Aegilops is capable of hybridizing with many species of Triticum, including T. aestivum (wheat) and producing fertile offspring, although fertility rates are low. Germination rates of Aegilops cylindrical/ wheat hybrid spikelets in an Okalahoma study ranged between 0.42 and 1.10% (Stone and Peeper 2004).
Hybridization needs to be considered in a determination of risk management as it could reduce the effectiveness of control methods if a herbicide-tolerant trait in wheat has the potential to be transferred to Aegilops cylindrica, potentially reducing the effectiveness of current control options.
The CFIA Plant Biosafety Office has reviewed the safety of herbicide-tolerant Clearfield wheat which is tolerant to the herbicide imidazolinone. The review examined the potential for gene flow of the imidazolinone-tolerant genes to wild relatives, including Aegilops cylindrica. These documents conclude "that there is no increased risk of consequences of gene flow between imidazolinone-tolerant wheat and Aegilops cylindrica, given the non-persistent nature of the introduction in British Columbia" and "the non-agricultural nature of the Ontario site" (CFIA 2007).
Although hybridization is currently unlikely the risks should not be underestimated if population of Aegliops cylindrica are allowed to expand into Canadian agricultural systems.
The large area occupied by both winter wheat and Aegilops cylindrica in the United States could lead to a significant number of hybridization events and, subsequently, the transfer of the herbicide resistance gene (Guadagnuolo et al. 2001). The weediness of Aegilops cylindrica could increase while control options decrease. It is possible that herbicide-tolerant Aegilops cylindrica populations evolved in the United States may be introduced into Canada via the pathways previously discussed.
Host for Pests that attack Winter Wheat
Aegilops cylindrica is considered an over-wintering host for some pests of winter wheat. The species has been shown to act as an alternate host for Russian wheat aphid (Diuraphis noxia Mordvilko) (Hammon et al. 1989). Aegilops cylindrica also acts as an over-wintering host for a number of fungal pathogens, such as those that cause pink mold, foot rot, root browning, damping off, dwarf bunt and karnal bunt (Donald and Ogg 1991).
Wheat Production Areas of Canada at Risk
During 2008-2009, Canada's wheat production was estimated at 27.3 million metric tons, an increase of 36% from 2007 (USDA 2008). Farm cash receipts indicated that the value of all wheat production in Canada was $4.2 and $5.7 million dollars in 2007 and 2008 respectively (Statistics Canada 2009). Wheat production areas at risk from Aegilops cylindrica include Alberta, Saskatchewan, Manitoba, Ontario, Quebec and the Atlantic provinces (Figure 2 & Figure 3). The majority of Canadian winter wheat is sown in southern Ontario and in the southern Prairie provinces.

Description for Figure 3:
This image shows the average wheat yields in Canada for the years of 1997 through 2001. The wheat yields can be predominantly found in the Prairie region consisting of lower to mid Manitoba, Saskatewan and Alberta the average wheat yield ranging from greater than 2.00 and less than 3.01 within that region. High wheat yields averaging more than 3.01 can also be found in the Atlantic Provinces as well.
Source: USDA 2004.
Canada is one of the world's largest wheat exporters with more than 95% of Canada's wheat produced in western Canada. According to the 2006 Census of Agriculture, wheat was Canada's largest field crop. Canada produces an average of over 25 million tonnes and exports approximately 19 million tonnes of wheat annually. As production significantly exceeds requirements for domestic consumption, the Canadian wheat industry is export-oriented.
If Aegilops cylindrica becomes established in export-oriented agricultural systems in Canada, exports of Canada's major crops and agricultural commodities could potentially be subject to phytosanitary measures by countries where Aegilops cylindrica is a regulated pest. For example, Aegilops cylindrica appears on the published regulated pest lists of China and Mexico; both countries have imported Canadian wheat in the past (Industry Canada 2009). While Aegilops cylindrica has the greatest impact on the production of winter wheat, phytosanitary measures could be applied by NPPOs of importing country to all wheat shipments; regardless of planting time or variety.
Previous imports
Intentional Introduction Pathways
- Aegilops cylindrica does not have a significant commercial use as a commodity itself; therefore it is not known to be intentionally imported for economic gain.
- In 2004 and 2008 import permits were issued for Aegilops cylindrica under the authority of section 43 of the Plant Protection Regulations for research purposes.
Unintentional Introduction Pathways
Grains and Field Crops
The total value of grain and field crop commodities Footnote 1 imported into Canada in 2008 was approximately $368 million. Of this amount, 3% of imports consisted of wheat from the United States, where Aegilops cylindrica is present. A small percentage of wheat was imported from other countries where Aegilops cylindrica is present, namely Lebanon, Russia, Türkiye, Bosnia, Italy, and Mexico. This represents less than 1% of the remaining imports of wheat in 2008 (Industry Canada 2009).
Seed
Canada imported approximately $1 million worth of wheat seed in 2007/2008, the majority of that from the United States (Canadian Seed Trade Association 2008).
Hay and Straw
The total value of hay and straw imported into Canada was approximately $13 million in 2008; 98% of this value came from states in the United States where Aegilops cylindrica is present (Industry Canada 2009).
Washington State was the greatest exporter of hay and straw to Canada with an average value of approximately $10 million per year in the last 5 years (Industry Canada 2009).
Used Vehicles and Farm Machinery
Used farm machinery moves across the United States-Canada border every year. Information is not available on the volume of imports of used farm machinery.
Recommended Risk Mitigation Measures
Natural Means of Dispersal
Natural range expansion could occur from infested counties in the United States along the U.S.-Canada border (USDA, NRCS 2006). It is recommended that risk mitigation include regular surveys targeting high risk areas along the Canada-United States border where the adjacent United States County is infested.
Education and outreach programs will also work to limit establishment in Canada, through early detection of new incursions.
Intentional and Unintentional Means of Introduction
Regulatory measures under the Plant Protection Act
- Regulate Aegilops cylindrica as a quarantine pest under the Plant Protection Act and add this species to the List of Pests Regulated by Canada (CFIA 2009);
- Prevent the importation, movement, and cultivation of this species in Canada either as an intentional import for food, feed or processing, or as a contaminant of a regulated commodity;
- Enable inspectors to take appropriate action for the purposes of eradicating the pest or preventing its spread;
- A permit to import issued under the authority of the Plant Protection Regulations, indicating specific import conditions for the handling and use of the commodity. It is the importer's responsibility to apply for and obtain the permit;
- The National Plant Protection Organization (NPPO) for the exporting country could be required to provide a phytosanitary certificate with or without an Additional Declaration stating freedom from Aegilops cylindrica;
- Phytosanitary agreements could be negotiated to certify imports from pest free areas and/or recognize noxious weed certification in countries or states of origin;
- End use does have an impact on risk mitigation measures. For example, wheat containing Aegilops cylindrica imported for milling would be considered lower risk than that coming in for direct use as pastured/rangeland animal feed. For milling, risk mitigation measures could include: the milling process itself, CFIA approved transport of the grain to the mill in a leak proof container and CFIA approved disposal of any Aegilops cylindrica parts removed from the commodity prior to the milling process;
- Additional measures may be waived for those commodities that have been treated or processed such that the risk of introduction of Aegilops cylindrica has been reduced to an acceptable level, for example heat treating grains or pelletization of hay or straw;
- Importation of grain known to be contaminated with Aegilops cylindrica may be permitted for research, processing, industrial or educational uses under a section 43 permit, and would be evaluated on a case by case basis;
- All risk mitigation measures forAegilops cylindrica must also take into consideration requirements/measures for other pests (e.g. pathogens and insects);
- The importation of used farm machinery would continue to fall under enforcement of Directive 95-26: "Phytosanitary requirements for soil and related matter and for items contaminated with soil and related matter" (CFIA, 2008). In 2003, the Canada Border Services Agency (CBSA) assumed responsibility for the initial import inspection services in respect of the Acts and Regulations administered by the CFIA to the extent that they are applicable at Canadian border points. The inspections of goods that may be contaminated with soil are among the responsibilities that were transferred to the CBSA in 2003. The Food, Plant and Animals Programs Section of the CBSA its Standard Operating Procedures (SOP) concerning the "Inspection of Imported Goods Potentially Contaminated with Soil." This SOP provides the CBSA's Border Services Officers with formal procedures for the inspection and disposition of goods that may be contaminated with soil, including used agricultural machinery and vehicles.
Regulatory measures under the Seeds Act
Continue to regulate Aegilops cylindrica as a prohibited noxious weed under the Weed Seeds Order of the Seeds ActFootnote 2. The importation of Aegilops cylindrica seed is prohibited under the Seeds Act and the Seeds Regulations have specific information requirements (CFIA 2009) to verify that seed imported into Canada is free of prohibited noxious weeds and meets the minimum standards for purity and germination for the crop kind in question. A certificate of analysis is required to demonstrate freedom from all prohibited noxious weeds.
Non regulatory measures
Education and outreach programs can assist in preventing and detecting new populations of Aegilops cylindrica. This can include providing information on identifying Aegilops cylindrica in the field, as well as educating people using custom combines and moving used farm machinery to clean all plant debris and soil that could contain seeds or spikelets of Aegilops cylindrica prior to moving the machines.
Trade Implications
- Not controlling import and spread of the species may result in infestations of field crops for export; if an importing country prohibits Aegilops cylindrica Canadian market access could be compromised. In 2010, Canada exported approximately $4.6 billion worth of wheat (Industry Canada 2011);
- Continued regulation of Aegilops cylindrica under the Seeds Act and Seeds Regulations secures the continued export of wheat seed from Canada to countries that regulate Aegilops cylindrica;
- The regulation of Aegilops cylindrica under the Plant Protection Act is not expected to have a major impact on seed trade, as it is already classified as a Class 1 noxious weed seed and is not cultivated in Canada;
- Regulation of Aegilops cylindrica under the Plant Protection Act will help to secure the potential export of hay and straw to the seven states in the United States that regulate the plant. In 2008, approximately $4.2 million worth of hay and straw were exported to Arizona, California, Colorado, Idaho, and Oregon (Industry Canada 2009);
- Since 98% of hay and straw imports come into Canada from the United States with the majority from Washington and Oregon where Aegilops cylindrica is present, there could be significant market losses if commodities are found to be contaminated;
- National Plant Protection Organizations (NPPO) of the exporting country could need to ensure freedom of Aegilops cylindrica in field crops; otherwise CFIA could refuse import;
- Trading partners would be impacted if Canada regulates Aegilops cylindrica as a quarantine pest, particularly the United States. This could potentially lead to a reduction or loss of imported product for Canadians should the NPPO of exporting countries be unable to meet proposed regulatory requirements.
Cost effectiveness and feasibility
- Aegilops cylindrica spikelets and winter wheat seed are quite similar in size, shape and density. These similarities make it difficult to separate the two. Standard sieve type seed cleaners do not sufficiently remove Aegilops cylindrica from wheat. For complete separation, special length graders and gravity tables should be used (NAPPO 2003);
- Foreign NPPOs will need to devote resources toward inspection of field crops and issuance of phytosanitary certificates;
- Aegilops cylindrica spikelets are easily identifiable making detection in samples quite easy;
- The CFIA Seed Program is already in place to prevent the entry of prohibited noxious weeds. The CFIA monitors compliance with the Canadian standards through the Marketplace Monitoring Program Footnote 3;
- For the regulation of Aegilops cylindrica in grain, Canada currently regulates the importation of grain under D-99-01: Barley, Oats, Rye, Triticale and Wheat - Phytosanitary Requirements on Import, Transhipped, In-Transit and Domestic Movement (CFIA 2009). D-99-01 has already been implemented to prevent the entry of regulated pests of cereals into areas of Canada not infested with the specified pests. Minor revisions will be needed to add requirements for Aegilops cylindrica;
- Phytosanitary measures are already in place for other pests regulated by Canada. Minor costs will be incurred for identification training for inspectors, delivery of import inspection of grains and field crops and sample collection. There will also be potential increased costs to the CFIA in the issuance of import permits.
Appendix 2A: Pest Risk Assessment Summary for Alopecurus myosuroides (slender foxtail)
- Identity of Organism
- Organism Status
- Current Regulatory Status
- Probability of Entry
- Probability of Establishment
- Probability of Spread
- Potential Economic Consequences
- Potential Environmental and Social Consequences
- Uncertainty
- Conclusion
- Technical Issues for Consideration
Identity of Organism
Name: Alopecurus myosuroides Huds (family Poaceae, subfamily Pooideae, tribe Poeae) (USDA -ARS 2009)
Synonyms: Alopecurus agrestis L., Alopecurus coerulescens Steud. & Hochst. ex Steud (nom. nud.), Alopecurus purpurascens, Tozzettia agrestis (L.) Bubani (Tropicos 2008)
English common names: Slender foxtail, black grass, black twitch, large foxtail (USDA -ARS 2009)
French common names: Vulpin des champs (USDA-ARS 2009)
Description: Alopecurus myosuroides is an erect, winter annual, tufted grass with reddish-purple panicles, appearing black at a distance. It flowers in early spring (Hubbard 1968; Aldrich-Markham 1992).
Organism Status
Alopecurus myosuroides has been reported to occur in Canada but did not become established, and no evidence was found that it is cultivated in Canada (CFIA 2008; Scoggan 1979). For more information, see under Probability of Entry. Based on this information, it is considered absent from the pest risk assessment area.
Current Regulatory Status
Alopecurus myosuroides is not currently regulated in Canada. Alopecurus myosuroides is not regulated as a federal noxious weed in the U.S. but is regulated by the state of Washington.
Probability of Entry
Alopecurus myosuroides has been accidentally introduced in contaminated grass seed to fields on research stations in Saanichton, BC (1955) and Brandon, MB (1965), but did not become established (Scoggan 1979). The provincial Conservation Data Centers (CDCs) do not consider the species to be established in these two provinces (CFIA 2008).
Alopecurus myosuroides is propagated only by seed. The main pathway for entry into Canada, based on monitoring of seed lots, is as a contaminant in grass seed lots (Table 1).
| Type of pathway | Specific pathways |
|---|---|
| Natural dispersal | Alopecurus myosuroides is an annual that spreads entirely by seed. Wind is the main natural means of dispersal over short distances. This is an unlikely pathway for entry into Canada, given the current range. |
| Intentional introduction | None identified. |
| Unintentional introduction Pathways | Slender foxtail is an occasional contaminant in lots of grass seed. This is the most likely pathway for entry into Canada. Based on the CFIA seed laboratory database, Alopecurus myosuroides has been detected in 5 imported grass samples in the past 9 years. As many as 12 seeds were reported from a single sample. Alopecurus myosuroides could potentially occur as a contaminant in grain lots. Although the species is a serious weed of winter cereals in Europe, it is very unlikely that Alopecurus myosuroides would occur as a contaminant in processed grains, as it would be readily removed during combining and cleaning. Seeds have not been reported in cereal seed samples in the past 9 years in Canada. It could also occur in imported hay and straw as contaminant seeds in panicles. |
Probability of Establishment
Alopecurus myosuroides is native to northern Africa (n. Algeria, n. Egypt, n. Libya and Tunisia), Asia (Afghanistan, Cyprus, Iran, Iraq, Israel, Jordan, Lebanon, Syria, Türkiye, Armenia, Azerbaijan, Georgia, Ciscaucasia, Dagestan, Turkmenistan, Uzbekistan, n. India and Pakistan), and Europe (United Kingdom, Belgium, Germany, Netherlands, Russian Federation, Ukraine, Albania, Bulgaria, Former Yugoslavia, Greece, Italy, Romania, France, Portugal and Spain) (USDA -ARS 2009).
It is widely naturalized in temperate parts of the world, including North America (U.S. and Mexico), South America (Bolivia, Peru, Argentina), China, Australia and New Zealand (Häfliger and Hildemar 1981). It has been widely introduced in the U.S. (see figure 1) as a weed of cultivation (Barkworth 2006).
In Europe, Alopecurus myosuroides grows in moist meadows, deciduous forests, and cultivated or disturbed ground and is a significant weed species in temperate cereal crops (Barkworth 2006). In the U.S., the species is reported mostly from cultivated fields (Barkworth 2006) but it is now commonly found in wetter sites of pasture, and along roadsides in Oregon and Washington (Lass and Prather 2007).
See Appendix 2C for a breakdown of HS codes and values of each commodity originating from countries where Alopecurus myosuroides is present. No data were available on imports of winter cereal varieties, Appendix 2D for Import data for grass seed and Appendix 2E for Import data for hay and straw.
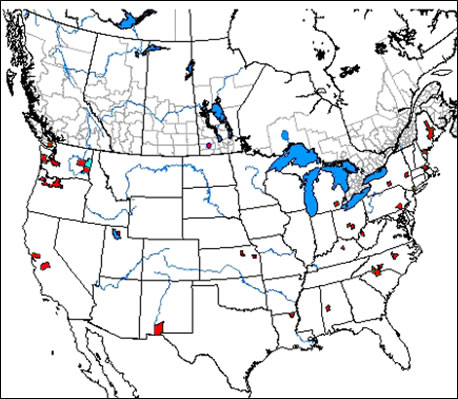
Description for Figure 1:
This is an image of North Arica showing the introduction of Alopecurus myosuroides to the United States. Red is used to demonstrate the regions in which it is found. It is scattered and minimal in nature covering approximately less thamen 3% of country.
Source: Barkworth 2006
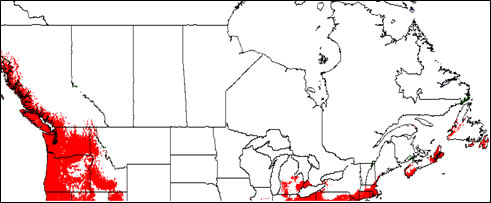
Description for Figure 2:
This image shows the potential range of Alopecurus myosuroides in Canada and upper America. Red is used to signify the regions in which it could survive according to the Canadian Plant Hardiness Zones map. In this map it would include coastal and extreme southern British Columbia, extreme southwestern Ontario and the coasts of Nova Scotia and Newfoundland. It would also include parts of the Western and Eastern coast of United States as well as areas located near the Great Lakes.
Source: NAPPFAST zones 6-9
Based on the world range, Alopecurus myosuroides is likely to survive to NAPPFAST hardiness zone 6 (see figure 2). This would include coastal and extreme southern British Columbia, extreme southwestern Ontario and the coasts of Nova Scotia and Newfoundland. As a weed of winter cereals, the most likely area to be affected would be southwestern Ontario, where winter cereals are grown. The winter wheat growing areas of the southern prairies appear to be outside the species' current range of climatic tolerance.
Probability of Spread
The main means of spread of Alopecurus myosuroides is by human activities. Seeds can be spread in contaminated crop seed, straw or manure. In cereal fields, the seeds of slender foxtail are blown out of the combine with the chaff. Cultivation equipment can also spread seeds. Seeds could easily move in soil during cultivation and in soil attached to farm equipment (CAB International 2007). Wind is the main natural means of dispersal, but seems to function only over relatively short distances (Colbach and Sache 2001).
There are several changes in cultivation practices that have favoured the spread of Alopecurus myosuroides in cereal fields in Europe (Aldrich-Markham 1992; CAB International 2007). Some that would also favour spread in Canada include:
- Suppression of many broadleaf weeds with herbicides has reduced competition for grass weeds such as Alopecurus myosuroides.
- Alopecurus myosuroides responds to high nitrogen fertilization by increasing tiller and seed production.
- Modern harvesting machines favour the dispersal of seeds as most of the seeds are shed by the time of harvest, and the rest are spread over the field with the chaff.
- Increase of autumn sown crops, especially cereals.
- Increased use of reduced tillage systems.
- Banning of straw burning.
- The development of herbicide-resistant populations of the weed.
Potential Economic Consequences
Alopecurus myosuroides is considered one of the most damaging weeds of winter cereals in England (Barkworth 2006) and is also now a major problem in Germany, Belgium and France, as well as parts of Italy, Yugoslavia, and Türkiye (Aldrich-Markham 1992). Its spread through Europe has been tied to the expansion of winter wheat and barley acreage, under modern, mechanized cultivation (Aldrich-Markham 1992).
Alopecurus myosuroides occurs in a wide range of crops, but is mainly associated with cereal dominated rotations. It is much commoner in autumn-sown cereal crops, such as winter wheat. It is also found on disturbed land in waste places but does not persist in grassland or pasture (CAB International 2007). It infests other winter crops, including grass seed, rapeseed, and forage legumes (Aldrich-Markham 1992) and is difficult to eradicate from cultivated fields that are repeatedly planted in cereals (Hubbard 1968).
In North America, Alopecurus myosuroides has become a major weed problem in western Oregon and eastern Washington, mostly in cereals, but it has also spread into pastures (Lass and Prather 2007).
Potential Environmental and Social Consequences
Alopecurus myosuroides does not seem to be an environmental weed either in its native or introduced range, being confined to disturbed habitats such as waste places and cultivated fields (Hubbard 1968; Barkworth 2006). There are no obvious potential social consequences to be expected from slender foxtail.
Uncertainty
No uncertainties potentially affecting the outcome of the risk assessment were identified.
Conclusion
Based on the outcome of this pest risk assessment, Alopecurus myosuroides is likely to become weedy or invasive in parts of southern Canada (Figure 2). It is recommended that the pest risk analysis process continue for this plant with the completion of a Risk Management Document.
Technical Issues for Consideration
Alopecurus species. The plants are also readily identifiable by trained personnel.
Appendix 2B: Risk Management Considerations for Alopecurus myosuroides (slender foxtail)
- Values at Risk
- Potential Mitigation Measures for Natural Means of Dispersal
- Potential Mitigation Measures for Intentional Introduction Pathways
- Potential Mitigation Measures for Non-intentional Introduction Pathways
Values at Risk
Alopecurus myosuroides is considered the most serious weed of winter cereals in Europe, reducing crop yield (CAB International 2007). Alopecurus myosuroides will most likely have the greatest impact on crops in Ontario, where winter wheat is produced. In 2008, approximately 650,000 tonnes (or 24 million bushels) of winter wheat were produced in the southwestern Ontario counties of Chatham-Kent, Essex and Lambton where Alopecurus myosuroides could potentially occur. This represents almost a quarter of winter wheat production in Ontario (OMAFRA 2009). Between the year 2000 and 2004, the average value of winter wheat production in Ontario was $250 million per year (OMAFRA 2009). Other crops that could be affected (to a lesser extent) by Alopecurus myosuroides in the respective counties include oats and barley with production values of 5,300 tonnes (85,000bu and 2,400 tonnes (112,000bu respectively in 2008 (OMAFRA 2009).
Potential Mitigation Measures for Natural Means of Dispersal
Wind is the main natural means of dispersal for Alopecurus myosuroides, but only over short distances. Given the current range, wind dispersion is an unlikely pathway for entry into Canada (Allison 2009); thus, no mitigation measures for natural means of dispersal are required at this time.
Potential Mitigation Measures for Intentional Introduction Pathways
No intentional introduction pathways were identified (Allison 2009). Alopecurus myosuroides is not available in Canada as an ornamental plant (CNLA 2009). Although no mitigation measures are currently needed, this pathway will still be regulated under the Plant Protection Act if this species is placed on the List of Pests Regulated by Canada and the Seeds Act if placed on the Weed Seeds Order.
Potential Mitigation Measures for Non-intentional Introduction Pathways
Field Crops Not Intended for Propagation
Previous imports
- The total value of cereal grain commodities that have winter varieties (wheat, rye, barley, and other unmilled cereals) imported in 2008 was approximately $39 million or 8.8 million metric tonnes. Approximately $7.7 million or 1.9 tonnes worth of imports were from countries where Alopecurus myosuroides is present (Statistics Canada 2009). See Appendix 2C for a breakdown of HS codes and values of each commodity originating from countries where Alopecurus myosuroides is present. No data were available on imports of winter cereal varieties.
- The level of risk associated with imports of cereal grain into Canada is relatively low since the majority of imports (82%) originate from areas where Alopecurus myosuroides is absent (Statistics Canada 2009).
Potential risk mitigation measures
Regulate Alopecurus myosuroides as a quarantine pest under the Plant Protection Act and add this species to the List of Pests Regulated by Canada (CFIA 2009) in order to:
- Prevent the importation, movement, and cultivation of this species in Canada.
- Enable inspectors to take appropriate action for the purposes of eradicating the pest or preventing its spread Footnote 1.
Regulatory actions under the Plant Protection Act could include one or more of the following:
- Exporters could be required to provide a Phytosanitary Certificate with or without an Additional Declaration stating freedom from Alopecurus myosuroides.
- Requirement for an import permit issued by the CFIA indicating specific import requirements and conditions for the pest status, handling and use of the commodity. It is the importer's responsibility to apply for and obtain the permit.
- Provisions for importation of grain contaminated with Alopecurus myosuroides for research, processing, industrial or educational uses under a section 43 permit on a case-by-case basis.
- Recognition of Pest Free Areas – if Alopecurus myosuroides can be shown to be absent from the country/state/region from which the field crop product was propagated, then risk is negligible and additional phytosanitary requirements may be waived.
- Note: End uses impact risk and may therefore impact required risk mitigation measures. Phytosanitary measures may be waived for those commodities that have been treated or processed such that the risk of introduction of Alopecurus myosuroides has been reduced to an acceptable level.
Combine harvesting equipment may blow a certain proportion of the weed seeds out of the grain during harvest due to their small size and low weight. It has been speculated that this may be up to 100%, but no study has been done (K. Allison, personal communication). The seeds may enter Canada in field crops not intended for propagation; a study examining the efficacy of removal during harvest should be done.
Trade implications
- Since a relatively small value of grain imports originate from areas where Alopecurus myosuroides is present, reduction and or loss of import markets if exporting countries are unable to meet proposed phytosanitary requirements would be low.
- Regulation of Alopecurus myosuroides under the Plant Protection Act will facilitate future exports of grain (average value of $1.6 million per year over the past 10 years; Industry Canada 2009) to the State of Washington, where Alopecurus myosuroides is regulated.
- Not controlling import and spread of the species may result in infestations of field crops (e.g. winter wheat) for export; if the importing country prohibits that species, Canadian market access could be compromised.
- Exporting countries may have to devote resources toward the inspection of grain commodities and the issuance of phytosanitary certificates.
Cost-effectiveness and Feasibility
- Resources will be needed by CFIA for marketplace monitoring and sampling, inspector training, and communication material development.
- Grain contaminated with Alopecurus myosuroides could be re-cleaned to remove the contaminant seeds. Alopecurus myosuroides seeds can be readily screened out of grass seed lots as the size of the seeds are 4.5-7.5 mm (Hubbard 1968). The seeds are readily identifiable by trained analysts. This should be done in the exporting country.
Seed
Previous imports
- The Seed Science and Technology Section of the CFIA Saskatoon laboratory maintains a record of contaminants through the marketplace monitoring of domestic and imported seed. Alopecurus myosuroides has been recorded in 5 imported grass seed samples in the past 9 years.
- The total value and quantity of grass seed commodities (Kentucky bluegrass, timothy, ryegrass and fescues) imported in 2008 from countries where Alopecurus myosuroides is present was approximately $22 million and 11 million kilograms respectively (Appendix 2D; Statistics Canada 2009). This represents 84% of the total value of grass seeds imported into Canada. The highest proportion of seed was imported from the states of Oregon and Washington with values of $17 million (9 million kilograms) and $4.4 million (2 million kilograms) respectively. The total value of imported grass seed has increased between $6 and $8 million over the last 5 years (Industry Canada 2009).
- The level of risk associated with imports of grass seed into Canada is relatively high since they originate primarily from areas where Alopecurus myosuroides is present.
Potential risk mitigation measures
Regulate Alopecurus myosuroides as a prohibited noxious weed (Class 1) under the Weed Seeds Order of the Seeds Act Footnote 2.
This species meets the definitions for Class 1 Footnote 3 species under the Weed Seeds Order.
All imported and domestic seed lots must be free of prohibited noxious weed seeds. Imported seed lots would require a certificate of analysis stating Alopecurus myos uroides is absent from the seed lot before it can be imported.
Regulate Alopecurus myosuroides as a quarantine pest under the Plant Protection Act and add this species to the List of Pests Regulated by Canada (CFIA 2009) in order to:
- Prevent the importation, movement, and cultivation of this species in Canada.
- Enable inspectors to take appropriate action for the purposes of eradicating the pest or preventing its spread Footnote 4.
Regulatory actions could include one or more of the following:
- Exporters could be required to provide a Phytosanitary Certificate with or without an Additional Declaration stating freedom from Alopecurus myosuroides.
- Requirement for an import permit indicating specific import requirements and conditions for the pest status, handling and use of the commodity. A section 43 import permit will be required for the importation of devitalized seed, preserved specimens for scientific research purposes by recognized herbaria and research facilities and the importation of seed for research in containment facilities.
- Recognition of Pest Free Areas – if Alopecurus myosuroides can be shown to be absent from the country/state/region from which the seed was harvested, then risk is negligible and additional regulatory requirements may be waived.
- Recognition of foreign government phytosanitary noxious weed certification in countries or states of origin may allow for additional phytosanitary requirements to be waived.
Trade implications
- Lack of Canadian regulation could compromise market access for Canadian commodities to those countries that regulate Alopecurus myosuroides, should this species become established in Canada.
- Regulation of Alopecurus myosuroides under the Seeds Act and Plant Protection Act will facilitate seed trade with the State of Washington, where Alopecurus myosuroides is regulated as a noxious weed. The value of grass seed exported to Washington in 2008 was low at approximately $960, 000 (Industry Canada 2009).
- There is potential for reduction and or loss of some import markets for Canadians if exporting countries are unable to meet proposed phytosanitary requirements. This loss will be significant as a large proportion of seed imports originate from countries where Alopecurus myosuroides is present.
- Exporting countries currently devote resources towards inspection of seed lots and issuance of Phytosanitary Certificates. Laboratories in foreign countries will need to be able to identify seeds of Alopecurus myosuroides within a seed sample. Exporters may need to ensure freedom of Alopecurus myosuroides in seed lots, otherwise CFIA can refuse import.
Cost-effectiveness and Feasibility
- The CFIA Seed Program is already in place to prevent the entry of prohibited noxious weeds. The CFIA monitors compliance with the Canadian standards through the Marketplace Monitoring Program Footnote 5.
- Seed contaminated with Alopecurus myosuroides could be re-cleaned to remove the contaminant seeds. Alopecurus myosuroides seeds can be readily screened out of grass seed lots as the size of the seeds are 4.5-7.5 mm (Hubbard 1968). The seeds are readily identifiable by trained analysts and can be separated from seeds of other Alopecurus species. This should be done in the exporting country.
Hay and Straw
Previous imports
The total value of hay and straw imports was around $13 million in 2008; 98% of this value came from the U.S. (Industry Canada 2009).
- Between 2004 and 2008, high quantities of hay and straw (an approximate average value of $11.1 million per year) were imported from Washington and Oregon, where Alopecurus myosuroides is present. This represents the majority of hay and straw imported into Canada. Since 2004, imports of hay and straw have remained relatively constant (Industry Canada 2009). Consult Appendix 2E for a detailed breakdown of hay and straw imports for the last 3 years.
- The level of risk associated with imports of hay and straw into Canada is relatively high since they originate primarily from areas where Alopecurus myosuroides is present.
Potential risk mitigation measures
Regulate Alopecurus myosuroides under the Plant Protection Act as a quarantine pest by placing it on the List of Pests Regulated by Canada in order to:
- prevent the importation, movement, and cultivation of this species in Canada.
- enable inspectors to take appropriate action for the purposes of eradicating the pest or preventing its spread Footnote 6.
Regulatory actions could include one or more of the following:
- Exporters could be required to obtain a Phytosanitary Certificate with or without an Additional Declaration stating freedom from Alopecurus myosuroides.
- Requirement for an import permit indicating specific import requirements and conditions for the pest status, handling and use of the commodity. It is the importer's responsibility to apply for and obtain the permit.
- Recognition of Pest Free Areas – if Alopecurus myosuroides can be shown to be absent from the country/state/region where the hay or straw was produced, then risk is negligible and additional phtyosanitary requirements may be waived.
- Requirement for heat treatment or pelletization for hay or straw intended for use as a biofuel stock.
Trade implications
- Regulation of Alopecurus myosuroides under the Plant Protection Act will address the potential export of hay and straw (value of approximately $3.6 million in 2008; Industry Canada 2009) to the State of Washington, where Alopecurus myosuroides is regulated.
- Exporting countries may have to devote resources towards inspection of hay and straw commodities and issuance of Phytosanitary Certificates. Inspectors in foreign countries need to be able to identify seeds of Alopecurus myosuroides that could be associated with hay and straw. Pre-harvest field inspections may be the most feasible option to ensure pest freedom. Exporters need to ensure freedom of Alopecurus myosuroides in hay and straw, otherwise CFIA can refuse import.
- Since 98% of hay and straw imports come from the U.S., and the majority from the state of Washington where Alopecurus myosuroides is invasive, there could be significant market losses of this commodity if it is found to be contaminated.
Cost-effectiveness and Feasibility
Resources will be needed by CFIA for marketplace monitoring and sampling, inspector training, and communication material development.
Vehicles and Used Farm Machinery
Previous imports
- A considerable volume of vehicles cross the U.S.-Canada border every year.
- Information is not available on the volume of imports of used farm machinery.
Potential Risk mitigation measures
Enforcement of the Directive 95-26: "Phytosanitary requirements for soil and related matter, alone or in association with plants" (CFIA 2008).
In 2003, the Canada Border Services Agency (CBSA) assumed responsibility for the initial import inspection services in respect of the Acts and Regulations administered by the CFIA to the extent that they are applicable at Canadian border points. The inspections of goods that may be contaminated with soil are among the responsibilities that were transferred to the CFIA in 2003. The Food, Plant and Animals Programs Section of the CFIA is currently finalizing its Standard Operating Procedures (SOP) concerning the "Inspection of Imported Goods Potentially Contaminated with Soil." This SOP provides the CFIA's Border Services Officers with formal procedures for the inspection and disposition of goods that may be contaminated with soil, including used agricultural machinery and vehicles.
Appendix 2C: Import data for wheat, rye, barley and other unmilled cereals
Import data for wheat, rye, barley and other unmilled cereals originating from countries where Alopecurus myosuroides is present
Value in Canadian Dollars ($)
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| U.S. – Washington | 433,287 | 2,317,101 | 1,825,056 |
| Argentina | 0 | 0 | 250,050 |
| Lebanon | 3,152 | 1,633 | 10,124 |
| Russia | 0 | 4,890 | 6,998 |
| New Zealand | 4,250 | 7,625 | 6,762 |
| Netherlands | 0 | 0 | 1,765 |
| U.S. – Oregon | 1,508,006 | 422,496 | 1,707 |
| Germany | 483 | 4,383 | 90 |
| Mexico | 0 | 1,172 | 4 |
| United Kingdom | 457 | 27,372 | 212 |
| China, Türkiye, Jordan, Italy, Ukraine, Bulgaria, Peru, France | 721 | 1,695 | 1,225 |
| Subtotal: | 1,950,356 | 2,788,367 | 2,103,993 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| Germany | 0 | 9 | 2,050,723 |
| India | 0 | 0 | 203 |
| U.S. – Oregon | 68 | 82 | 94 |
| Subtotal: | 68 | 91 | 2,051,020 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| China | 3,127 | 16,939 | 15,429 |
| India | 317 | 638 | 2,852 |
| Italy, Australia, Germany, New Zealand, Russia, Netherlands, Peru, Ukraine, U.S. – Oregon | 964 | 814 | 1,491 |
| U.S. – Washington | 4,787 | 0 | 21 |
| United Kingdom | 1,376 | 143 | 9 |
| Subtotal: | 10,571 | 18,534 | 19,802 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| Bolivia | 717,165 | 1,005,275 | 1,640,901 |
| China | 277,389 | 307,425 | 503,778 |
| U.S. – Oregon | 112,846 | 198,547 | 476,661 |
| Peru | 67,587 | 210,797 | 366,051 |
| Netherlands | 40,728 | 320,144 | 227,418 |
| India | 287,150 | 181,626 | 150,134 |
| Egypt | 106 | 0 | 57,357 |
| Russia | 25,498 | 24,587 | 51,599 |
| Mexico | 11,164 | 11,392 | 28,825 |
| France | 64,279 | 31,242 | 23,686 |
| Italy | 7,116 | 15,760 | 6,877 |
| United Kingdom | 781 | 5,502 | 5,253 |
| Ukraine | 19,433 | 4,461 | 2,024 |
| U.S. – Washington | 1,619 | 3,780 | 482 |
| Türkiye, Syria, Spain, Argentina, Bulgaria, Pakistan, Australia, Tunisia | 1,649 | 2,189 | 70 |
| Germany | 2,858 | 19,220 | 52 |
| New Zealand | 2,268 | 0 | 0 |
| Subtotal: | 1,639,636 | 2,341,947 | 3,541,168 |
| Total 2006 | Total 2007 | Total 2008 |
|---|---|---|
| 3,600,631 | 5,148,939 | 7,715,983 |
Quantity in metric tonnes (TNE)
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| U.S. – Washington | 2,200 | 8,916 | 5,334 |
| Argentina | 0 | 0 | 320 |
| Lebanon | 11 | 3 | 13 |
| Russia | 0 | 4 | 5 |
| New Zealand | 82 | 12 | 12 |
| Netherlands | 0 | 0 | 4 |
| U.S. – Oregon | 9,463 | 2,009 | 1 |
| Germany | 0 | 3 | 0 |
| Mexico | 0 | 0 | 0 |
| United Kingdom | 0 | 17 | 0 |
| China, Türkiye, Jordan, Italy, Ukraine, Bulgaria, Peru, France | 4 | 3 | 8 |
| Subtotal: | 11,760 | 10,967 | 5,697 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| Germany | 0 | 0 | 4,015 |
| India | 0 | 0 | 1 |
| U.S. – Oregon | 0 | 0 | 0 |
| Subtotal: | 0 | 0 | 4,016 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| China | 3 | 85 | 59 |
| India | 0 | 0 | 0 |
| Italy, Australia, Germany, New Zealand, Russia, Netherlands, Peru, Ukraine, U.S. – Oregon | 4 | 2 | 4 |
| U.S. – Washington | 32 | 0 | 0 |
| United Kingdom | 7 | 0 | 0 |
| Subtotal: | 46 | 87 | 63 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| Bolivia | 401,314 | 579,050 | 630,320 |
| China | 213,063 | 409,492 | 540,074 |
| U.S. – Oregon | 108,541 | 43,881 | 347,798 |
| Peru | 40,095 | 109,596 | 131,443 |
| Netherlands | 21,248 | 221,320 | 141,553 |
| India | 226,297 | 271,519 | 145,113 |
| Egypt | 0 | 0 | 20,650 |
| Russia | 3,941 | 6,092 | 5,961 |
| Mexico | 1,913 | 1,916 | 4,186 |
| France | 17,351 | 8,604 | 6,460 |
| Italy | 2,329 | 4,595 | 3,149 |
| United Kingdom | 487 | 558 | 4,668 |
| Ukraine | 4,569 | 2,569 | 2,600 |
| U.S. – Washington | 367 | 825 | 107 |
| Türkiye, Syria, Spain, Argentina, Bulgaria, Pakistan, Australia, Tunisia | 1,601 | 249 | 8 |
| Germany | 1,070 | 25,537 | 31 |
| New Zealand | 4 | 0 | 0 |
| Subtotal: | 1,044,190 | 1,685,803 | 1,984,121 |
| Total 2006 | Total 2007 | Total 2008 |
|---|---|---|
| 1,055,996 | 1,696,857 | 1,993,897 |
Source: Statistics Canada 2009. Countries with an export value of less than $1,000 per year are grouped together and a summed value for all countries is shown
Appendix 2D: Import data for grass seed
Import data for grass seed originating from countries where Alopecurus myosuroides is present
Value in Canadian Dollars ($)
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| U.S. - Oregon | 1,925,464 | 1,898,365 | 2,722,623 |
| U.S. - Washington | 7,210 | 26,523 | 46,936 |
| Germany | 80,272 | 89,726 | 67,197 |
| Italy | 0 | 4,284 | 37,816 |
| Netherlands | 3,420 | 24,291 | 15,640 |
| France | 0 | 10,613 | 3,606 |
| Subtotal: | 2,016,366 | 2,053,802 | 2,893,818 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| U.S. - Oregon | 4,181,870 | 4,755,449 | 7,836,065 |
| U.S. - Washington | 3,130,349 | 2,808,083 | 4,252,270 |
| Germany | 0 | 21,186 | 0 |
| Subtotal: | 7,312,219 | 7,584,718 | 12,088,335 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| U.S. -Oregon | 5,256,248 | 3,814,246 | 6,626,765 |
| U.S. - Washington | 2,779 | 2,974 | 85,305 |
| Germany | 355,869 | 339,445 | 222,094 |
| Netherlands | 79,587 | 106,655 | 144,991 |
| New Zealand | 34,589 | 109,624 | 63,301 |
| China | 97,129 | 63,127 | 28,963 |
| Belgium | 35,887 | 0 | 23,113 |
| France | 0 | 15,020 | 15,044 |
| Spain | 0 | 6,121 | 1,827 |
| Argentina | 0 | 17,242 | 1 |
| Italy | 61,300 | 32,132 | 0 |
| United Kingdom | 0 | 4,114 | 0 |
| Egypt | 6,121 | 0 | 0 |
| Subtotal: | 5,929,509 | 4,510,700 | 7,211,404 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| Netherlands | 14,689 | 0 | 0 |
| Germany | 2,090 | 0 | 0 |
| U.S.-Oregon | 1,379 | 0 | 0 |
| Subtotal: | 18,158 | 0 | 0 |
| Total 2006 | Total 2007 | Total 2008 |
|---|---|---|
| 15,276,252 | 14,149,220 | 22,193,557 |
Quantity in Kilograms per Cubic Metre (KGM)
Import data for grass seed originating from countries where Alopecurus myosuroides is present (2006-2008 - Quantity in Kilograms (KGM)).
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| U.S. - Oregon | 975,900 | 921,911 | 1,270,884 |
| U.S. - Washington | 2,583 | 9,628 | 14,786 |
| Germany | 38,091 | 40,000 | 36,947 |
| Italy | 0 | 1,800 | 22,665 |
| Netherlands | 1,721 | 10,612 | 5,606 |
| France | 0 | 5,209 | 1,515 |
| Subtotal: | 1,018,295 | 989,160 | 1,352,403 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| U.S. - Oregon | 1,468,614 | 1,822,725 | 2,654,033 |
| U.S. - Washington | 2,010,877 | 1,639,238 | 1,943,822 |
| Germany | 0 | 4,500 | 0 |
| Subtotal: | 3,479,491 | 3,466,463 | 4,597,855 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| U.S. - Oregon | 3,491,358 | 2,485,844 | 5,227,573 |
| U.S. - Washington | 1,460 | 1,665 | 57,564 |
| Germany | 321,092 | 197,979 | 91,268 |
| Netherlands | 62,310 | 60,564 | 56,649 |
| New Zealand | 27,350 | 73,875 | 25,000 |
| China | 59,360 | 33,930 | 6,067 |
| Belgium | 46,582 | 0 | 10,000 |
| France | 0 | 3,968 | 3,431 |
| Spain | 0 | 2,000 | 1,015 |
| Argentina | 0 | 24,990 | 0 |
| Italy | 21,000 | 7,500 | 0 |
| United Kingdom | 0 | 4,500 | 0 |
| Egypt | 1,500 | 0 | 0 |
| Subtotal: | 4,032,012 | 2,896,815 | 5,478,567 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| Netherlands | 16,276 | 0 | 0 |
| Germany | 771 | 0 | 0 |
| U.S.-Oregon | 658 | 0 | 0 |
| Subtotal: | 17,705 | 0 | 0 |
| Total 2006 | Total 2007 | Total 2008 |
|---|---|---|
| 8,547,503 | 7,352,438 | 11,428,825 |
Source: Statistics Canada 2009.
Appendix 2E: Import data for hay and straw
Import data for hay and straw originating from countries where Alopecurus myosuroides is present
Value in Canadian Dollars ($)
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| U.S. - Washington | 5,056,544 | 5,685,096 | 6,397,081 |
| U.S. - Oregon | 4,875 | 1,750 | 18,492 |
| India | 284,039 | 258,851 | 302,025 |
| Mexico | 9,709 | 28,495 | 23,099 |
| China | 263 | 0 | 5,744 |
| Germany | 3,079 | 2,453 | 3,013 |
| Pakistan | 108 | 2,089 | 1,131 |
| United Kingdom | 2,518 | 8,773 | 408 |
| Subtotal: | 5,361,135 | 5,987,507 | 6,750,993 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| U.S. -Washington | 3,849,245 | 4,045,927 | 4,115,211 |
| U.S. -Oregon | 1,086,048 | 630,851 | 330,770 |
| China | 13,007 | 12,363 | 32,527 |
| India | 2,933 | 28,297 | 76 |
| Australia | 0 | 19,132 | 0 |
| Germany | 60 | 3,000 | 0 |
| France | 19 | 2,014 | 0 |
| Mexico, U.K., Portugal, Belgium, Peru, Lebanon | 583 | 841 | 95 |
| Subtotal: | 4,951,895 | 4,742,425 | 4,478,679 |
| Total 2006 | Total 2007 | Total 2008 |
|---|---|---|
| 10,313,030 | 10,729,932 | 11,229,672 |
Source: Statistics Canada 2009.
Appendix 3A: Pest Risk Assessment Summary for Centaurea iberica (Iberian starthistle)
- Identity of the organism
- Organism Status
- Current Regulatory Status
- Probability of Entry
- Probability of Establishment
- Probability of Spread
- Potential Economic Consequences
- Potential Environmental and Social Consequences
- Uncertainty
- Conclusion
- Technical Issues for Consideration
Identity of the organism
Name: Centaurea iberica Trevir. ex Spreng (family Asteraceae) (USDA-ARS 2009)
Synonyms: Calcitrapa iberica (Trevir. ex Spreng.) Schur, Leucantha iberica (Trevir. ex Spreng.) Á. Löve & D. Löve (Tropicos 2008)
English common names: Iberian star-thistle, Iberian star thistle, pale starthistle, Spanish centaury-thistle, Iberian knapweed, Spanish thistle.
French common name: None found.
Description: Centaurea iberica is a biennial herbaceous plant, but may behave as an annual or short-lived perennial in some environments (Graham and Johnson, 2003). In Oregon, a rosette forms in May and June and the plants bolt and bloom from midsummer to fall. The plants grow from 30 to 200 cm tall. The leaves are divided into narrow linear segments. The rosettes have spines in the centre. The flower heads are white, pink, or pale purple, and 15–20 mm long with straw-coloured, spine-tipped bracts. The spines are more than 2.5 cm long. The achenes (cypselae) are white- or brown-streaked, 3–4 mm long, and have a white pappus which is 1–2.5 mm long (FNA Editorial Committee 1993+, ODA 2007).
Organism Status
Centaurea iberica is not reported to be naturalized in Canada, and no evidence was found that it is cultivated in Canada (CFIA 2008). Based on this information, the species is considered to be absent from the PRA area.
Current Regulatory Status
- Canada:
- Centaurea iberica is not regulated in Canada.
- United States:
- Centaurea iberica is not regulated as a Federal Noxious Weed in the U.S. but it is regulated in Arizona, California, Nevada and Oregon (USDA-NRCS 2009). No person may move Centaurea iberica or commodities found to contain this species or parts of it into or through these states (ADA 2006; CDFA 2003; State of Nevada 2003; ODA 2006).
Probability of Entry
No information could be found on the probable pathway for entry of Centaurea iberica into North America (Table 8).
| Type of pathway | Specific pathways |
|---|---|
| Natural dispersal |
|
| Intentional introduction |
|
| Unintentional introduction |
|
Probability of Establishment
In Europe, Centaurea iberica is native in Bulgaria, Greece, former Yugoslavia, Romania and Ukraine (Crimea) (Pankhurst, 1998). It is also native in Asia (Afghanistan, Cyprus, Iran, Iraq, Israel, Jordan, Lebanon, Syria, Türkiye, Armenia, Azerbaijan, Georgia, Russia (Ciscaucasia and Dagestan), southeastern Kazakhstan, Kyrgyzstan, Tajikistan, Turkmenistan, Uzbekistan, China (Xinjiang), northern India and Pakistan) (USDA-ARS 2009). Centaurea iberica has been introduced in North America in California, Kansas, Oregon, Washington and Wyoming (FNA Editorial Committee, 1993+) (see Figure 1). It spread abundantly in some counties of California during the early 1950's (Graham and Johnson 2003). Centaurea iberica is extremely competitive along roadsides and in range and pastures (ODA 2007).
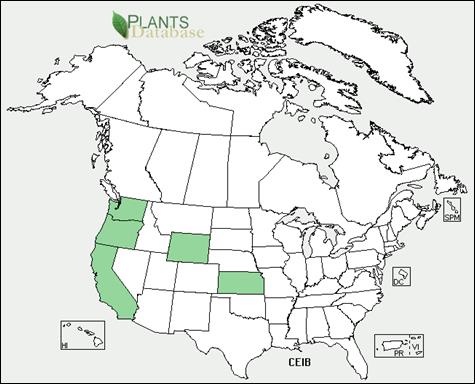
Description for - Figure 1
This map shows the range of Centaurea iberica in North America indicating its presence through the use of the colour green. The five states covered in green are Washington, Oregon, California, Wyoming and Nebraska.
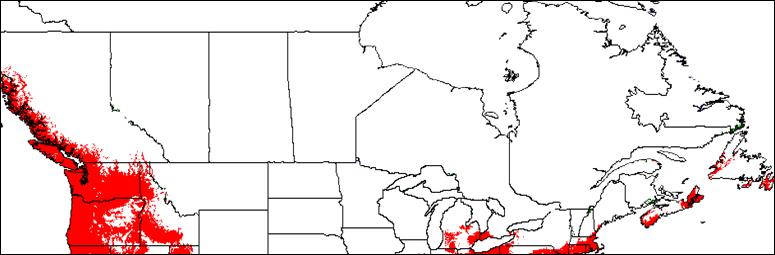
Description for - Figure 2
This image shows the potential range of Centaurea iberica in Canada and northern America through the use of a map. Red is used to indicate the regions in which Centaurea iberica could survive according to the Canadian Plant Hardiness Zones map, in this case NAPPFAST Hardiness Zone 6. In Canada, this includes coastal and southernmost BC, extreme southwestern Ontario and small areas on the coasts of the Maritime Provinces, including Newfoundland. In the U.S., it includes parts of the Eastern and Western coast as well as areas located near the Great Lakes.
Source: (USDA-NRCS 2009)
Probability of Spread
Centaurea iberica is a biennial plant that spreads by seed. The achenes have a pappus which suggests that they are dispersed by wind. The spiny involucres could be trapped in hair or clothing and be transported by animals or people.
Potential Economic Consequences
Centaurea iberica is considered a common weed in Lebanon (Holm et al., 1991). In Oregon, Centaurea iberica displaces valuable forage species in pastures and rangelands. The sharp spines deter grazing animals, restricting access for livestock and reducing the value of hay (ODA, 2007).
Potential Environmental and Social Consequences
Centaurea iberica seems to be confined to disturbed areas, including over-grazed rangelands (Uygur, 2001). The sharp spines mean that infestations can impede recreational use and restrict access for wildlife (ODA, 2007).
No other potential social consequences were identified.
Uncertainty
There are a number of areas of uncertainty in this risk assessment. This species is not well-covered in weed books or websites in North America. Basic information on self-compatibility, potential for apomixis, seed productivity and seed bank persistence are lacking. However, better information in these poorly-documented characteristics would not change the outcome from "reject" as the species scored very high in the assessment.
Conclusion
Based on the outcome of this pest risk assessment, Centaurea iberica is likely to establish and become weedy or invasive in some parts of southern and coastal British Columbia, southwestern Ontario and the east coast of Canada if it is introduced to these areas.
Technical Issues for Consideration
The plants of Centaurea iberica are very similar to those of purple star-thistle (Centaurea calcitrapa) as both have purple flower heads. New infestations have been misidentified in the past due to observers being familiar with purple star-thistle but unaware of Centaurea iberica. The plants of Centaurea iberica are more robust on average (FNA Editorial Committee 1993+).
However, the plants are best identified on the basis of the more elongated the flower heads (more rounded in purple star-thistle) and the paler colour of the corollas (Graham and Johnson 2003). The achenes can be easily separated by the presence of a pappus and the shinier pericarp in Centaurea iberica (ODA 2007).
In Canada, Centaurea calcitrapa L. (purple star-thistle) has been previously reported from southwestern BC and southern Ontario (Scoggan 1979). As it is not listed in the Illustrated Flora of British Columbia (Douglas et al. 1998), it seems that no persistent populations have become established there.
Appendix 3B: Risk Management Considerations for Centaurea iberica (Iberian starthistle)
- Values at Risk
- Potential Mitigation Measures for Natural Means of Dispersal
- Potential Mitigation Measures for Intentional Introduction Pathways
- Potential Mitigation Measures for Non-intentional Introduction Pathways
Values at Risk
Livestock Industry
Centaurea iberica could establish in pastures and fodder crops, and diminish the quality of forage. It is difficult to evaluate the value of forage because the harvest is usually intended to feed livestock on the farm where it is produced. Feed is the largest single cost of a beef operation, accounting for 60% of expenditures (Potter 2004). The increase in cost of production or a decrease in forage quality could directly impact dairy, cattle and sheep farm incomes.
Almost 250,000 ha of pasture and fodder crops are at risk in Canada. This area, mostly located in Southern Ontario, currently feeds around 261,500 cattle (dairy and beef production) and 36'000 sheep and lambs (see Table 2). In 2007, farm cash receipts were worth $410 million for dairy production and $160 million for the beef industry in Southern Ontario (OMAFRA 2006).
Seed Trade
The presence of Centaurea iberica in forage crops in Canada could affect trade of forage seed with the states of Arizona, California, Nevada and Oregon where Centaurea iberica is prohibited. The value of Canadian forage seed exported to these states fluctuates between $14.9 and $28.5 million/year (see figure 3).
| Canadian Provinces Table Note a | Hay and fodder crops Table Note b (ha) | Natural and tame pastures Table Note b (ha) | Cattle and calves Table Note b (nb) | Sheep and lambs Table Note b (nb) |
|---|---|---|---|---|
| Ontario | 157,000 | 52,000 | 220,000 | 34,000 |
| Nova Scotia | 4,500 | 4,000 | 7,000 | 2,000 |
| British Columbia | 4,500 | 42,000 | 34,500 | 0 |
| Total: | 166,000 | 98,000 | 261,500 | 36,000 |
Table Notes
- Table note 2
-
Areas located in hardiness zone 6 and higher (Agriculture and Agri-Food Canada 2009).
-
Statistics Canada 2001.

Description for Figure 3
This figure shows the total Canadian exports of forage seed (in $'000) to Arizona, California, Nevada and Oregon for the years of 2004 through 2008. This line graph relies that California, Nevada and Arizona have a forage seed export value of less than 5,000 while Oregon and the overall total increase steadily surpassing 15,000 for the four years with a notable increase in 2007 when it topped 25,000. Seed of clover, alfalfa, fescue, ray-grass (Lolium multiflorum and Lolium perenne) and other forage, except beet and Kentucky grass (Poa pratensis).
Source: Statistics Canada in Industry Canada 2009
Potential Mitigation Measures for Natural Means of Dispersal
Allison (2009) has not identified any risk associated with natural means of dispersal.
Potential Mitigation Measures for Intentional Introduction Pathways
According to Allison (2009), Centaurea iberica is not a cultivated plant. It is not available in Canada as an ornamental (CNLA 2009).
Potential Mitigation Measures for Non-intentional Introduction Pathways
Seed of Clover
Previous imports
For the past ten years, the value of clover seed imported from areas where Centaurea iberica is present fluctuated between $0.8 and $2 million (see Figure 4 and Appendix 3C) (Industry Canada 2009).

Description for Figure 4:
This figure shows the imports of clover seeds from countries and areas where Centaurea iberica is present for the years of 2006, 2007 and 2008. This bar graph relies the import values for China (light green), U.S. California (black), U.S. Washington (red and white strips), and U.S. Oregon (turquoise). This graph shows that the predominant import values (over 800,000) comes from U.S. Oregon; moreover, the import values have been rising steadily and the remaining of the actors accounts for less than 10% of the imports for those years.
Source: Statistics Canada in Industry Canada 2009
Potential risk mitigation measures
Regulate Centaurea iberica as a prohibited noxious weed (Class 1) under the Weed Seeds Order of the Seeds Act Footnote 1.
- This species meets the definitions for Class 1 Footnote 2 species under the Weed Seeds Order
- All imported and domestic seed lots must be free of prohibited noxious weed seeds. Imported seed lots would require a certificate of analysis stating Centaurea iberica is absent from the seed lot before it can be imported.
Regulate Centaurea iberica as a quarantine pest under the Plant Protection Act and add this species to the List of Pests Regulated by Canada (CFIA 2009):
- This could prevent the importation, movement, and cultivation of this species in Canada.
- Exporters of clover seed could be required to obtain a phytosanitary certificate stating freedom from Centaurea iberica.
- The only exceptions would be for the importation of preserved specimens for scientific research purposes by recognized herbaria and research facilities and the importation of seed for research in containment facilities.
- Phytosanitary agreements could be negotiated to certify imports from pest free areas and/or recognize noxious weed certification in countries or states of origin.
Trade implications
- Exporters in other countries will need to devote resources towards inspection of seed lots and issuance of phytosanitary certificates. Laboratories in exporting countries will need to be able to identify seeds of Centaurea iberica within a seed sample. Exporters need to ensure freedom of Centaurea iberica in seed lots, otherwise CFIA can refuse import.
- This measure will facilitate seed trade with the American states of Arizona, California, Nevada and Oregon, where Centaurea iberica is prohibited.
Cost-effectiveness and feasibility
- The CFIA Seed Program is already in place to prevent the entry of prohibited noxious weeds. The CFIA monitors compliance with the Canadian standards through the Marketplace Monitoring Program Footnote 3
- The seeds of Centaurea iberica are easily identified
- Centaurea iberica seeds can be readily screened out of clover seed due to the relatively large size of the seeds (3 to 4 mm long).
Livestock
Previous imports
Since 2004, around 8000 live animals intended for reproduction have been imported into Canada from areas where Centaurea iberica is present (see Figure 5). This represents around 2% of total imports of live animals for reproduction (CFIA internal data).
Potential risk mitigation measures
No mitigation measures needed. The level of risk associated with this pathway is relatively low due to the small number of livestock imported into Canada.
This sampling program helps to ensure that seeds sold in, imported into and exported from Canada meet established standards for quality, including varietal purity and germination, and are labelled so that they are properly represented in the marketplace, and in the case of most agricultural crop varieties, are registered prior to sale in Canada.

Description for Figure 5:
This figure displays, through the use of a bar graph, the number of live animals intended for reproduction that were imported from countries where Centaurea iberica is present. The United States's value is indicated using light blue and other countries using black. Cattle, in the U.S., has approximately 500 and is nonexistent in other counties. While goats, sheep and hog have hardly any visible presence in both the U.S. and in other countries on the table. Horses, on the other hand, surpass 5,000 in other countries while surpassing 1,000 in the U.S.
Source: CFIA internal data
Raw Wool and Raw Hides
Previous imports
The total value of raw wool imports was around $3.1 million in 2008; less than 0.1% of that value came from countries and American states where Centaurea iberica is present (Industry Canada 2009).
Potential risk mitigation measures
- No measures are required. Imports at risk are not significant.
- Raw wool and raw hides imported into Canada from all countries require CFIA inspection. It must be free from feces, blood, ectoparasites and dirt (plant and plant parts are considered to be dirt) or it will be refused of entry (Health of Animals Act, Directive AHPD-DSAE-2001-1-1 (CFIA 1990)).
- The vegetative matter is removed from commercial wool by carbonization Footnote 4.
Appendix 3C: Import data for Centaurea iberica (Iberian star-thistle)
Import data for red clover seeds for sowing originating from countries where Centaurea iberica is present (2006-2008).
| Exporting Country | Value ($CAN) 2006 | Quantity (kg) 2006 |
Value ($CAN) 2007 | Quantity (kg) 2007 |
Value ($CAN) 2008 | Quantity (kg) 2008 |
|---|---|---|---|---|---|---|
| U.S. – Washington | 316,192 | 226,457 | 217,368 | 126,359 | 443,638 | 242,069 |
| U.S. – Oregon | 0 | 0 | 4,298 | 1,814 | 224 | 112 |
| Sub-total: | 316,192 | 226,457 | 221,666 | 128,173 | 443,862 | 242,181 |
| Exporting Country | Value ($CAN) 2006 | Quantity (kg) 2006 |
Value ($CAN) 2007 | Quantity (kg) 2007 |
Value ($CAN) 2008 | Quantity (kg) 2008 |
|---|---|---|---|---|---|---|
| China | 28,345 | 12,598 | 0 | 0 | 52,892 | 23,508 |
| Sub-total: | 28,345 | 12,598 | 0 | 0 | 52,892 | 23,508 |
| Exporting Country | Value ($CAN) 2006 | Quantity (kg) 2006 |
Value ($CAN) 2007 | Quantity (kg) 2007 |
Value ($CAN) 2008 | Quantity (kg) 2008 |
|---|---|---|---|---|---|---|
| U.S. – Oregon | 396,191 | 106,022 | 651,354 | 204,622 | 468,110 | 135,354 |
| U.S. – California | 0 | 0 | 0 | 0 | 0 | 0 |
| U.S. – Washington | 18,741 | 3,625 | 0 | 0 | 8,261 | 2,268 |
| Sub-total: | 414,932 | 109,647 | 651,354 | 204,622 | 476,371 | 137,622 |
| Exporting Country | Value ($CAN) 2006 | Quantity (kg) 2006 |
Value ($CAN) 2007 | Quantity (kg) 2007 |
Value ($CAN) 2008 | Quantity (kg) 2008 |
|---|---|---|---|---|---|---|
| U.S – Oregon | 138,292 | 40,667 | 28,619 | 7,745 | 94,871 | 23,546 |
| U.S. – California | 6 | 2 | 56,460 | 10,343 | 22,911 | 4,990 |
| U.S. – Washington | 26,021 | 5,000 | 4,463 | 1,588 | 17,014 | 4,538 |
| Sub-total: | 164,319 | 45,669 | 89,542 | 19,676 | 134,796 | 33,074 |
| Value ($CAN) 2006 | Quantity (kg) 2006 |
Value ($CAN) 2007 | Quantity (kg) 2007 |
Value ($CAN) 2008 | Quantity (kg) 2008 |
|---|---|---|---|---|---|
| 895,443 | 381,773 | 962,562 | 352,471 | 1,055,029 | 412,877 |
Source: Statistics Canada
Appendix 4A: Pest Risk Assessment Summary for Centaurea solstitialis (yellow star-thistle)
- Identity of Organism
- Organism Status
- Current Regulatory Status
- Probability of Entry
- Probability of Establishment
- Probability of Spread
- Potential Economic Consequences
- Potential Environmental and Social Consequences
- Uncertainty
- Conclusion
- Technical Issues for Consideration
Identity of Organism
Name: Centaurea solstitialis (L.) (Asteraceae) (USDA-ARS, 2009)
Synonyms: Leucantha solstitialis (L.) A. Löve & D. Löve (USDA-NRCS, 2009)
English Common Names: Yellow star-thistle (USDA-NRCS, 2009), Centaurea solstitialis, golden star-thistle, St. Barnaby's thistle, yellow centaury, yellow cockspur (USDA-ARS, 2009)
French Common Names: Centaurée du solstice, chardon-doré, auriole
Description: Centaurea solstitialis is a winter annual, herbaceous plant, rarely a biennial or short-lived perennial. Populations in the United States are variable in their plant characteristics. The stems are stiff and erect, 15-200 cm in height. Centaurea solstitialis produces rosette leaves that lie close to the ground in the first fall. The inflorescences are borne in solitary flowerheads on stem tips, although vigorous plants may produce flowerheads in branch axils. The involucre is about 1.2-1.8 cm long. The involucral bracts have one long central spine 1.0-2.5 cm long and two or more pairs of short lateral spines and are covered with hairs. The achenes (fruits) are of two types, both glabrous and about 2 to 3 mm long. The disc florets produce most of the achenes (75-90%). These disc achenes are shiny and have a short (2-5 mm), stiff pappus. Achenes produced by the outer (ray) florets are duller, darker in colour and have no pappus. Centaurea solstitialis has a large taproot that grows to soil depths of 1 m or more, allowing access to deep soil moisture during dry summer and fall months. The dead stalks are persistent and usually remain standing through the winter (Zouhar 2002).
Organism Status
It is known historically from a few locations in Canada, but those populations have not established. No evidence was found that this species is cultivated in Canada (CNLA 2009). Based on this information, it is considered absent from Canada.
Current Regulatory Status
Federally, it is regulated by the CFIA as Class 1 prohibited noxious weed in the Weed Seeds Order under the Seeds Act. Provincially, it is regulated as a "Noxious Weed" in British Columbia, as a "Prohibited Noxious Weed" in Alberta, and as a "Prohibited Weed" in Saskatchewan under their respective Weed Control Acts and Regulations.
It is not regulated as a federal noxious weed in the U.S. but is regulated in the following states: AZ, CA, CO, HI, ID, MO, MT, ND, NM, NV, OR, SD, UT, WA, WV and WY.
Probability of Entry
It is an annual that spreads only by seed. Seeds have been found as contaminants in alfalfa and grain crops, and contaminated seeds are thought to have been the pathway for introduction of the species outside the native range (CAB International 2007).
| Type of pathway | Specific pathways |
|---|---|
| Natural dispersal | Seeds can disperse 1.5 m by wind and are dispersed over longer distances by animal activity. (CAB International 2007). The pappus bristles are covered with stiff, microscopic barbs that readily adhere to hair (Zouhar 2002). |
| Intentional introduction | None identified. |
| Unintentional introduction | Long-distance dispersal of seed is often directly related to human activities and occurs by movement of livestock, vehicles and equipment (Zouhar 2002). This is a likely pathway for entry into Canada. Seeds have been found as contaminants in alfalfa and grain crops, and contaminated seeds are thought to have been the pathway for introduction of the species outside the native range. There have been multiple introductions of Centaurea solstitialis into California, from Europe and Chile, usually as a contaminant in alfalfa seed (CAB International 2007). This would be the most likely pathway for entry into Canada. |
Probability of Establishment
It is native to Eurasia, including Armenia, Azerbaijan, Georgia, Iran, Iraq, Lebanon, Syria, Tajikistan, Türkiye, Turkmenistan, Ukraine, Algeria, Tunisia, Albania, Bulgaria, France, Greece, Italy, Spain and former Yugoslavia (USDA-ARS 2009). It is now found throughout Europe, in North and South America and Africa. It has spread to Central Asia, but does not persist in cold northerly areas. The earliest specimens in the U.S. were found in California in 1869 (CAB International 2007).
It has been reported from at least 40 U.S. states and four Canadian provinces. However, in eastern North America, infestations are sporadic and localized, and populations have failed to become established, in spite of repeated introductions. In California over 6.9 million hectares are infested, and the species is spreading in Idaho, Oregon and Washington (CAB International 2007).
In spite of historical reports of Centaurea solstitialis from Alberta, Saskatchewan, Manitoba and Ontario, there are no established populations anywhere in Canada.
Based on the results of the North Carolina State APHIS Plant Pest Forecasting (NAPPFAST) System, it appears that Centaurea solstitialis would survive to global plant hardiness zone 5 (Figure 11). Data from the pacific northwest states indicates that the species is common in warmer micro-climates on south and southwest facing slopes (Shafi et al. 2003; Zouhar 2002), therefore it is likely that it will be these micro-climates that will be vulnerable within zone 5 in Canada.
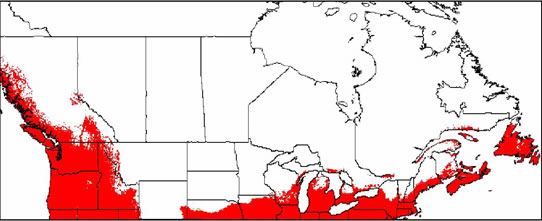
Description of Figure 1:
This image is a map of Canada and the Central United States with areas colored in red. The read areas are showing the establishment of the Yellow Star-thistle. Thy include the Canadian West Coat and some of the Mid-west States as well as the States south of the Great Lakes and the Canadian and U.S. East coast.
Probability of Spread
Seeds can disperse 1.5 m by wind, and seeds are dispersed over longer distances by animal or human activity. In California, it is thought that human activity accounts for most of the seed dispersal (CAB International 2007). The pappus bristles are covered with stiff, microscopic barbs that readily adhere to clothing and hair. Long-distance dispersal is often directly related to human activities and occurs by movement of livestock, vehicles, equipment, and contaminated hay and crop seed (Zouhar 2002).
Potential Economic Consequences
Centaurea solstitialis is toxic in large amounts to horses. The muscles of the lips, face and tongue become stiff and swollen, giving the horse a fixed expression. Poisoning can result in permanent brain damage, and severely affected animals eventually die of thirst and starvation (CAB International 2007).
Losses result from interference with livestock grazing and forage harvesting procedures, and lower yield and forage quality of rangelands. Livestock and wildlife avoid grazing in heavily infested areas resulting in slower weight gain and reduced quality of meat, milk, wool and hides (CAB International 2007).
Centaurea solstitialis is the most important roadside weed problem in much of central and northern California and causes occasional problems in cereals, orchards, vineyards, cultivated crops and wastelands (CAB International 2007).
Potential Environmental and Social Consequences
Centaurea solstitialis can reduce wildlife habitat and forage, displace native plants, and decrease native plant and animal diversity. Dense infestations threaten natural ecosystems by fragmenting plant and animal habitats (CAB International 2007). Infestations can also limit access to recreational areas, and reduce land value. Several rare and sensitive plant species in Oregon, Nevada and Idaho are thought to be threatened by C.solstitialis. Large populations of Centaurea solstitialis can alter the water cycle in annual grassland and foothill woodland ecosystems in California, by using more water to a greater soil depth than native vegetation (Zouhar 2002).
Uncertainty
There is some doubt about its potential range in Canada. It is very likely that the potential range is less than that shown in Figure 11, based on NAPPFAST zone 5. However, it is also very likely that there are suitable environments for persistent populations in southern British Columbia.
Conclusion
Based on the outcome of the CFIA's pest risk assessment, Centaurea solstitialis is likely to establish and become invasive in parts of Canada, including southern British Columbia if introduced. This plant should be considered for regulation under the Plant Protection Act (note that it is regulated by the CFIA as a prohibited noxious weed seed under the Seeds Act).
Technical Issues for Consideration
- Centaurea solstitialis is already regulated as a prohibited noxious weed seed under the Weed Seeds Order of the Seeds Act and Seeds Regulations.
- The seeds of Centaurea solstitialis can be readily identified visually by trained seed analysts.
Appendix 4B: Risk Management Considerations for Centaurea solstitialis (yellowstar-thistle)
- Values at Risk
- Potential Mitigation Measures for Natural Means of Dispersal
- Potential Mitigation Measures for Intentional Introduction Pathways
- Potential Mitigation Measures for Non-intentional Introduction Pathways
Values at Risk
Rangelands: Rangelands are important ecosystems that provide an abundance and variety of products, such as browse and forage for both wild and domesticated animals and wood fibre. Rangelands also provide drinking water, habitat for wildlife, biodiversity, nutrient cycling and recreational opportunities (Horton 1996). Centaurea solstitialis has the ability to invade rangelands, reducing their native biodiversity, wildlife habitat and forage and affecting the natural water cycle. One of the most common and profitable uses of rangeland is livestock grazing. Table 11 shows the number of cattle and calves, sheep and lambs and horses and ponies in areas of B.C. in hardiness zones five to nine. Of concern is that Centaurea solstitialis is toxic to horses if consumed in large amounts and can interfere with livestock grazing and is recognized as one of the "worst weeds in the West" by the Centre for Invasive Plant Management (CIPM 2009).
Potential Mitigation Measures for Natural Means of Dispersal
Natural dispersal is a possible pathway of entry of Centaurea solstitialis into Canada, particularly into British Columbia. Currently, there are four bordering counties in Washington, one bordering county in Idaho and one bordering county in Montana (adjacent to B.C.) that are infested with the weed (USDA-NRCS 2009). It is listed as a Class B Noxious Weed in the State of Washington (NWCB 2009), a noxious weed in Idaho on the Statewide Containment List (ISDA 2009) and a Category 3 noxious weed in Montana (MDA 2009). In Washington and Idaho, the goal is to prevent the spread of existing populations and the establishment of new populations, but not necessarily eradication. In Montana, a management criterion of Category 3 noxious weeds includes awareness and education, early detection and immediate action to eradicate infestations. Depending on how close existing populations are to the border, these programs could reduce the risk of Centaurea solstitialis naturally dispersing into Canada. It is also regulated as a noxious weed under the B.C. Weed Control Act, which requires all land occupiers to control designated noxious plants if found on their property (BCMAL 2002).
As there are populations of Centaurea solstitialis in U.S. counties adjacent to B.C., it is recommended that an early detection and rapid response (EDRR) program should be employed by the province.
| Type of Animal | Number of Animals | Percentage of B.C. Total |
|---|---|---|
| Cattle and Calves | 355,871 | 44% |
| Sheep and Lambs | 38,612 | 63% |
| Horses and Ponies | 53,246 | 49 % |
Source: Statistics Canada, 2007
Note: data is based on Census Agricultural Regions and Census Divisions, some of which extend beyond hardiness zone 5.
Potential Mitigation Measures for Intentional Introduction Pathways
No intentional introduction pathways for Centaurea solstitialis were identified. Although no mitigation measures are currently required, this pathway will still be regulated under the Plant Protection Act if this species is placed on the List of Pests Regulated by Canada (CFIA 2009).
Potential Mitigation Measures for Non-intentional Introduction Pathways
Field Crops Not Intended for Propagation
Previous imports
It is unknown which grain commodities could be potentially contaminated with seed of Centaurea solstitialis, but the weed has been noted to affect cereal production.
Potential risk mitigation measures
Regulate Centaurea solstitialis as a quarantine pest under the Plant Protection Act and add this species to the List of Pests Regulated by Canada (CFIA 2009) in order to:
- Prevent the importation, movement, and cultivation of this species in Canada.
- Enable inspectors to take appropriate action for the purposes of eradicating the pest or preventing its spread Footnote 1.
- Regulatory actions under the Plant Protection Act could include one or more of the following:
- Exporters could be required to provide a Phytosanitary Certificate with or without an Additional Declaration stating freedom from Centaurea solstitialis.
- Recognition of Pest Free Areas – if Centaurea solstitialis can be shown to be absent in the area from which the field crop product was propagated, then risk is negligible and additional phytosanitary requirements may be waived.
- An import permit indicating specific import requirements and conditions for the pest status, handling and use of the commodity could be required. It is the importer's responsibility to apply for and obtain the permit.
- Provisions for importation of grain contaminated with Centaurea solstitialis for research, processing, industrial or educational uses under a section 43 Permit to Import on a case-by-case basis.
- End uses impact risk and may therefore impact required risk mitigation measures. Phytosanitary measures may be waived for those commodities that have been treated or processed such that the risk of introduction of Centaurea solstitialis has been reduced to an acceptable level.
All risk mitigation measures for field crop commodities containing Centaurea solstitialis must be taken with consideration for requirements/measures for pests other than plants (e.g. pathogens and insects).
Trade implications
- Exporting countries may have to devote resources towards inspection and issuance of Phytosanitary Certificates. Laboratories in foreign countries will need to be able to identify seeds of Centaurea solstitialis. Exporters will need to ensure freedom of Centaurea solstitialis in field crop shipments; otherwise the CFIA can refuse entry.
- Potential for reduction and/or loss of some import markets for Canada should exporting countries be unable to meet proposed phytosanitary requirements.
- Not controlling import and spread of the species may result in infestations of field crops for export; if the importing country prohibits that species, market access for Canadian commodities could be compromised.
- Canadian regulation will facilitate trade with those countries and states which have established regulatory control of Centaurea solstitialis.
Cost-effectiveness and Feasibility
- Resources will be needed by the CFIA for marketplace monitoring and sampling, inspector training, and communication material development.
- Seeds could be screened out of grain lots, depending on the size of the contaminated grain. The seeds are on average 2.5 mm long, 1-1.2 mm wide and 0.5 mm thick. It may be more difficult to clean small-seeded crop kinds. Grain can be cleaned prior to export to remove contaminant seeds.
- Field inspections or laboratory testing in the exporting country could be used to ensure freedom from Centaurea solstitialis.
- A determination of which grains are more prone to contamination of Centaurea solstitialis would be useful to have a more targeted approach for this pathway.
Seed
Two specimens of Centaurea solstitialis were collected in a hay field in Campbellford, Ontario in 1971, prior to the listing of this species on the Weed Seeds Order in 1986.
- The Seed Science and Technology Section of the CFIA Saskatoon laboratory maintains a record of contaminants found through marketplace monitoring of domestic and imported seed and Centaurea solstitialis has not previously been recorded as a seed contaminant.
- From 2006 to 2008, an average of approximately $4.2 million per year or 852,000 kilograms of alfalfa seed was imported from countries where Centaurea solstitialis is present (Statistics Canada 2009). The majority of these imports (approximately 90%) were from infested states in the U.S. (Appendix 4D), with the highest volume arriving from Idaho. An average of approximately $3.1 million worth (574,000 kilograms) of alfalfa seed was imported from Idaho from 2006 to 2008 (Statistics Canada 2009).
- The level of risk associated with imports of alfalfa seed into Canada is relatively low since Centaurea solstitialis has been listed on the Weed Seeds Order since 1986. Although 84% of alfalfa seed imports originate from countries where the weed is present (Industry Canada 2009), exporting countries are required to screen seed shipments to ensure freedom from Centaurea solstitialis.
Previous imports
It is unknown which grain commodities could be potentially contaminated with seed of Centaurea solstitialis, but the weed has been noted to affect cereal production.
Potential risk mitigation measures
- Continue to regulate Centaurea solstitialis as a prohibited noxious weed (Class 1 Footnote 2 ) under the Weed Seeds Order of the Seeds Act Footnote 3.
- All imported and domestic seed lots must be free of prohibited noxious weed seeds. Imported seed lots require a certificate of analysis stating Centaurea solstitialis is absent from the seed lot before being imported.
- Regulate Centaurea solstitialis as a quarantine pest under the Plant Protection Act.
- Add this species to the List of Pests Regulated by Canada (CFIA 2009) in order to prevent the importation, movement, and cultivation of this species in Canada.
- Enable inspectors to take appropriate action for the purposes of eradicating the pest or preventing its spread.
Regulatory actions under the Plant Protection Act could include one or more of the following:
- Exporters could be required to provide a Phytosanitary Certificate with or without an Additional Declaration stating freedom from Centaurea solstitialis.
- Requirement for a Permit to Import indicating specific import requirements and conditions, handling and use of the commodity.
- Exceptions may be made for the importation of devitalized seed, preserved specimens for scientific research purposes by recognized herbaria and research facilities and the importation of seed for research in containment facilities.
- Recognition of Pest Free Areas – if Centaurea solstitialis can be shown to be absent from the area from which the seed was harvested, then risk is negligible and additional phytosanitary requirements may be waived.
- Recognition of foreign government phytosanitary noxious weed certification in countries or states of origin. This may allow for additional phytosanitary requirements to be waived.
Trade implications
- Exporting countries currently devote resources towards inspection of seed lots and issuance of Phytosanitary Certificates. Laboratories in foreign countries need to be able to identify seeds of Centaurea solstitialis within a seed sample. Exporters currently need to ensure freedom of Centaurea solstitialis in seed lots, otherwise the CFIA can refuse entry.
- Since Centaurea solstitialis is already listed as a prohibited noxious weed, no further trade implications are expected.
- Lack of Canadian regulation could compromise market access for Canadian commodities to those countries that regulate Centaurea solstitialis, should this species become established in Canada.
- Canadian regulation facilitates trade with states in the U.S. that regulate C. solstitialis (see Appendix 4A section III - Current Regulatory Status). Canada exported approximately $28 million worth (65% of the total value exported) of alfalfa seed to these states in 2008 (Industry Canada, 2009).
Cost-effectiveness and Feasibility
- The CFIA Seed Program is already in place to prevent the entry of prohibited noxious weeds, including Centaurea solstitialis. The CFIA monitors compliance with the Canadian standards through the Marketplace Monitoring Program Footnote 4.
- Seed can be easily identified by trained seed analysts.
- Prior to export, seed is typically cleaned to remove contaminant seeds.
- Seeds of Centaurea solstitialis can be readily screened out of seed lots. This should be done in the exporting country.
Hay and Straw
Previous imports
- From 2006 to 2008, an average value of approximately $12.2 million per year of hay and straw was imported from countries and U.S. states where this weed is present. This represents 97% of the total value of hay and straw coming into Canada (Industry Canada 2009).
- The majority of hay and straw is exported from the State of Washington. In 2008, $10.5 million worth of hay and straw was imported into Canada from this state (Industry Canada 2009).
- Due to the high percentage of hay and straw coming from places infested with Centaurea solstitialis, the risk associated with this pathway is relatively high.
Potential risk mitigation measures
Regulate Centaurea solstitialis under the Plant Protection Act as a quarantine pest by placing it on the List of Pests Regulated by Canada (CFIA 2009) in order to:
- Prevent the importation, movement, and cultivation of this species in Canada.
- Enable inspectors to take appropriate action for the purposes of eradicating the pest or preventing its spread Footnote 5.
- Regulatory actions under the Plant Protection Act could include one or more of the following:
- Exporters could be required to obtain a Phytosanitary Certificate with or without an Additional Declaration stating freedom from Centaurea solstitialis.
- Requirement for a Permit to Import indicating specific import requirements and conditions for the pest status, handling and use of the commodity. It is the importer's responsibility to apply for and obtain the permit.
- Recognition of Pest Free Areas – if it can be shown to be absent from the area from which the hay or straw was harvested, then risk is negligible and additional phytosanitary requirements may be waived.
- Requirement for heat treatment or pelletization for hay or straw intended for use as a biofuel stock.
Trade implications
- Exporting countries will need to devote resources towards inspection of hay and straw commodities and issuance of Phytosanitary Certificates. Inspectors in foreign countries need to be able to identify seeds that could become associated with hay and straw. Pre-harvest field inspections may be the most feasible option to ensure pest freedom. Exporters need to ensure freedom of Centaurea solstitialis in hay and straw, otherwise the CFIA can refuse entry.
- Since 97% of hay and straw imports come from U.S. (mostly Washington) where Centaurea solstitialis is invasive, there could be significant market losses of this commodity if it is found to be contaminated.
- Regulation under the Plant Protection Act will secure the export of hay and straw (value of approximately $5.5 million in 2008; Industry Canada 2009) to states where it is regulated.
Cost-effectiveness and Feasibility
- Resources will be needed by the CFIA for marketplace monitoring and sampling, inspector training, and communication material development.
Livestock
Previous imports
- Between 2004 and 2008, approximately 22,000 live horses, assess, bovine animals, swine, sheep and goats for reproduction were imported into Canada.
- Just over half of the imported livestock came from the eastern U.S., where populations of Centaurea solstitialis have not been able to establish.
- The risk associated with the livestock pathway is relatively low due to the small number of animals for reproduction being imported into Canada from places where Centaurea solstitialis is established.
Potential risk mitigation measures
- All regulated animals (including livestock) receive inspection upon their arrival in Canada either by the Canada Border Services Agency (CBSA) or the CFIA according to the Animal Health Directive APHD-DSAE-IE-2004-6-2 (CFIA 2008). This inspection could include a visual check (e.g. for ectoparasites) of the animals. A visual check for plant material could be conducted at this time. Inspection for plant material could be targeted at those animals originating from places infested with Centaurea solstitialis.
- An awareness campaign targeted toward livestock exporters on the CFIA website could be put in place. It would encourage people bringing livestock into the country to ensure their animals are cleaned and free from plant material, particularly seeds.
Trade implications
Exporters are already required to undergo inspection of livestock at the border.
Cost-effectiveness and Feasibility
- CBSA officials, veterinarians or animal health inspectors already perform inspection of livestock entering Canada. Integration of plant material inspection into routine veterinarian checks could be done in order to reduce time and costs.
- Seeds have pappus bristles that are barbed that enable them to adhere to livestock. It may be difficult to detect small seeds, especially on animals with long hair. The time and cost commitment may be too great to control this pathway in this manner.
- Resources will be needed by the CFIA for the development of communication material should a public awareness campaign be put in place.
Vehicles and Used Farm Machinery
Previous imports
- A considerable number of vehicles cross the Canada-U.S. border every year.
- Information is not available on the number of imported used farm machinery or harvesting equipment used in custom combining operations.
Potential Risk mitigation measures
Enforcement of the Directive 95-26: "Phytosanitary requirements of soil and related matter and for items contaminated with soil and related matter" (CFIA 2008).
In 2003, CBSA assumed responsibility for the initial import inspection services in respect to the Acts and Regulations administered by the CFIA to the extent that they are applicable at Canadian border points. The inspection of goods that may be contaminated with soil are among the responsibilities that were transferred to the CBSA in 2003. The Food, Plant and Animals Programs Section of the CBSA is currently finalizing its Standard Operating Procedures (SOP) concerning the "Inspection of Imported Goods Potentially Contaminated with Soil." This SOP provides the CBSA's Border Services Officers with formal procedures for the inspection and disposition of goods that may be contaminated with soil, including used agricultural machinery and vehicles.
Nursery Stock with Soil
There are two documented references of Centaurea solstitialis being found in gardens or landscaped areas. The references indicate that the pest plant may be introduced as an ornamental plant or as a root ball of shrubs or trees.
Previous imports
- The total value of imports of nursery stock was approximately $173 million in 2008 Footnote 6. Approximately 78% of this value came from states in the U.S. where Centaurea solstitialis is present (Industry Canada 2009).
- While soil from the non-continental U.S. is prohibited as per Directive D-95-26: Phytosanitary requirements of soil and related matter and for items contaminated with soil and related matter, soil associated with nursery stock from the continental U.S is permitted in most cases. It is not possible to determine the amount of nursery stock from the continental U.S. arriving with soil.
Potential Risk mitigation measures
Regulate Centaurea solstitialis under the Plant Protection Act as a quarantine pest and add it to the List of Pests Regulated by Canada in order to:
- Prevent the importation, movement, and cultivation of this species in Canada
- Enable inspectors to take appropriate action for the purposes of eradicating the pest or preventing its spread.
Regulatory actions could include one or more of the following:
- Requirement for exporters to obtain a Phytosanitary Certificate with or without an Additional Declaration stating freedom from Centaurea solstitialis. Centaurea solstitialis could be added to Appendix 6 of D-08-04: Conditions for importation from the continental United States of plant material (other than potatoes) in association with soil and related matter. All soil and related matter in association with plants requires a Phytosanitary Certificate stating freedom from pests named in this appendix.
- Requirement for nursery stock imported from areas infested with Centaurea solstitialis to enter Canada only after the importer obtains a CFIA issued Permit to Import indicating specific import requirements and conditions for pest status, handling and use of the commodity. Prior to the issuance of a Permit to Import, a facility inspection by the CFIA and verification of the importer's ability to meet permit requirements may be required. It is the importer's responsibility to apply for and obtain a Permit to Import.
- Recognition of Pest Free Areas; if the exporting country shows Centaurea solstitialis to be absent from the area from which the nursery stock is produced, then additional phytosanitary requirements may be waived.
- Recognition of nursery certification programs.
Trade implications
- Potential for reduction and/or loss of some import markets for Canada should exporting countries be unable to meet proposed phytosanitary requirements. Since 78% of nursery stock imports originate from infested areas of the U.S., there could be significant market losses of this commodity if Canadian import requirements cannot be met.
- The exporting country may have to devote resources to establish pest-free areas of production and the issuance of Phytosanitary Certificates.
Cost-effectiveness and Feasibility
Phytosanitary Certificates are currently issued by exporting countries for nursery stock and exporting countries currently comply with the phytosanitary requirements set out in D-95-26, including the pest-free area.
Appendix 4C: Import Data for Alfalfa seeds
Import data for alfalfa seeds from countries where Centaurea solstitialis (yellow star-thisle) is present (2006-2008).
Value in Canadian Dollars ($)
| Exporting country/US states | 2006 | 2007 | 2008 |
|---|---|---|---|
| Idaho | 4,772,722 | 1580344 | 2799074 |
| California | 421,346 | 649464 | 197474 |
| Washington, state | 26,447 | 404826 | 199382 |
| South Dakota | 104,015 | 310093 | 57649 |
| Minnesota | 242,639 | 1107 | 20 |
| Tennessee | 7,357 | 62330 | 19394 |
| Michigan | 9,367 | 9130 | 9438 |
| Montana | 7,709 | 10583 | 18097 |
| Kentucky | 0 | 11841 | 14976 |
| Oregon | 50,023 | 1008 | 1355 |
| North Carolina | 23,119 | 10 | 1329 |
| Iowa | 58,787 | 53781 | 851 |
| Maine | 0 | 5 | 9 |
| Missouri | 0 | 1734 | 2403 |
| North Dakota | 0 | 0 | 17276 |
| Wyoming | 3,002 | 0 | 0 |
| Indiana | 2,859 | 1487 | 0 |
| Wisconsin | 1,962 | 25049 | 925 |
| Maryland, New York | 12 | 30 | 25 |
| Subtotal: | 5,731,366 | 3,122,822 | 3,339,677 |
| Exporting country/US states | 2006 | 2007 | 2008 |
|---|---|---|---|
| France | 27 | 2529 | 52220 |
| Italy | 23,986 | 21697 | 69814 |
| Spain | 0 | 3662 | 0 |
| Netherlands | 125 | 12 | 33081 |
| Germany | 0 | 30812 | 42065 |
| Austria | 0 | 11101 | 6046 |
| Subtotal: | 24,138 | 69,813 | 203,226 |
| Exporting country/US states | 2006 | 2007 | 2008 |
|---|---|---|---|
| Argentina | 56 | 3122 | 0 |
| Uruguay | 2,524 | 0 | 0 |
| Subtotal: | 26,718 | 3,122 | 0 |
| Exporting country/US states | 2006 | 2007 | 2008 |
|---|---|---|---|
| Total: | 5,758,084 | 3,195,757 | 3,542,903 |
Quantity in kilograms (KGM)
| Exporting country/US states | 2006 | 2007 | 2008 |
|---|---|---|---|
| Idaho | 955171 | 327406 | 440995 |
| California | 114149 | 166799 | 43479 |
| Washington, state | 5457 | 80165 | 45398 |
| South Dakota | 44600 | 90867 | 17010 |
| Minnesota | 96537 | 260 | 9 |
| Tennessee | 1814 | 12412 | 2653 |
| Michigan | 1317 | 999 | 8769 |
| Montana | 971 | 2158 | 5275 |
| Kentucky | 0 | 1999 | 1950 |
| Oregon | 21900 | 198 | 134 |
| North Carolina | 5936 | 3 | 181 |
| Iowa | 11430 | 5967 | 115 |
| Maine | 0 | 1 | 1 |
| Missouri | 0 | 272 | 465 |
| North Dakota | 0 | 0 | 3969 |
| Wyoming | 590 | 0 | 0 |
| Indiana | 579 | 703 | 0 |
| Wisconsin | 272 | 3674 | 114 |
| Maryland, New York | 2 | 6 | 1 |
| Subtotal: | 1,260,725 | 693,889 | 570,518 |
| Exporting country/ US states | 2006 | 2007 | 2008 |
|---|---|---|---|
| France | 19 | 180 | 6335 |
| Italy | 6013 | 3421 | 21212 |
| Spain | 0 | 600 | 0 |
| Netherlands | 10 | 2 | 3200 |
| Germany | 0 | 3451 | 3975 |
| Austria | 0 | 2149 | 1000 |
| Subtotal: | 6,042 | 9,803 | 35,722 |
| Exporting country/ US states | 2006 | 2007 | 2008 |
|---|---|---|---|
| Argentina | 11 | 513 | 0 |
| Uruguay | 325 | 0 | 0 |
| Subtotal: | 336 | 513 | 0 |
| Exporting country/US states | 2006 | 2007 | 2008 |
|---|---|---|---|
| Total: | 1 267 103 | 704 205 | 606 240 |
Source: Statistics Canada 2009
Appendix 4D: Import Data for Hay and Straw
Import data for hay and straw from countries where Centaurea solstitialis (yellow star-thisle) is present (2006-2008)
Value in Canadian Dollars ($)
| Exporting country/US states | 2006 | 2007 | 2008 |
|---|---|---|---|
| Washington, state | 5,056,544 | 5,685,096 | 6,397,081 |
| North Carolina | 272,458 | 316,202 | 212,617 |
| North Dakota | 856,800 | 326,304 | 180,360 |
| Illinois | 40,714 | 114,980 | 111,185 |
| Oregon | 4,875 | 1,750 | 18,492 |
| New York | 8,984 | 10,260 | 16,288 |
| Michigan | 2,656 | 30,169 | 458 |
| All other statesTable Note 1 | 31,374 | 33,178 | 50,069 |
| Mexico Germany Ghana | 9,709 | 28,495 | 23,099 |
| United Kingdom | 5,796 | 11,226 | 4,855 |
| Subtotal: | 6,289,910 | 6,557,660 | 7,014,504 |
| Exporting country/US states | 2006 | 2007 | 2008 |
|---|---|---|---|
| Washington, state | 3,849,245 | 4,045,927 | 4,115,211 |
| Wisconsin | 107,918 | 238,895 | 386,684 |
| Oregon | 1,086,048 | 630,851 | 330,770 |
| Ohio | 250,851 | 262,863 | 303,452 |
| Pennsylvania | 22,699 | 20,272 | 101,167 |
| New Jersey | 44,162 | 37,891 | 48,900 |
| Nebraska | 29,892 | 24,792 | 48591 |
| Kansas | 0 | 13,410 | 46,197 |
| Colorado | 32,133 | 26,652 | 28,890 |
| Kentucky | 619 | 30 | 15,743 |
| Iowa | 412 | 483 | 13,852 |
| California | 12740 | 23511 | 9755 |
| Arizona | 134345 | 131764 | 3429 |
| New York | 24965 | 1808 | 228 |
| Idaho | 6317 | 55099 | 53 |
| All other states Table Note 2 | 27704 | 17677 | 21449 |
| Chile | 0 | 41354 | 32946 |
| Albania | 0 | 41354 | 32946 |
| Germany, France, Colombia | 3592 | 10030 | 3888 |
| Subtotal: | 5,633,642 | 5,583,309 | 5,511,205 |
Source: Industry Canada 2009
Appendix 5A: Pest Risk Assessment Summary for Crupina vulgaris (common crupina)
- Identity of Organism
- Organism Status
- Current Regulatory Status
- Probability of Entry
- Probability of Establishment
- Probability of Spread
- Potential Economic Consequences
- Potential Environmental and Social Consequences
- Uncertainty
- Conclusion
- Technical Issues for Consideration
Identity of Organism
Name: Crupina vulgaris Cass. (Asterales: Asteraceae)
Synonyms: Centaurea crupina L., Serratula crupina
English common names: Common crupina, crupina, bearded-creeper, starry scabious (Darbyshire, 2003; CABI, 2006)
French common names: Crupine, crupine vulgaire (Darbyshire 2003)
Description: Crupina vulgaris Cass. (Asterales: Asteraceae) is a facultative winter annual weed that is native to the Mediterranean region and has been recently (1968) introduced in the United States in Washington, Idaho, Oregon, California and Massachusetts. It is a serious weed of pastures, grasslands, rangelands, hayfields, and undisturbed roadsides and waste places.
Crupina vulgaris grows a slender taproot from one to several metres deep. The main flowering stem can range in height from 0.3-1.2 m. The top of the main flowering stem branches into 1-15 branches. In addition to short flowering branches at the top of the main flowering stem, additional flowering branches are produced in upper leaf axils.
Three to 130 capitula (flower heads) are produced per plant. Each capitulum bears 3-8 sterile flowers and 1-8 fertile flowers, with a normal combination of three sterile and two fertile flowers. Rose or purple petals partially protrude from the scaly floral bracts of the capitulum.
Seeds are cypselae with a dense pappus of blackish-brown bristles at the apex. Cypselae are a type of achene, a one-celled, one-seeded dry, hard fruits that do not open when ripe. The shape of the achene is roughly cylindrical, tapering at the base, and measuring 3-5 X 1.5-3 mm. The surface of the achene is black or silvery beige and covered with fine hairs.
Organism Status
Current native distribution of Crupina vulgaris is centred around the Mediterranean, ranging from southern Europe (Iberian Peninsula, France, Italy, Greece), across Türkiye, and extending as far east as Xinjiang Province of China in Asia. In Africa, it is also considered native to Morocco, Libya and Algeria.
Crupina vulgaris is not present in Canada, but is invasive in several U.S. states (California, Oregon, Washington, and Idaho (USDA, NRCS 2009)).
The first successful introduction of Crupina vulgaris in the U.S. was discovered in Idaho in 1968. Later discoveries were from Sonoma County, California, in 1976 (declared eradicated in 1982), Chelan County, Washington, in 1983, Umatilla County, Oregon, in 1987, Sonoma County, California, in 1989 (a re-discovery), Modoc County, California, in 1990, and Wallowa County, Oregon and locations in Nez Perce County and the Snake River Canyon, Idaho, in 1995. It is estimated that Crupina vulgaris has invaded over 26,000 ha in Idaho, Oregon, California and Washington. The major populations of Crupina vulgaris remain widely separated and discrete, despite local expansion and dispersal to new satellites.
Two reports of Crupina vulgaris in Canada appear to be erroneous. Zamora et al. (1989) state that common crupina was found in British Columbia (no further details given). Garnatje et al. (2002) indicate, however, that this report appears to be in error, based on a personal communication reference with R. Cranston (a weed specialist previously with the B.C. Ministry of Agriculture and Food). Secondly, Thill et al. (1999) state that an infestation of common crupina was found near Pincher Creek, British Columbia, Canada, and eradicated between 1980 and 1986 (Pincher Creek is actually located in the province of Alberta). The personal communication reference for this statement, however, has indicated that he has no knowledge of an occurrence of common crupina in Canada (R. Cranston, previously of B.C. Ministry of Agriculture and Food, pers. comm. with K. Castro, Apr. 4, 2006). Furthermore, there are no records for common crupina in the municipal district of Pincher Creek (K. Cooley, Agricultural Fieldman, M.D. of Pincher Creek, pers. comm. with K. Castro, Apr. 4, 2006). Without further evidence to the contrary, common crupina is considered absent from Canada.
Current Regulatory Status
Crupina vulgaris is not currently regulated under the Plant Protection Act. Under the Seeds Act, Weed Seeds Order, 2005, it is listed as a Class 1 Prohibited Noxious Weed. All seed sold or offered for sale in Canada, as well as imported into Canada for the purpose of planting must be free of prohibited noxious weed seeds as set out in the Weed Seeds Order. At the provincial level, Crupina vulgaris is regulated in British Columbia.
In the United States, Crupina vulgaris is on the USDA's Regulated Pest List (USDA, APHIS 2009) and the Federal Noxious Weed List. It is also considered a noxious weed or plant pest in 15 states: Alabama, California, Colorado, Florida, Idaho, Massachusetts, Minnesota, Montana, Nevada, North Carolina, Oregon, South Carolina, South Dakota, Vermont and Washington (USDA, NRCS 2009).
Probability of Entry
| Type of pathway | Specific pathways |
|---|---|
| Natural dispersal | Crupina vulgaris achenes can be dispersed by one or more natural means. (Achenes are small, one-seeded dry fruits. In the case of Crupina vulgaris, they resemble a seed.) Wind dispersal of the large, heavy achenes is limited to approximately 2 m or less. Achenes may be transported up to 15 m by rodents, which collect them in food caches, and 100 m or more by cattle and deer, on hooves and hair. Seeds have also survived passage through the digestive tracts of most animals except sheep. Water is a potential dispersal mechanism, although riparian zones are not a major habitat of Crupina vulgaris. |
| Intentional introduction | Achenes may be transported by recreationists or tourists as a curiosity because they are similar in appearance to a fly fishing lure. |
| Unintentional introduction | Canada imports live sheep, sheep wool, live cattle, hay and other forage products from the four states in which Crupina vulgaris is present. These imports represent potential unintentional pathways for this species into Canada. Crupina vulgaris could also be accidentally introduced in association with contaminated fabric, screens, farm machinery, trucks, rail cars, ballast soil, and/or sheep dogs moving across the border from the United States into Canada. Recreationists or tourists may unintentionally transport seeds of Crupina vulgaris on clothing and equipment. |
Probability of Establishment
If introduced, Crupina vulgaris would most likely become established in parts of Canada. Based on current distribution, Crupina vulgaris could establish in five plant hardiness zones in Canada (zones 4-8) (Figure 1). This species may be able to establish in harsher climatic zones, an area of uncertainty that could be addressed through scientific research.
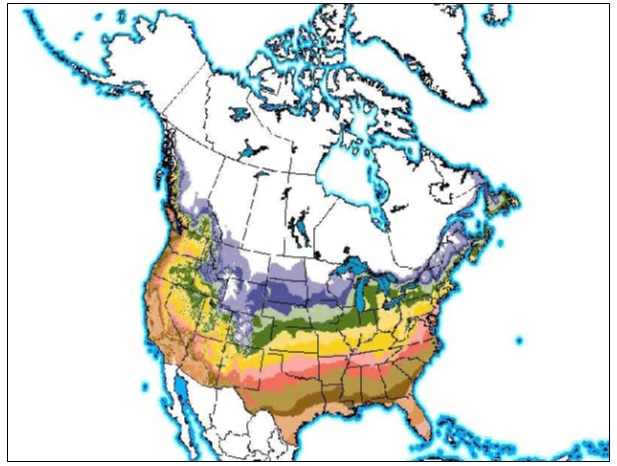
Description of Figure 1:
This image is a map of Nord America showing the hardiness zones from 1 to 11 in different colours.
Probability of Spread
Crupina vulgaris reproduces by seed only. Relative to many other weedy species, Crupina vulgaris has a low seed output. Seed output ranges from 3-27 per plant in dry grasslands to as many as 850 per plant under favourable conditions (Roché 1996, cited in Roché and Thill 2001). Despite relatively low seed output, the large, heavy seeds have a relatively high success rate (Roché and Thill 2001). Dispersal potential is also greatly amplified by human-mediated dispersal pathways (Ising 1937; Sorrie and Sommers 1999). In the United States, Crupina vulgaris has proven to be invasive, and is a serious weed of grasslands, hayfields, pastures and rangelands (CABI 2006). The ability of Crupina vulgaris to establish in dry, sparsely vegetated areas and to compete with more desirable species are additional factors favouring the spread of this species.
Potential Economic Consequences
There is a high potential for serious economic consequences if Crupina vulgaris were to be introduced into Canada. Depending on the level of infestation and the potential range of the species, Crupina vulgaris could have serious negative economic impacts on at least two major industries in Canada: forage and livestock production (AAFC 2007). The marketing of seed commodities could also be affected, although it is considered unlikely that Crupina vulgaris would be found in cleaned seed lots.
Potential Environmental and Social Consequences
Crupina vulgaris contributes to the degradation of native and managed plant communities. It can be highly competitive and dominate sites, displacing other plant species and reducing biodiversity in the ecosystem. It also increases the risk of soil erosion (CABI 2006). Crupina vulgaris is not considered to have sociological impacts on aesthetics, recreation or property values (CABI 2005).
Uncertainty
There are three main areas of uncertainty for this risk assessment. One is the climatic amplitude for Crupina vulgaris. Further refinement of the range of temperatures over which Crupina vulgaris can survive would allow a more precise definition of the potential range of Crupina vulgaris in Canada. Its preference for warmer, more Mediterranean-type climates may limit its potential for invasiveness in Canada. Similarly, rainfall regime tolerances should be clarified, i.e., whether or not high rainfall (greater than 950 mm) is a limiting factor for Crupina vulgaris. Secondly, there is little information on economic impacts of Crupina vulgaris elsewhere where introduced. While it is known to impact livestock and forage production, the monetary value of potential economic impacts of Crupina vulgaris in Canada would be difficult to determine. Lastly, if introduced, Crupina vulgaris could potentially have negative impacts on species at risk in Canada, but it is unknown at this time if, how, or to what degree these impacts might manifest themselves.
Conclusion
It is considered likely that, without specific measures, Crupina vulgaris will be introduced into Canada via one or more of several pathways that have been identified for this species. British Columbia appears to be at the greatest risk, due to the combined factors of imported sheep, cattle and hay from states in which Crupina vulgaris occurs, as well as the availability of suitable habitats. However, it is possible that pathways for Crupina vulgaris could lead to its introduction in other parts of Canada as well.
If Crupina vulgaris became established in Canada, it would have negative economic impacts, especially to the hay and livestock industries. However, its limited natural dispersal ability and relatively low seed production suggest that, if detected early, eradication could be successful.
It is recommended that Crupina vulgaris be added to the List of Regulated Pests by Canadato prevent what would otherwise be a likely introduction of a damaging invasive plant species. This species should be regulated because 1) it is not yet present in Canada, 2) if detected early, there is a relatively good chance of eradication, 3) its potential economic impact is high, 4) no economic benefits of this species have been identified, and 5) it is known to have negative impacts on biodiversity in the areas it invades. Regulation of this species under the Plant Protection Act would also complement its regulation as a prohibited noxious weed under the Seeds Act.
Technical Issues for Consideration
More scientific research is needed to determine if this species is able to establish in harsher climatic zones and if it will have deleterious effects on endangered species and species at risk.
Appendix 5B: Risk Management Considerations for Crupina vulgaris (common crupina)
- Values at Risk
- Potential Mitigation Measures for Natural Means of Dispersal
- Potential Mitigation Measures for Intentional Introduction Pathways
- Potential Mitigation Measures for Non-intentional Introduction Pathways
Values at Risk
No values at risk were identified.
Potential Mitigation Measures for Natural Means of Dispersal
The natural dispersal of the achenes would remain fairly local as the seeds fall close to the parent plant and dispersal by wildlife occurs only over short distances. These methods of dispersal are difficult to control. No specific mitigation measures are proposed for natural means of dispersal of Crupina vulgaris.
Potential Mitigation Measures for Intentional Introduction Pathways
Transport of plant parts by tourists and recreational users
A potential intentional pathway for the introduction of Crupina vulgaris is due to the appearance of the achenes. They are tapered and slightly iridescent, and with their stiff bristles at the wide end, somewhat resemble dry flies used for fishing.
Previous imports
It is not possible to assess the quantity of achenes that enter Canada for fly fishing, but it is most likely small.
Risk mitigation measures
Increase public awareness of the regulation and the risk posed by this plant and distribute awareness material at border crossings to the public. This is not considered effective by itself if the risk is high, but is a viable action if the level of risk is low.
Trade implications
As most imports of this type are non-commercial, there would be no trade implications.
Cost-effectiveness and Feasibility
A public awareness campaign is the most feasible option, whereas inspection would be very disruptive, difficult to implement, and expensive.
Potential Mitigation Measures for Non-intentional Introduction Pathways
Hay and Straw
Previous imports
- The total value of hay and straw imports from 2006 to 2008 was over $ 44 million; over 84% of this value came from the U.S. (Industry Canada, 2009).
- During the same period, large quantities of hay and straw (about $31 million value) were imported from Washington and Oregon while smaller amounts were imported from Idaho and California (just over $111,000), all states where Crupina vulgaris is present. This represents the majority of hay and straw imported into Canada. Since 2002, imports of hay and straw have remained relatively constant (Industry Canada, 2009).
- The level of risk associated with imports of hay and straw into Canada is moderate to high. The majority of hay and straw shipments do not originate from counties in Washington and Oregon where Crupina vulgaris is present. However, a smaller amount of imported hay and straw does originate in the infested areas of Idaho and California. The associated risk will increase if Crupina vulgaris continues to spread. As Crupina vulgaris is found not only in range land but also non-crop disturbed areas such as field edges and roadsides, land use changes such as road building and increased recreational use will increase the risk of spread.
Potential risk mitigation measures
Regulate Crupina vulgaris under the Plant Protection Act as a quarantine pest by adding it to the List of Pests Regulated by Canada. This will:
- Enable inspectors to take appropriate action for the purposes of eradicating the pest or preventing its spreadFootnote 1.
- Require exporters to obtain a Phytosanitary Certificate with or without an Additional Declaration stating freedom from Crupina vulgaris. (Note: Recognition of Pest Free Areas – if Crupina vulgaris can be shown to be absent in the area from which the field crop product was propagated, then risk is negligible and additional phytosanitary requirements may be waived.)
Additional requirements may include:
- A Permit to Import indicating specific import requirements and conditions for the pest status, handling and use of the commodity. It is the importer's responsibility to apply for and obtain the permit.
- For hay or straw intended for use as a biofuel stock, heat treatment or pelletization could be required.
Trade implications
- Regulating Crupina vulgaris as a quarantine pest would align Canada with the U.S. where this species is already on their Regulated Pest List.
- This measure will facilitate trade of hay and straw with the states of California, Oregon and Washington, where Crupina vulgaris is regulated.
- NPPOs of exporting countries will need to devote resources towards inspection of hay and straw commodities and issuance of Phytosanitary Certificates. Most straw and some hay from the four U.S. states where Crupina vulgaris is present currently requires phytosanitary certification. Inspectors in exporting countries need to be able to identify seeds of Crupina vulgaris that could attach themselves to hay and straw. NPPOs of the exporting country need to ensure freedom of Crupina vulgaris in hay and straw, otherwise CFIA can refuse import.
- Since most of hay and straw imports come from the U.S., and the majority from California, Oregon, Idaho and Washington where Crupina vulgaris is considered invasive, there could be significant market losses of this commodity if it is found to be contaminated.
- The U.S. could object to the new requirements, necessitating negotiation.
Cost-effectiveness and Feasibility
Regulation under the Plant Protection Act is already in place for other pests regulated by Canada. Minor costs will be incurred for training of inspectors for weed identification. Costs may be incurred during the negotiation of requirements with the U.S. or other affected trading partners.
Seed
Previous imports
Between 2004 and 2008, an average of $42 million worth of seed was imported into Canada from the four US states where Crupina vulgaris occurs.
Risk mitigation measures
Crupina vulgaris seeds are not likely to contaminate seed as it does not tolerate cultivation and would not be prevalent in crop fields, although it can become established along field edges. However, it also matures earlier than crop plants and the achenes are easily removed by screening due to its bristly pappus. Seeds suspected of being contaminated with Crupina vulgaris could be re-cleaned to remove the contaminant seeds.
Crupina vulgaris is listed as a Class 1 prohibited noxious weed in the Weed Seeds Order Footnote 2, and therefore, due to current regulations and requirements under the Seeds Act, imported seed is unlikely to be a pathway.
Other options include regulating Crupina vulgaris as a quarantine pest under the Plant Protection Act. Adding this species to the List of Pests Regulated by Canada (CFIA, 2009) would:
- Prevent the importation, movement, and cultivation of this species in Canada.
- Enable inspectors to take appropriate action for the purposes of eradicating the pest or preventing its spreadFootnote 3.
Regulatory actions under the Plant Protection Act could include one or more of the following:
- Exporters could be required to obtain a Phytosanitary Certificate stating freedom from Crupina vulgaris.
- Exceptions may be made for the importation of preserved specimens for scientific research purposes by recognized herbaria and research facilities and the importation of seed for research in containment facilities.
- Could negotiate phytosanitary agreements to certify imports from pest free areas and/or recognize noxious weed certification in countries or states of origin.
Trade implications
NPPOs of exporting countries and seed certification agencies currently devote resources towards inspection of seed lots and issuance of Phytosanitary Certificates. Laboratories in foreign countries will need to be able to identify seeds of Crupina vulgaris within a seed sample. Exporters need to ensure freedom of Crupina vulgaris in seed lots, otherwise CFIA can refuse import.
Field Crops Not Intended for Propagation
Included in this section are cereal grains, oilseeds, pulses, forage, and new crops not covered under the Seed section above.
Previous imports
Between 2004 and 2008, an average of $1.7 million worth of field crops not intended for propagation were imported into Canada from the four U.S. states where Crupina vulgaris occurs.
Risk mitigation measures
Crupina vulgaris seeds are not likely to be a contaminant as they do not tolerate cultivation and would not be prevalent in crop fields, although Crupina vulgaris can become established along field edges. However, it also matures earlier than crop plants and the achenes are easily removed by screening due to its bristly pappus. Grain suspected of being contaminated with Crupina vulgaris could be re-cleaned to remove the contaminant seeds.
Livestock
Previous imports
- The total value of sheep and cattle imports varies widely by year. Live sheep imports ranged from $30,244 in 2004 to $2,106,614 in 2006 (Industry Canada, 2007). Live cattle imports have ranged from $3 million in 2004 to $267 million in 2000 (Industry Canada, 2007).
- Between 1999 and 2008, almost 260,000 sheep, goats, and cattle were imported from the four states where Crupina vulgaris is present (CFIA, internal data).
Potential risk mitigation measures
Regulate Crupina vulgaris under the Plant Protection Act as a quarantine pest by placing it on the List of Pests Regulated by Canada. This would:
- Enable inspectors to take appropriate action for the purposes of eradicating the pest or preventing its spread Footnote 4.
- Require importers to obtain certification stating freedom from Crupina vulgaris.
Trade implications
- NPPOs of the exporting countries would need to devote resources towards inspection of livestock commodities. Inspectors in exporting countries would need to be able to identify seeds of Crupina vulgaris that could attach themselves to livestock. Exporters would need to ensure freedom of Crupina vulgaris on livestock, otherwise CFIA could refuse import.
- Since almost all imports of livestock come from the U.S., including California, Oregon, Washington and Idaho where Crupina vulgaris is invasive, there could be significant market losses of this commodity if it is found to be contaminated.
Cost-effectiveness and Feasibility
- The regulation of Crupina vulgaris on livestock is not considered feasible. Inspection of livestock for Crupina vulgaris achenes would be disruptive, difficult to implement, and expensive.
- This measure is not currently in place for other plant pests regulated by Canada. Substantial costs would be incurred if an inspection system for invasive plant seeds on livestock were to be implemented.
Raw Wool and Raw Skin
Previous imports
- The total value of sheep and goat raw wool imports was around $3.05 million in 2008; 76% of this value came from U.S. (Industry Canada 2009).
- Between 1997 and 2006, significant quantities of raw wool and raw skins were imported from California, where Crupina vulgaris is present.
Potential risk mitigation measures
No measure is required:
- The vegetative matter is removed from commercial wool by carbonization Footnote 5.
- Raw wool and raw hides imported into Canada from all countries require CFIA inspection. They must be free from feces, blood, ectoparasites and dirt (plant and plant parts are considered to be dirt) or they will be refused entry (Health of Animals Act, Directive AHPD-DSAE-2001-1-1 (CFIA 1990)). No further measures are considered necessary.
Vehicles and Used Farm Machinery
Previous imports
- A considerable volume of vehicles cross the U.S.-Canada border every year.
- Information is not available on the volume of imports of used farm machinery.
Potential Risk mitigation measures
Enforcement of the Directive 95-26: "Phytosanitary requirements for soil and related matter, and for items contaminated with soil and related matter" (CFIA 2010).
In 2003, the Canada Border Services Agency (CBSA) assumed responsibility for the initial import inspection services in respect to the Acts and Regulations administered by the CFIA to the extent that they are applicable at Canadian border points. The inspection of goods that may be contaminated with soil are among the responsibilities that were transferred to the CBSA in 2003. The Food, Plant and Animals Programs Section of the CBSA is currently finalizing its Standard Operating Procedures (SOP) concerning the "Inspection of Imported Goods Potentially Contaminated with Soil." This SOP provides the CBSA's Border Services Officers with formal procedures for the inspection and disposition of goods that may be contaminated with soil, including used agricultural machinery and vehicles.
Trade implications
- Exporting countries will need to devote resources towards inspection of used vehicles and farm machinery. Inspectors in exporting countries need to be able to identify seeds of Crupina vulgaris that could attach themselves to used vehicles and farm machinery. Exporters need to ensure freedom of Crupina vulgaris, otherwise CFIA can refuse import.
- The number of used vehicles and farm machinery imported from all states where Crupina vulgaris is present is unknown; however, there could be significant market losses of this commodity if it is found to be contaminated.
Cost-effectiveness and Feasibility
As vehicles and used farm machinery are already required to be free from soil, this would address one of the likely sources of Crupina vulgaris contamination, resulting in no additional costs. However, ensuring that used vehicles and farm machinery are free from Crupina vulgaris achenes not in association with soil would require additional resources and training as well as renegotiating the Memorandum of Understanding (MOU) between CFIA and CBSA.
Used Recreational Equipment and Clothing
Previous imports
It is not possible to assess the quantity of used recreational equipment and clothing that enters Canada, but it is most likely significant.
Risk mitigation measures
Increase public awareness of the regulation and the risk posed by this plant and distribute awareness material at border crossings to the public. This is not considered effective by itself if the risk is high, but is a viable action if the level of risk is low.
Trade implications
As most imports of this type are non-commercial, there would be no trade implications.
Cost-effectiveness and Feasibility
A public awareness campaign is the most feasible option, whereas inspection would be very disruptive, difficult to implement, and expensive.
Appendix 5C: Import Data for Field Crops
Import data for field crops not intended for propagation originating from U.S. states where Crupina vulgaris is present (2006-2008).
Value in Canadian Dollars ($)
| Exporting U.S. State | 2006 | 2007 | 2008 |
|---|---|---|---|
| Washington | 433,287 | 2,317,101 | 1,825,056 |
| Oregon | 1,508,006 | 422,496 | 1,707 |
| Idaho | 0 | 279,928 | 0 |
| California | 4,974 | 8,701 | 5,260 |
| Subtotal: | 1,946,267 | 3,028,226 | 1,832,023 |
| Exporting U.S. State | 2006 | 2007 | 2008 |
|---|---|---|---|
| Oregon | 68 | 82 | 94 |
| Subtotal: | 68 | 82 | 94 |
| Exporting U.S. State | 2006 | 2007 | 2008 |
|---|---|---|---|
| Idaho | 119,901 | 152 | 147 |
| California | 4,600 | 5,043 | 4,512 |
| Oregon | 0 | 5 | 449 |
| Washington | 4,787 | 0 | 21 |
| Subtotal: | 129,288 | 5,200 | 5,129 |
| Exporting U.S. State | 2006 | 2007 | 2008 |
|---|---|---|---|
| Washington | 0 | 5 | 689 |
| California | 552 | 0 | 67 |
| Idaho | 2 | 29 | 8 |
| Oregon | 15,805 | 27,901 | 11,700 |
| Subtotal: | 16,359 | 27,935 | 12,464 |
| Exporting U.S. State | 2006 | 2007 | 2008 |
|---|---|---|---|
| California | 1,247 | 251 | 374 |
| Oregon | 0 | 289 | 0 |
| Subtotal: | 1,247 | 540 | 374 |
| Exporting U.S. State | 2006 | 2007 | 2008 |
|---|---|---|---|
| California | 360,752 | 593,333 | 1,263,130 |
| Oregon | 112,846 | 198,547 | 476,661 |
| Washington | 1,619 | 3,780 | 482 |
| Idaho | 2,984 | 44,047 | 22,951 |
| Subtotal: | 478,201 | 839,707 | 1,763,224 |
| Exporting U.S. State | 2006 | 2007 | 2008 |
|---|---|---|---|
| Washington | 41,915 | 180 | 2,043 |
| Idaho | 0 | 0 | 4,481 |
| Oregon | 7,994 | 4,441 | 26,005 |
| California | 64,492 | 36,753 | 165,357 |
| Subtotal: | 114,401 | 41,374 | 197,886 |
| Exporting U.S. State | 2006 | 2007 | 2008 |
|---|---|---|---|
| Idaho | 2,278,429 | 6,622,763 | 22,504,709 |
| Washington | 447,941 | 2,152,692 | 4,950,358 |
| Oregon | 893,316 | 2,272,041 | 1,233,939 |
| California | 257,817 | 155,498 | 49,527 |
| Subtotal: | 3,877,503 | 11,202,994 | 28,738,533 |
| Exporting U.S. State | 2006 | 2007 | 2008 |
|---|---|---|---|
| California | 489,204 | 523,238 | 565,491 |
| Oregon | 285,632 | 652,761 | 349,070 |
| Washington | 95,604 | 201,037 | 167,368 |
| Idaho | 30,668 | 8,062 | 147,337 |
| Subtotal: | 901,108 | 1,385,098 | 1,229,266 |
| Exporting U.S. State | 2006 | 2007 | 2008 |
|---|---|---|---|
| Total: | 7,464,442 | 16,531,156 | 33,778,993 |
Quantity in metric tonnes (TNE)
| Exporting U.S. State | 2006 | 2007 | 2008 |
|---|---|---|---|
| Washington | 2,200 | 8,916 | 5,334 |
| Oregon | 9,463 | 2,009 | 1 |
| Idaho | 0 | 1,225 | 0 |
| California | 8 | 17 | 10 |
| Subtotal: | 11,671 | 12,167 | 5,345 |
| Exporting U.S. State | 2006 | 2007 | 2008 |
|---|---|---|---|
| Oregon | 0 | 0 | 0 |
| Subtotal: | 0 | 0 | 0 |
| Exporting U.S. State | 2006 | 2007 | 2008 |
|---|---|---|---|
| Idaho | 531 | 2 | 0 |
| California | 18 | 11 | 11 |
| Oregon | 0 | 0 | 2 |
| Washington | 32 | 0 | 0 |
| Subtotal: | 581 | 13 | 13 |
| Exporting U.S. State | 2006 | 2007 | 2008 |
|---|---|---|---|
| Washington | 0 | 0 | 1 |
| California | 1 | 0 | 0 |
| Idaho | 0 | 0 | 0 |
| Oregon | 120 | 84 | 81 |
| Subtotal: | 121 | 84 | 82 |
| Exporting U.S. State | 2006 | 2007 | 2008 |
|---|---|---|---|
| California | 4 | 1 | 0 |
| Oregon | 0 | 1 | 0 |
| Subtotal: | 4 | 2 | 0 |
| Exporting U.S. State | 2006 | 2007 | 2008 |
|---|---|---|---|
| California | 100,535 | 172,970 | 241,323 |
| Oregon | 108,541 | 43,881 | 347,798 |
| Washington | 367 | 825 | 107 |
| Idaho | 1,701 | 15,564 | 6,994 |
| Subtotal: | 211,144 | 233,240 | 596,222 |
| Exporting U.S. State | 2006 | 2007 | 2008 |
|---|---|---|---|
| Washington | 14 | 0 | 1 |
| Idaho | 0 | 0 | 5 |
| Oregon | 18 | 3 | 23 |
| California | 21 | 14 | 130 |
| Subtotal: | 53 | 17 | 159 |
| Exporting U.S. State | 2006 | 2007 | 2008 |
|---|---|---|---|
| Idaho | 2,968 | 6,597 | 5,003 |
| Washington | 692 | 2,068 | 4,654 |
| Oregon | 670 | 7,963 | 3,640 |
| California | 107 | 495 | 187 |
| Subtotal: | 4,437 | 17,123 | 13,484 |
| Exporting U.S. State | 2006 | 2007 | 2008 |
|---|---|---|---|
| California | 1,063 | 1,246 | 616 |
| Oregon | 130 | 524 | 226 |
| Washington | 442 | 745 | 332 |
| Idaho | 32 | 5 | 234 |
| Subtotal: | 1,667 | 2,520 | 1,408 |
| Exporting U.S. State | 2006 | 2007 | 2008 |
|---|---|---|---|
| Total: | 229,678 | 265,166 | 616,713 |
Source: Industry Canada 2009
Appendix 5D: Import Data for Hay and Straw
Import data for hay and straw originating from U.S. states where Crupina vulgaris is present (2006-2008).
Value in Canadian Dollars ($)
| Exporting U.S. State | 2006 | 2007 | 2008 |
|---|---|---|---|
| California | 12,740 | 23,511 | 9,755 |
| Idaho | 6,317 | 55,099 | 53 |
| Oregon | 1,086,048 | 630,851 | 330,770 |
| Washington | 3,849,245 | 4,045,927 | 4,122,801 |
| Subtotal: | 4,954,350 | 4,755,388 | 4,463,379 |
| All U.S. States: | 5,630,050 | 5,532,203 | 5,481,961 |
| Exporting U.S. State | 2006 | 2007 | 2008 |
|---|---|---|---|
| California | 2,472 | 501 | 597 |
| Idaho | 0 | 0 | 0 |
| Oregon | 4,875 | 1,750 | 18,492 |
| Washington | 5,056,544 | 5,685,096 | 6,397,081 |
| Subtotal: | 5,063,891 | 5,687,347 | 6,416,170 |
| All U.S. States: | 6,629,605 | 6,938,311 | 7,498,629 |
| Exporting U.S. State | 2006 | 2007 | 2008 |
|---|---|---|---|
| CA, ID, OR, WA | $10,018,241 | $10,442,735 | $10,879,549 |
| All U.S. States | $12,259,655 | $12,470,514 | $12,980,590 |
| All Other Countries | $2,317,592 | $2,186,384 | $2,250,154 |
| % From U.S. | 84% | 85% | 85% |
Source: Industry Canada 2009
Appendix 6A: Pest Risk Assessment Summary for Dioscorea polystachya (Chinese yam)
- Identity of Organism
- Organism Status
- Current Regulatory Status
- Probability of Entry
- Probability of Establishment
- Probability of Spread
- Potential Economic Consequences
- Potential Environmental and Social Consequences
- Uncertainty
- Conclusion
- Technical Issues for Consideration
Identity of Organism
Name: Dioscorea polystachya Turcz. (Family Dioscoreaceae) (USDA-ARS, 2009)
Synonyms: Dioscorea batatas Decne., Dioscorea cayenensis Lam. var. pseudobatatas Hauman, Dioscorea decaisneana Carrière, Dioscorea doryphora Hance, Dioscorea opposita auct., Dioscorea oppositifolia auct., Dioscorea potaninii Prain & Burkill, Dioscorea rosthornii Diels, Dioscorea swinhoei Rolfe, Dioscorea trinervia Roxb. ex Prain & Burkill (Global Invasive Species Database, 2009; USDA-ARS, 2009)
English common names: Chinese yam, Chinese-potato, cinnamon-vine (USDA-ARS, 2009)
French common name: Igname de Chine (CAB International, 2007; USDA-ARS, 2009)
Description: The plants are vines growing from spindle-shaped tubers which are on long stalks and are deeply buried. The twining stems are up to 5 m in length. Small bulbils (less than 2 cm in diameter) are produced in the leaf axils. The lower leaves are alternate, becoming opposite higher up the stem and are 3–9 cm long × 3–11 cm wide. The petioles are as long as the blade. The blade is 7(–9)-veined, glabrous and 3-lobed. Inflorescences are borne in the leaf axils. The flowers are small, yellowish and have a cinnamon fragrance. Staminate and pistillate flowers are on separate plants. The fruit is an ovate capsule. The seeds are winged. Only one pistillate specimen has been documented from North America and it is assumed that the plants propagate vegetatively by means of the bulbils (FNA Editorial Committee, 1993+; Gleason and Cronquist, 1963).
Dioscorea polystachya is native to eastern Asia, where it is cultivated for its edible tubers. In North America, it is planted as a garden ornamental and has become naturalized throughout much of the eastern United States (FNA Editorial Committee, 1993+).
Organism Status
Dioscorea polystachya has been imported for sale in Canada on a very limited scale. The UBC Botanical Garden offered it for sale (as Dioscorea batatas) in their catalogue in 2006. There are no records of plants outside of cultivation. Based on this information, for the PRA area, this species is considered to be possibly present only in cultivation.
Current Regulatory Status
Dioscorea polystachya is not regulated in Canada. It is not regulated as a federal noxious weed in the U.S., nor is it regulated in any of the states (USDA-NRCS, 2009).
It is currently listed in the Southeast Exotic Pest Plant Council's Invasive Exotic Pest Plant List for Tennessee as a Rank 1-Severe Threat species, indicating that it is an exotic species that possesses characteristics of an invasive species and could spread easily into native plant communities and displace native vegetation (Tu, 2002).
Probability of Entry
Dioscorea polystachya is native to eastern Asia, where it is cultivated for its edible tubers. In North America, it is planted as a garden ornamental and has become naturalized throughout much of the eastern United States (FNA Editorial Committee, 1993+).
| Type of pathways | Specific pathways |
|---|---|
| Natural dispersal | Naturalspread is by seeds and aerial bulbils, but sexual reproduction by means ofseeds has not been detected in North America. The aerial bulbils can bespread by rodents who feed on them. Thispathway allows only local movement and is not likely to be a conduit forentry into Canada. |
| Intentional introduction | Plantshave been intentionally introduced into the U.S. as an ornamental ormedicinal plant. In eastern Asia it has been widely planted as a starchy foodtuber crop. This isthe most likely pathway for entry into Canada. |
| Unintentional introduction | As withany weed, introduction is possible via soil imported with nursery stock frominfested areas of the U.S. The riskfrom this pathway is likely to be minor, however. Asreproduction does not appear to occur through seed in North America,contaminated seed imported from the U.S. is not a potential pathway. Seed imported from other countries wheresexual reproduction does occur could be contaminated, however. The risk from this pathway is likely to beminor, though, as the plant is not known to be a weed of crops and so wouldprobably not be harvested along with seeds for planting. |
Probability of Establishment
Dioscorea polystachya is native to China (Anhui, Fujian, eastern Gansu, northern Guangdong, Guangxi, Guizhou, Hebei, Henan, Hubei, Hunan, Jiangsu, Jiangxi, Jilin, Liaoning, southern Shaanxi, Shandong, Sichuan, northern Yunnan and Zhejiang), Japan (Hokkaido, Honshu, Kyushu, Shikoku), Korea and Taiwan (USDA-ARS, 2009). The species is widely cultivated for food in temperate eastern Asia (Bailey and Bailey, 1976).
The species is introduced and cultivated as an ornamental, food and medicinal plant and is naturalized in temperate regions, including the U.S. (USDA-ARS, 2009). It is described as root hardy to USDA Plant Hardiness Zone 5 (Bailey and Bailey, 1976).
In its native range, Dioscorea polystachya grows in forests, scrub, herbaceous plant communities, on mountain slopes, along rivers and roadsides (Wu and Raven, 2000).
Dioscorea polystachya has not become established outside cultivation in Canada (CFIA, 2008; Scoggan, 1979).
Dioscorea polystachya has a wide range of environmental adaptability and few pests and predators in North America. It has a high degree of asexual reproductive vigour, and is difficult to manage once firmly established (Tu, 2002).
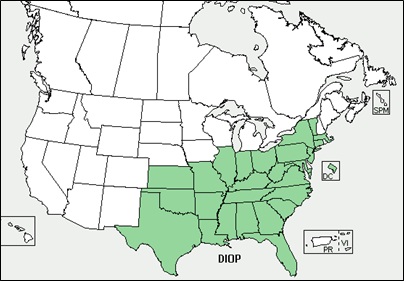
Description for Figure 1:
This map shows the range of Dioscorea polystachya in North America indicating its presence through the use of the colour green. The eastern half of the United States is almost completely covered in green along with a fifth of the lower western half.
Source : USDA-NRCS, 2009.
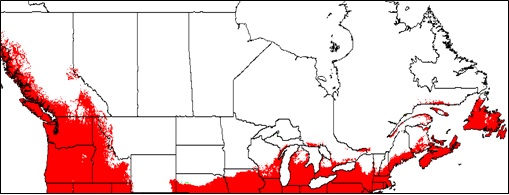
Description for Figure 2:
This image shows the potential range of Dioscorea polystachya in Canada and northern America through the use of a map. Red is used to indicate the regions in which Centaurea iberica could survive according to the Canadian Plant Hardiness Zones map, in this case NAPPFAST Hardiness Zones 5-9. In Canada, Dioscorea polystachya is likely to become weedy or invasive in parts of Canada, including southern and coastal British Columbia, southern Ontario, southern Quebec and parts of the Maritime Provinces. Most of the red can be found in the U.S., this includes the greater parts of the eastern and western coasts as well as areas located near the Great Lakes extending downwards beyond the image.
Source : USDA-NRCS, 2009.
Probability of Spread
Dioscorea polystachya is found in thickets, ravines, stream banks, creek bottoms, limesinks, granite outcrops, alluvial woods, roadsides, drainage canals, waste places and fence rows in its introduced range in the southeastern U.S. (FNA Editorial Committee, 1993+).
Initial infestations are generally associated with human disturbances, such as old home sites and along roadways. From these areas, the species can easily spread into nearby riparian areas and undisturbed habitats. In 1970, the species had not yet been documented as escaping from cultivation in the U.S., but by 1986 it had become naturalized (Tu, 2002).
As Dioscorea polystachya can grow in both disturbed and natural sites, habitats will not be a limiting factor for spread in Canada.
Potential Economic Consequences
Both the tuber and bulbils are edible, although the bulbils are not generally used as food. The edible tuber, which can measure up to 1 m long and weigh up to 2 kg or more, is flavourful and nutritious. The tubers are sometimes used in herbal medicine. The species is frequently planted for its ornamental value (Tu, 2002).
In forested areas, branches can be broken off of trees by the weight of the vines (ISSG, 2009).
Manual and mechanical methods of plant removal can effectively control small isolated patches of Dioscorea polystachya. However, these methods are extremely time and labour-intensive, as the large, deeply-buried tuber makes removal very difficult. Herbicide application appears to be the most effective means for controlling large infestations. Repeated treatments are usually necessary to completely kill large underground tubers (ISSG, 2009), which increases the cost.
Potential Environmental and Social Consequence
Dioscorea polystachya is a fast-growing vine that has the ability to rapidly invade undisturbed habitats, such as riparian forests. It spreads prolifically by asexual reproduction via bulbils. In the U.S., it reduces native species richness and abundance by outcompeting and eliminating native plant species. It thickly blankets adjacent vegetation, and excludes light from understory vegetation. It can break branches of large trees and shrubs. It is able to completely cover the ground so that the growth of native herbaceous ground cover is prevented (ISSG, 2009).
Uncertainty
Although it is listed on one nursery website there is no definite information to confirm that Dioscorea polystachya is being grown in gardens in Canada or if it is present. An online search found no other nurseries or seed suppliers that offer the species for sale in Canada.
Conclusion
Based on the outcome of this pest risk assessment, Dioscorea polystachya is likely to become weedy or invasive in parts of Canada, including southern and coastal British Columbia, southern Ontario, southern Quebec and parts of the Maritime Provinces. This plant should be considered for regulation under Canada's Plant Protection Act. Regulation under the Seeds Act may also be warranted, although seed is not currently considered to be a likely pathway for this species.
Technical Issues for Consideration
The very large number of species in Dioscorea complicates identification. There is one native species of this genus in southern Ontario, Dioscorea villosa L. This species is distinguishable from Dioscorea polystachya, with care and training.
Appendix 6B: Risk Management Considerations for Dioscorea polystachya (Chinese yam)
- History of invasiveness
- Potential Mitigation Measures for Natural Means of Dispersal
- Potential Mitigation Measures for Intentional Introduction Pathways
- Potential Mitigation Measures for Non-intentional Introduction Pathways
History of invasiveness
In the 25 or so years that Dioscorea polystachya has been present in the U.S., it has spread across many of the eastern states. In that short period of time, it has been identified as a species of concern by various state invasive plant councils:
- Alabama: Scattered and localized infestations in urban environments, managed forests, rights-of-way and aquatic/wetland environments; extensive and dense infestations in natural areas and parks in Alabama or severe invasion in an adjacent state (AIPC, 2007).
- Georgia: Moderate problem in natural areas (GEPPC, 2006).
- Kentucky: Exotic, invasive, spreads easily into native plant communities and displaces native vegetation; includes species which are or could become widespread in Kentucky (KY-EPPC , 2008).
- South Carolina: Limited infestations, posing a potential threat to natural areas and exhibiting invasive characteristics (SC-EPPC, 2008).
- Tennessee: Exotic, invasive, spreads easily into native plant communities and displace native vegetation) (TN-EPPC, 2001).
As described in Crooks and Soulé, 1999, some invasive plant populations have historically shown a "lag" period after first establishing, during which the population remains relatively small and has a low impact. Following the lag, the population then explodes, its range and impacts rapidly increasing. In brief, "past performance of an exotic is a poor predictor of potential population growth, range expansion and ecological impact" (Crooks and Soulé, 1999).
The CFIA is concerned that this may be the case with Dioscorea polystachya and that the early reports of invasiveness described above may be signs that the plant could become a much more significant threat in the future, both in the U.S. and in Canada.
Potential Mitigation Measures for Natural Means of Dispersal
Though populations are present in U.S. states that adjoin the U.S.-Canada border (see Figure 1), Dioscorea polystachya has only been recorded, within those states, in counties that are not immediately adjacent to the U.S.-Canada border (USDA-NRCS, 2009). Natural dispersal is therefore not expected to be an important factor in the spread of Dioscorea polystachya to Canada. If populations continue to spread in the U.S., however, dispersal of floating bulbils via waterways may become a concern and therefore the species can easily spread into nearby riparian areas. This means of spread would be extremely difficult to control.
Potential Mitigation Measures for Intentional Introduction Pathways
Plants for Planting Excluding Seed
Previous imports
Based on the information available in the CFIA's Import Permit System, Import Retrieval System, and information compiled at the CFIA's Import Service Centres, Dioscorea polystachya may have been recently imported into Canada. Three Permits to Import were issued by the CFIA over the last five years for cuttings of plants from the genus Dioscorea, but the precise species were not indicated. No plants from the genus Dioscorea are listed as available in Canadian nurseries (CNLA, 2009).
Potential risk mitigation measures
Non-regulatory measures
- Encourage voluntary cessation of the sale of Dioscorea polystachya. Voluntary cessation is not effective by itself, but could support other measures.
- Increase public awareness of the risk posed by Dioscorea polystachya, publish a factsheet online, and distribute awareness material to garden centers, botanical gardens, gardeners associations, horticulture industry groups, etc. Not considered effective by itself, but could support other measures.
Regulatory measures
Allow sale of Dioscorea polystachya with special conditions such as not to be grown close to natural areas and mandatory control of adventives. Allowing sale with special conditions is not considered effective because once grown in private gardens, CFIA does not have adequate resources to monitor.
Regulate Dioscorea polystachya under the Plant Protection Act as a quarantine pest:
- Prohibit importation of Dioscorea polystachya plants.Footnote 1
- Will require that importers of material from regions other than the continental U.S. specify the scientific name when applying for a Permit to Import plant and propagative material;
- Will require that the scientific name of all Dioscorea species be provided on the Phytosanitary Certificate for plants exported to Canada from the continental U.S.
Trade Implications
As described above under Previous imports, Dioscorea polystchya has only rarely been imported into Canada. Prohibiting the species should therefore not significantly impact Canadian importers and vendors. In addition, other ornamental species could potentially be substituted. If the intent is cultivation for consumption or for medicinal purposes, roots (processed or dried) could be imported rather than grown in Canada (see Section below. Plant parts for consumption or for medicinal use, for more information).
Cost-effectiveness and Feasibility
It may be useful for CFIA inspectors to check imported Dioscorea plants to ensure that Dioscorea polystachya is not being imported under an incorrect name or synonym. This is particularly the case with Dioscorea polystachya because it has been widely sold in the U.S. as Dioscorea polystachya. This would require training inspectors and providing them with identification material. As described above under Previous imports, however, Dioscorea plants are only rarely imported; the impact on CFIA resources should therefore be minimal.
Seed
Previous imports
There is no record in the information available in the CFIA's Import Permit System, Import Retrieval System, and data compiled at the CFIA's Import Service Centres that Dioscorea polystachya seed has ever been imported to Canada. Seed of other Dioscorea species is known to have been imported at least three times in recent years.
Potential risk mitigation measures
Regulate Dioscorea polystachya as a prohibited noxious weed (Class 1) under the Weed Seeds Order of the Seeds Act Footnote 2.
- This species meets the definitions for Class 1Footnote 3 species under the Weed Seeds Order.
- All imported and domestic seed lots must be free of prohibited noxious weed seeds. Imported seed lots require a certificate of analysis stating that the seed lot is free of all prohibited noxious weeds in order to be imported.
Regulate as a quarantine pest under the Plant Protection Act. Add this species to the List of Pests Regulated by Canada (CFIA 2009):
- This would prevent the importation, movement, and cultivation of this species in Canada. Currently, seed of many new crop species, such as field crops, can be imported without a Permit to Import or a Phytosanitary Certificate from throughout the world Footnote 4.
- It would enable inspectors to take appropriate action for the purposes of eradicating the pest or preventing its spread Footnote 5.
- Seed of horticultural plants is not within the scope of CFIA's current directive on plants for planting (D-08-04). Therefore phytosanitary requirements must be specified either in a revision of D-08-04 or in a new regulatory policy.
Sufficient information about the use of Dioscorea polystachya as a potential crop does not exist. If the proponent, located in Canada, needs to collect more information about the plant (e.g. to generate data for a determination of environmental safety), then confined research field trials under Part V of the Seeds Regulations could be authorized by the Plant Biosafety Office.
Trade Implications
As described above under Previous imports, Dioscorea polystachya seed has only rarely been imported to Canada. Prohibiting the species should therefore not significantly impact Canadian importers and vendors. In addition, other ornamental species could potentially be substituted. If the intent is cultivation for consumption or for medicinal purposes, roots (processed or dried) could be imported rather than grown in Canada (see Section below. Plant parts for consumption or for medicinal use).
Cost-effectiveness and Feasibility
The CFIA Seed Program is already in place to prevent the entry of prohibited noxious weeds. As well, as described in Section Appendix 6A, Probability of Entry, Dioscorea polystachya is not known to be a weed of crops. Negligible effort would therefore be required to identify the seeds or take action on non-compliant shipments because it would be unlikely to find Dioscorea polystachya seeds as a contaminant in sampled seed or grain lots.
It may be useful for the CFIA to occasionally sample imported Dioscorea seeds to ensure that Dioscorea polystachya seeds are not imported under an incorrect name or synonym. As described above under Previous imports, however, Dioscorea seeds are only rarely imported; the impact on CFIA resources should therefore be minimal.
Plant parts for consumption or for medicinal use
Background
Importers are required to specify the intended end-use (e.g., consumption, planting) of imported products when requesting a Permit to Import, and a Permit to Import issued for plant parts imported for consumption specifies that the plant parts cannot be used for propagation. However, once the plant parts are distributed, the CFIA has no control over how they are used. Dioscorea polystachya tubers imported for consumption could therefore conceivably be planted for cultivation as an ornamental, or to grow tubers for consumption or medicinal use.
Previous imports
Based on the information available in the CFIA's Import Permit System, Import Retrieval System, and information compiled at the CFIA's Import Service Centres, Dioscorea polystachya tubers for consumption or medicinal use may have been recently imported into Canada. Only one Permit to Import was issued by the CFIA over the last five years for tubers for consumption from the genus Dioscorea, but the precise species was not specified. No shipments of Dioscorea polystachya tubers are known to have been imported, though approximately a dozen shipments of tubers of other Dioscorea species have been imported in recent years.
Potential risk mitigation measures
Non-regulatory measures
Encourage voluntary cessation of the sale of Dioscorea polystachya. This would not be effective by itself, but could support other measures.
Regulatory measures
Regulate Dioscorea polystachya under the Plant Protection Act as a quarantine pest and, as a supporting measure, prohibit importation of tubers for consumption.
- Refuse to issue Permits to Import for viable plant parts of Dioscorea polystachya intended for consumption or medicinal use.
- Allow importation of plant parts of Dioscorea polystachya intended for consumption or medicinal use if they are processed in such as way as to render them non-viable.
- Will require that importers of material from the non-continental U.S. specify the scientific name when applying for a Permit to Import for plant parts for consumption.
Trade Implications
As described above under Previous imports, Dioscorea polystachya plant parts for consumption or medicinal use have only rarely been imported to Canada. Prohibiting the species should therefore not significantly impact Canadian importers and vendors.
Cost-effectiveness and Feasibility
It may be useful for CFIA inspectors to occasionally check imported Dioscorea tubers to ensure that Dioscorea polystachya is not being imported under an incorrect name or synonym. This would require training inspectors and providing them with identification material. As described above under Previous imports, however, Dioscorea tubers are only rarely imported; the impact on CFIA resources should therefore be minimal.
Potential Mitigation Measures for Non-intentional Introduction Pathways
Note that, as described in Section Appendix 6A, in the section called Probability of Entry, unintentional introduction pathways (through contaminated soil, seed, grain, etc.) are considered only minor pathways, posing a minimal risk to Canada. These pathways will therefore not be detailed here.
Appendix 7A: Pest Risk Assessment Summary for Echium plantagineum (Paterson's curse)
CFIA Pest Risk Assessment
English Common Names: Paterson's curse, purple viper's bugloss, salvation Jane, blue weed, Lady Campbell weed, purple bugloss, purple echium, riverina bluebell, dwarf blue bedder.
French Common Names: Vipérine à feuilles de plantain, vipérine faux plantain
Common Names: Echium plantagineum L. (LAMIALES: Boraginaceae), known as Echium plantagineum, is an annual or biennial broadleaved weed. It has been widely introduced around the world as a garden plant and pasture species. More recently, interest in this species has increased due to a unique fatty acid composition that includes both gamma linolenic acid and stearidonic acid. The seed oil has potential for use in health food, cosmetic and pharmaceutical industries. Some of the major issues surrounding Echium plantagineum include its ability to dominate pastures in its exotic range, toxicity to livestock, and potential control issues due to herbicide resistance.
Description: Echium plantagineum L. (LAMIALES: Boraginaceae), known as Echium plantagineum, is an annual or biennial broad-leaved weed with purple flowers.
Distribution: Echium plantagineum has a broad, circum-Mediterranean native distribution. It is particularly successful in invading other countries with Mediterranean-type climates, and has become invasive in Australia, South Africa, Chile, Uruguay, Argentina, and Brazil. In Australia, it has invaded an estimated 33 million hectares. There have also been a number of introductions of Echium plantagineum to the United States, and it is currently invasive in California.
There are records of at least three prior occurrences of Echium plantagineum in Canada: Brandon, Manitoba; Vineland, Ontario; and near St. John's, Newfoundland. It does not appear to have persisted at any of these locations. More recently, Echium plantagineum has been grown for several years in field trials near Saskatoon, Saskatchewan.
Likelihood of introduction: Echium plantagineum has been imported into Canada and there is further interest in importing this species for commercial production.
Establishment potential: Echium plantagineum favours warm temperate climates. Information on the native geographic distribution of this species suggests that it could potentially establish in four plant hardiness zones in Canada (USDA Plant Hardiness Zones 5-8). Although less likely to thrive there, this species may be able to establish in harsher climatic zones as well. Field trials in North America indicate that Echium plantagineum can flower and set seed up to Zone 2 or 3.
Spread potential: Echium plantagineum has both a high reproductive potential and high dispersal potential. This species produces seed over much of the growing season, which can result in large seed banks of as many as 30,000 seeds/m2 under favourable conditions. Seeds germinate under a wide range of temperatures and can remain viable for 10 years in the soil. There are numerous pathways through which Echium plantagineum may disperse, including attachment to animal wool or fur, as ingested seed, and via contaminated vehicles, equipment, hay, grain, soil, or gravel. It has also been known as a cereal and forage seed contaminant. Evidence from its exotic range also indicates a high potential for spread of Echium plantagineum.
Potential economic impact: Echium plantagineum has already caused major economic impacts in other countries where introduced. In Australia, lost pasture productivity, control costs and wool contamination due to Echium plantagineum are estimated to cost sheep and cattle producers $250 million AUD annually. At least six potential negative economic impacts have been identified for Echium plantagineum: pasture degradation, livestock and crop yield losses, hay and seed contamination, and increased costs of control.
Potential environmental impact: Echium plantagineum has the potential to have serious impacts on the environment. The most significant of these are considered to be the potential negative impacts on animal and human health due to the plant's toxic alkaloids and the potential consequences of herbicide resistance in this species. In addition, it appears that Echium plantagineum has the potential to affect ecosystem processes (erosion processes, fertility) and community composition.
Recommendations: Specific phytosanitary measures are strongly recommended. Therefore, it is recommended that Echium plantagineum be added to an official list of species that are not allowed to be imported into Canada. Furthermore, it is recommended that Echium plantagineum be considered for inclusion on Canada's Regulated Pest List and on the Weed Seeds Order as Class 1 Prohibited Noxious.
CFIA Pest Risk Assessment Update
Additional information relating to the CFIA pest risk assessment was gathered in April 2008 as a result of questions arising from a 30-day consultation on the initial version of this RMD. The summary from this document (Castro 2008) is incorporated below.
- There are three previous known introductions of Echium plantagineum in Canada. Examination of the specimens and label data show that one of the introductions, in Murray's Pond, NL, persisted for 12 years and was difficult to eradicate. A second specimen, from Brandon, MB, was described as producing occasional volunteers. A third specimen, from Vineland, ON appears to have been a garden escape.
- Based on its native distribution, Echium plantagineum appears to have the potential to establish in USDA Plant Hardiness Zones 5-8 in Canada.Evidence from previous introductions of Echium plantagineum in Canada supports this possibility, even though climatic conditions and cropping patterns are different than other countries where this species has proven invasive, such as Australia, South Africa, and several South American countries. The production of volunteers in Brandon, MB (USDA Hardiness Zone 3a) suggests that Echium plantagineum may be able to establish in even colder zones than this.
- Lack of invasiveness in the native range of Echium plantagineum cannot be used to support the claim that Echium plantagineum would not have the potential to become invasive in Canada. Species in their native ranges are generally held in check by factors such as native herbivores and native community composition. Also, some sources describe Echium plantagineum as a weed even in its native range.
- At present, field trial information from North America is inconclusive as to whether or not Echium plantagineum poses an invasive threat in Canada. A paper describing field trials in North Dakota since 2002 claims low invasive risk for Echium plantagineum and absence of volunteers, but no indication of the methods used to evaluate these factors was provided. A field trial in Maine in 2007 was mostly destroyed due to concerns over invasive risk. A request for confidentiality has precluded the use of information from field trials in Saskatchewan.
- Mitigation measures to prevent the spread of Echium plantagineum into the Canadian environment have been described by an import proponent. However, dispersal of the species by wildlife is still a concern, as is human error and the greater risk of loss of containment in the future when increased acreage may be grown. Additional mitigation measures are proposed. These include: long-term post-harvest monitoring of fields (10 years or more), planting away from ditches and watercourses, prevention of movement of soil from areas where the crop is sown, inspection of tire treads, and the use of fences.
Other Pest Risk Assessments
A pest risk assessment for Echium plantagineum by the Oregon Department of Agriculture (ODA 2004) indicated that this species received the highest possible rating ("A") according to the ODA Noxious Weed Rating System. ODA also assessed Echium plantagineum using the USDA-Animal and Plant Health Inspection Service (APHIS) Weed Risk Assessment Guidelines. The cumulative risk score was 35 out of a possible 37 points, indicating potential for rapid dispersal and growth rates as well as negative economic impacts to certain agricultural industries
Appendix 7B: Risk Management Considerations for Echium plantagineum (Paterson's curse)
- Potential Hardiness of Echium plantagineum in Canada
- Areas of Commercial Cultivation
- Field Trial Information
- Economic Impacts in Countries other than Australia
- Agricultural Values at Risk
- Herbicide Resistance
- Socio-economic Impacts
- Potential Trade Impacts
- Existing Domestic, Provincial or other Programs
Potential Hardiness of Echium plantagineum in Canada
Echium plantagineum should be able to establish and thrive in USDA Plant Hardiness Zones 5-8 in Canada, putting at risk pasture, livestock, crops, hay and honey production in the following areas: Vancouver Island, the Queen Charlotte Islands, the west coast of BC, a large area in south-central British Columbia, the southernmost parts of Ontario, Nova Scotia, Prince Edward Island, a small area of southern New Brunswick, and eastern Newfoundland. Echium plantagineum may also be able to establish in colder regions up to USDA Plant Hardiness Zones 2 or 3, but is less likely to flourish in them. Figure 1 shows the USDA hardiness zones 3a and higher.
In Canada, Echium plantagineum has been historically collected from sites in southern Manitoba, southern Ontario, and Newfoundland (Scoggan, 1978). It has not persisted indefinitely at any of these sites. Lack of persistence would seem to provide support for low invasive risk of Echium plantagineum in Canada. However, examination of the label data on the specimens provides evidence to the contrary.
A specimen collected in 1931 from Murray's Pond, Newfoundland (NFLD) (near St. John's) is located at the Harvard University Herbarium (GH). It was submitted with a note stating "planted 12 years ago", "have at last got it nearly all dug out" and "garden weed (pest)". These statements indicate that Echium plantagineum can persist as a weed from year to year in that area of Newfoundland, which is located in approximately USDA Plant Hardiness zone 6b. CLIMEX software was used to determine other locations in Canada with similar climates to St. John's, NFLD (Figure 2). Areas with similar climates include parts of New Brunswick and Nova Scotia, and to a lesser extent, southern Ontario, and coastal British Columbia.

Description for Figure 1:
This image shows locations in North America with a similar climate as St. John's, Newfoundland. This is done through the implementation of a Composite Match Index with four blue circles, each circle getting bigger than the last, symbolizing the ranges of 0.7, 0.8, 0.9, and 1 respectively.
The specimen collected in 1953 from Brandon, Manitoba (MB) is located at the Agriculture and Agri-food Canada Herbarium (DAO). The label on this specimen states "Roadside. Dry soil. Occasional volunteer plants [plants]." Brandon has a much harsher climate than St. John's, NFLD, but evidently Echium plantagineum is capable of producing volunteers there as well. Brandon is in USDA Hardiness zone 3a and has a climate similar to much of the Prairie provinces.
There are two specimens of Echium plantagineum in the University of Guelph herbarium (OAC). One was collected in 1940 from Vineland, ON, where it was collected in habitat described as "Waste ground, near garden." The second is a white form of the species, cultivated at the Ontario Agricultural College in Guelph, (Ontario) ON and collected in 1935. The former suggests that the Echium plantagineum is capable of becoming a garden escape. Vineland, ON is in approximately USDA Hardiness zone 5b.

Description for Figure 2:
This map contains scattered blue dots across the Maritime Provinces as well as throughout southern Ontario and into the United States deceasing in size as it proceeds downwards. Moreover, occasional spots can be found across the western coast.
Composite Match Index is the product of six component indices (total rainfall, rainfall pattern, humidity, soil moisture, and maximum and minimum temperature) (Sutherst et al., 2006). The value ranges from 0 to 1, with 1 representing a perfect climate match. Map generated using Climex software.
With the exception of the first specimen from Murray's Pond, NFLD, it is not known why Echium plantagineum did not persist in Brandon, MB or Vineland, ON. Lack of persistence may have been due to eradication efforts (physical, mechanical or chemical), as was the case in NFLD, rather than climatic factors. The label data suggests that the species can re-seed and potentially spread at both locations. A seed bank decay rate study by Sheppard and Smyth (2002) showed that Echium plantagineum seed longevity was closely linked with field conditions, but may last tens of years.
Several stakeholders indicated that Echium plantagineum is unlikely to become invasive in New Brunswick because New Brunswick differs significantly from this species' circum-Mediterranean distribution. As shown in Figure 2, there are many locations throughout the Atlantic Maritimes that have similar climates to St. John's, NFLD where this species was a known pest. It is also important to note that the scope of this risk management document for Echium plantagineum includes all of Canada, not just New Brunswick where trials have been proposed. Therefore, the invasive risk of Echium plantagineum must be considered for the entire country.
The CFIA weed risk assessment on Echium plantagineum determined that there is potential for this species to establish in USDA Plant Hardiness Zones 5-8 in Canada based on its native geographic distribution, which includes northern Africa, parts of eastern Asia and much of Europe, including central and southern Russia (Gibbs, 1972). Its center of origin is Mediterranean, but its native range clearly extends beyond this region. There are also records of Echium plantagineum in Norway and Sweden (GBIF, 2008). Evidence of volunteering in Manitoba suggests that this species may be capable of establishing in even colder hardiness zones. It should also be noted that Echium plantagineum has shown high levels of genetic diversity and phenotypic plasticity (Wood and Degabriele, 1985). Seed collected from harsher climatic regions of its native or introduced range may have a better chance of success in Canadian climates than those collected in warmer regions.
It should be noted that even if Echium plantagineum does not survive as an autumn germinated plant, and complete its lifecycle as a winter annual, there is a risk that the species may overwinter as seed, germinate in spring, and complete its lifecycle as an annual plant. Experience with this plant indicates that it reproduces quickly, and can begin flowering within two weeks of emergence in spring (B. Myers-Shenai, 21 Apr. 2008, pers. comm.).
In summary, the potential for adaptation of Echium plantagineum to the Canadian environment appears to be high due to several factors. The species has wide climatic amplitude and an extensive native range. It is capable of establishing new populations where introduced and, notably, it is capable of invading disturbed sites. Seed production is high. It behaves as an annual, winter annual or biennial, conferring greater adaptation potential than perennial species. Genetic variation is also high, and it is capable of a great deal of phenotypic plasticity in response to localized environmental factors. Collectively, these factors suggest a highly adaptable weed potentially capable of establishing throughout most of Canada's agriculturally productive lands.
Areas of Commercial Cultivation
Several stakeholders have indicated that Echium plantagineum is cultivated in the United Kingdom (UK) and New Zealand with few or no issues of invasiveness. IENICA (2000, 2002) report that Echium plantagineum has only been grown in pilot studies in the UK and that details about European production are unknown. Production in the UK was limited to 20 ha in 1997 and 190 ha in 1998. It appears that production in the UK in the past ten years has been small-scale. Experience with the plant in the UK indicates that it grows rapidly and is able to out compete weeds in early stages, and it tolerates infertile soils.
The United Kingdom is considered part of the native range of Echium plantagineum (Clapham et al., 1962; CABI , 2006). Species in their native ranges are generally kept in check by factors such as native community composition and natural predators. In the absence of these factors, some species become invasive where introduced. For this reason, lack of invasiveness in the United Kingdom is not compelling evidence to support the suggestion that Echium plantagineum would not have the potential to become invasive in Canada. Many of Canada's invasive plant species have been introduced from Europe and are not invasive in their native range.
However, it should also be noted that Echium plantagineum has been described as weedy even in its native range. It is considered a 'principal' weed in Tunisia (Holm et al., 1979). Echium plantagineum is also considered an arable weed of Europe (Hanf, 1983). It is introduced occasionally into central Europe from its main native range as a seed contaminant (Hanf, 1983). Clapham et al. (1962) indicate that Echium plantagineum can also occur as a garden-escape in some parts of the British Isles.
Echium plantagineum has also been cultivated in New Zealand but has not caused problems to the same degree as in Australia. However, it is still considered a weed in New Zealand and is described as a "Common weed of road-side and waste places in the north of NI" (Roy et al., 1998). Its habitat is old habitations, road-sides and waste places. Roy et al. (1998) also indicates that grazed pastures in New Zealand are "probably too competitive for it to invade." It is also sold in garden centers in New Zealand.
Field Trial Information
There is little information on the behaviour of Echium plantagineum in cultivation in North America to date. Trials have been conducted in North Dakota, Maine and Saskatchewan. Echium plantagineum has been grown in trials in at least four locations (Carrington, Langdon, Minot, and Prosper) in North Dakota since 2002 (Berti et al., 2007). Seed sources for the trials were from the United Kingdom and wild populations in Chile, collected between 35°S and 40°S. Trials were sown in late May and flowered 31 to 49 days later, with the exception of the Chilean lines, which flowered between 52 and 80 days after sowing or not at all (2 lines). The results state the following: "The harvest index for Echium plantagineum is extremely low indicating little domestication and plant breeding improvement. High lodging of the crop after full bloom contributes to seed shattering, white mold [Sclerotinia sclerotiorum (Lib.) De Bary] infection, and poor seed appearance." In addition, the following statements are made: "Echium invasiveness risk is low in North Dakota. No volunteer plants were observed from shattered seed the following season."
There is no indication in this article about the methodology used to determine low invasive risk and the absence of volunteers. The climate in North Dakota is similar to that of southern Manitoba where Echium plantagineum has indeed produced occasional volunteers. Attempts have been made to contact the author of this paper to inquire further about the trials and the method for evaluating invasive risk but no response has been given to date.
A trial on Echium plantagineum was also conducted on 8 acres in Aroostook County, Maine in 2007. The recommendation was given to destroy the trial prior to seed set (Sexton, 2007). The recommendation was based on several factors, including concern regarding infestations in Oregon and California, the species' prolonged seed longevity and prolific seed production, and the potential to harm grain and hay markets (Sexton, 2007). The final decision was to destroy most of the trial, but allow 3 acres to set seed and be harvested. It will not be replanted in the future, and measures will be taken to ensure that this species does not establish and spread from this site (A. Gibbs, ME Dept of Agriculture, 28 Apr. 2008, pers. comm.). Due to the short-term nature of this trial, there is insufficient evidence to indicate that Echium plantagineum has low invasive risk in Maine.
Echium plantagineum trials have also been grown near Saskatoon, Saskatachewan for several years. Information on these trials cannot be included because of a request for confidentiality from the owners of the data.
Economic Impacts in Countries other than Australia
In addition to Australia, where Echium plantagineum has caused significant economic impacts, this specieshas become invasive in South Africa, Chile, Uruguay, Argentina, Brazil, and the US (Oregon and California). There are few reports of economic impacts from this species in these other countries. However, reports that quantify economic impacts of invasive plant species are few in general. Echium plantagineum has been reported to cause harm to livestock in Brazil (Mendez et al., 1985; Santos et al., 2008). In the US, Echium plantagineum has also shown the capability to dominate in cattle pastures. In South Africa, Echium plantagineum is described as a roadside weed (Retief and Van Wyk, 1998).
The Australian experience did not occur under managed conditions, however it has been demonstrated that Echium plantagineum can become invasive as a result of a very limited introduction sometimes consisting of a few plants. Although best management practices can mitigate the risk, they cannot reduce it to an acceptable level. The potential for this species to escape into the wild or into other agricultural crops and pastureland will increase substantially if this species is allowed to be grown commercially throughout Canada.
Agricultural Values at Risk
Echium plantagineum has the potential to cause negative economic impacts including pasture degradation, livestock and crop yield losses, hay and seed contamination, and increased costs of control. Although in the past Echium plantagineum was considered a valuable honey plant, concerns have been raised about the levels of pyrrolizidine alkaloids that carry over into the honey. Due to the presence of toxic alkaloids in Echium plantagineum, Echium plantagineum honey must be blended with non-Echium plantagineum honeys. The Canadian Honey Council has issued a warning regarding the use of honey from Echium plantagineum based on information from Food Standards Australia New Zealand (Canadian Honey Council 2005). Canadian honey producers in proximity to Echium plantagineum crops may therefore be negatively affected. The Canadian Honey Council has expressed concern over the potential requirement for blending honey should Echium plantagineum be present in the Canadian environment, as blending would add significant costs to honey production (H. Clay, Canadian Honey Council, 22 Apr. 2008, pers. comm.).
Although a comprehensive cost-benefit analysis is not within the scope of this document, a rough assessment of the agricultural values that are at risk within the potential range of Echium plantagineum in Canada is shown in tables 1-3. Table 1 shows the number of beekeepers and value of honey production. Provinces within USDA Plant Hardiness Zones 5-8, its primary potential range, are indicated with an asterisk. In 2004 the value of Canadian honey production was $121 million. Recent correspondence with the Canadian Honey Council indicates that the beekeeping industry of Canada includes 8,000 apiculturists, 600,000 colonies of honey bees and has a current value of two million dollars in honey production and one billion dollars in the pollination of agricultural crops. Canadian pure honey is renowned world-wide for its quality and flavour. Approximately seventy-eight percent of Canadian honey production is concentrated in Alberta, Saskatchewan and Manitoba.
| Province | No. of beekeepers | Value $,000 |
|---|---|---|
| Nova ScotiaTable Note 1 | 375 | 1,100 |
| New BrunswickTable Note 1 | 225 | 355 |
| Prince Edward IslandTable Note 1 | 30 | 150 |
| Quebec | 205 | 5,600 |
| OntarioTable Note 1 | 2,650 | 14,005 |
| Manitoba | 580 | 16,905 |
| Saskatchewan | 1,055 | 21,000 |
| Alberta | 695 | 50,455 |
| British ColumbiaTable Note 1 | 2,110 | 11,535 |
| Total: Canada | 7,925 | 121,105 |
Source: Honey Council Canada.
Table Notes
- Table Note 1
-
Within primary potential range.
| Province | Hectares of Tame (Seeded) Pasture | Hectares of Natural Pasture |
|---|---|---|
| NewfoundlandTable Note 2 | 2,348 | 10,288 |
| Nova ScotiaTable Note 2 | 23,381 | 31,708 |
| New BrunswickTable Note 2 | 17,013 | 24,995 |
| Prince Edward IslandTable Note 2 | 10,847 | 12,315 |
| Quebec | 147,387 | 158,602 |
| OntarioTable Note 2 | 303,400 | 959,935 |
| Manitoba | 498,295 | 1,548,197 |
| Saskatchewan | 1,962,222 | 5,175,789 |
| Alberta | 2,483,702 | 6,529,863 |
| British ColumbiaTable Note 2 | 245,793 | 1,499,563 |
| Total: Canada | 5,694,388 | 15,951,255 |
Source: Statistics Canada.
Table Notes
- Table Note 2
-
Within primary potential range.
Tables 1 and 2 indicate the areas in forage production which is the foundation of Canada's beef and dairy industries. The beef and dairy industries have traditionally been the second and third ranking primary agriculture sectors in Canada after the grain sector.
According to the 2001 Census of Agriculture, throughout Canada, over 27 million hectares were devoted for livestock grazing and forage production (Agriculture and Agri-food Canada 2007). Of this, about 15.4 million hectares were native pasture, 4.8 million hectares were tame or seeded pasture, and nearly 7 million hectares were cultivated tame hay, including 5 million hectares located in Western Canada. In total, it is estimated that approximately 40% of Canada's total farm area is allocated for grazing and growing forage crops. Harvested area for tame hay was 6.7 million hectares in 2005.
| Province | Tonnes ('000) |
|---|---|
| NewfoundlandTable Note 3 | 18 |
| Nova ScotiaTable Note 3 | 339 |
| New BrunswickTable Note 3 | 354 |
| Prince Edward IslandTable Note 3 | 258 |
| Quebec | 3,435 |
| OntarioTable Note 3 | 4,563 |
| Manitoba | 3,193 |
| Saskatchewan | 4,477 |
| Alberta | 8,437 |
| British ColumbiaTable Note 3 | 1,556 |
| Total: Canada | 26,630 |
Source: Agriculture and Agri-food Canada 2007.
Table Notes
- Table Note 3
-
Within primary potential range.
Herbicide Resistance
In Australia, Echium plantagineum has developed resistance to a group of herbicides known as acetolactate synthase (ALS) inhibitors. This group of herbicides includes the sulfonylurea family of herbicides, to which this population of Echium plantagineum is highly resistant, and the imidazolinone family of herbicides, to which it exhibits low-level resistance (Preston 2006). Herbicide resistance in Echium plantagineum can lead to increased costs of control and reduced choices for herbicide use over the long term. In Australia, it is considered likely that ALS resistance will persist over the affected areas for many years even if ALS inhibitors are not used, due to the long lived seed bank (Preston 2006).
It should be noted, however that many cases of ALS herbicide resistance over several species are well documented as established in Canada. These are most prevalent in central and western Canada which historically uses multiple products in this herbicide group. Increased cost of control would not result if more cost-effective herbicide groups, such as phenoxy herbicides were used for Echium plantagineum. With any responsible management program, rotation of herbicide groups are highly suggested regardless of the crop. Canadian growers recognize this and practice this in their farming operations. Australia has experienced herbicide resistance to most groups of herbicides including the first case of glyphosate resistance in the world due in part to poor management practices.
Socio-economic Impacts
Negative Impacts:
Although impacts would take many years to accumulate, costs to Canadians relating to Echium plantagineum after several decades could eventually compare to those currently experienced in Australia. The plant has spread over millions of acres in New South Wales, Western Australia and Victoria provinces. In Australia, lost pasture productivity, control costs and wool contamination due to Echium plantagineum are estimated to cost sheep and cattle producers $250 million AUD annually which is the current equivalent of $220 million CAN annually.
It has been suggested that farmers will greatly benefit from increased revenue associated from growing Echium plantagineum as opposed to cereal crops, especially in New Brunswick. Since New Brunswick is a net importer of feed grains, the reduction of New Brunswick-based grain supplies because of potato farmers switching from cereal rotation to rotation with Echium plantagineum may actually have significant impacts on the New Brunswick livestock industry. The rising costs of feed grains are seriously compromising the Canadian livestock sector.
Canadian honey is world renowned as being exceptional in quality; however the introduction and spread of Echium plantagineum in Canada could potentially affect this reputation and impose a requirement for blending at additional cost to the apiculturist. The total value of Canadian honey in 2004 (the most recent year available from Statistics Canada) was $121.1 million. Recent correspondence with the Canadian Honey Council indicates that the beekeeping industry of Canada includes 8,000 apiculturists, 600,000 colonies of honey bees and has a current value of two million dollars in honey production and one billion dollars in the pollination of agricultural crops.
Potential environmental and social impacts include toxicity to animals and humans, increased or changed herbicide usage in association with herbicide resistance, decreased soil fertility and increased soil erosion, and changes in community composition in natural ecosystems. Pyrrolizidine alkaloids and cytochrome C allergens are two known potentially toxic constituents that are found in Echium plantagineum. Pyrrolizidine alkaloids are harmful to human health, when consumed in products such as milk or milk products, honey or cereals. Liver poisoning by pyrrolizidine alkaloids in humans is gradual and cumulative, and there is no known therapy (Klemow et al., 2002). Furthermore, pyrrolizidine alkaloids "inhibit mitosis, cause chromosomal abnormalities, and are mutagenic, teratogenic and carcinogenic (Culvenor 1985). Ingestion of pyrrolizidine alkaloids may result in cancerous lesions on the lung and liver (Huxtable 1980)" (Klemow et al., 2002). Pyrrolizidine alkaloids in herbal teas have caused poisoning of human infants (Huxtable, 1980, cited in Klemow et al., 2002). Cases of poisoning have been noted in rats (in studies), horses, pigs, cattle, and sheep. Monogastric animals such as horses and pigs are much more susceptible than ruminants, and can be lethally poisoned within a single season of grazing (Peterson 1985). Deaths in sheep may occur over the longer term, and are usually associated with chronic copper poisoning (Peterson 1985). Another invasive plant species in Canada also contains alkaloids (Senecio jacobaea, Tansy Ragwort), however this is not a sufficient justification to overlook the significance of introducing yet one more into the Canadian environment.
Pollen from plants in the genus Echium plantagineum has also been associated with respiratory allergies (cytochrome C allergens) (UK Advisory Committee on Novel Foods and Processes 2007). The hairiness of Echium plantagineum has caused dermatitis, inflammation and itching to both humans and animals (Castro 2007).
Positive Impacts:
Crop diversification is widely promoted by federal and provincial agricultural agencies throughout Canada, and the addition of Echium plantagineum as a potentially profitable market-niche crop in Canada would further that goal. The development of Echium plantagineum as a potato rotational crop in New Brunswick has, in particular, received a great deal of attention. While the cereal grains (barley, oats, wheat) have traditionally played a large role as rotation crops for potatoes in New Brunswick, the contribution of those crops to farm economics is marginal. Grain is priced largely in global markets, and New Brunswick/Atlantic Canada, with relatively cool summers and a small land base, does not support a vibrant grains industry. New Brunswick and Atlantic Canada are net importers of feed grain for livestock.
There is a need for new players to provide market access to enable the New Brunswick agri-food and agri-products sector to participate in the new bioeconomy with its emerging opportunities in natural health products, functional foods and industrial bioproducts. These markets are not easily negotiated by agricultural producers, or even cooperatives, which do not have market clout and easy access to global markets outside of commodity regimes. For this reason, the Strategic Innovation Group (SIG) of the New Brunswick Potato Cluster was formed in 2004. The SIG is chaired by the Minister of the New Brunswick Department of Agriculture and Aquaculture and consists of stakeholders from industry, government, university, R & D, consumer groups and additional external advisors.
While large by New Brunswick's standards, the 22,000 or so hectares that the New Brunswick potato industry has to offer for rotation, falls in to a category better described as "niche" on the scale of global crops. The relatively exacting requirements for potato production place the growers in a favourable situation for understanding the more exacting requirements of niche cropping and strict accountability in crop management. Cooler climates, such as found in Atlantic Canada, typically result in the production of seed oils that are more enriched in the polyunsaturated fatty acids of interest. New Brunswick's summers have warm (not hot) days and cool nights, a characteristic deemed vital for the accumulation of the desirable oil constituents in Echium plantagineum.
In addition to the role that that Echium plantagineum promises to offer in diversifying New Brunswick's potato industry, there are several other benefits including:
- Subject to the integration of E. plantagineum in the New Brunswick cropping mix, the investment for processing capacity is likely to be in the order of $15 M and creating 20-25 jobs. These investments bode well for workforce stabilization and agricultural sustainability in New Brunswick's potato belt.
- Being higher margin crops, the rotation crops being targeted can make a real contribution to the ecology and economy of the potato industry, by lowering the temptation to truncate the rotation cycle.
- The pollination window required for the crops marries well with the window of requirement for wild blueberries and cranberries, and these boraginaceous crops should enhance the health of hives, since they produce more nectar and are better suited to pollination by honeybees than the Vaccinium species.
- Diversifying and strengthening the beekeeping industry has positive downstream impact on the success and sustainability of NB's wild blueberry industry.
- One of the end uses of Echium plantagineum is pharmaceutical, and targets an important anti-inflammatory condition. Cultivation would address some of the health challenges that Canada and the world face; and
- Providing plant-based alternatives to synthetic industrial inputs.
It has been suggested that farmers will greatly benefit from increased revenue associated from growing Echium plantagineum as opposed to cereal crops, especially in New Brunswick. It is important to note, however that since cereal grain prices are rising in North America, the difference in revenue may not be significant.
Currently there are no concrete proposals to extract the seed oils in Canada. The construction of a processing plant would cost approximately $25 million and the plant would potentially employ up to 25 people. If a refinery was not constructed in Canada, however, the product would probably be exported and Canadian economic benefits would be restricted to crop production.
The cultivation of Echium plantagineum would provide a reliable source of essential fatty acids if a refinery was developed in Canada. The seed oil of Echium plantagineum contains a unique oil profile that is rich in essential fatty acids. Essential fatty acids are not produced by the body, but must be obtained through the diet or by supplementation. Of particular interest are the gamma linolenic (GLA) and stearidonic fatty acids present in the seed oil. Echium plantagineum seed oil contains 10-11% GLA; other sources include borage oil (22-25%), black current oil (about 15%), evening primrose oil (8-10%), and biotechnology-derived safflower oils (35-65%) and canola oils (36-40%) (Patterson 2006). GLA is considered to have many beneficial effects, and has been found effective as a treatment for eczema, hyperactivity disorders, certain cancers, high blood pressure, and swelling.
Stearidonic (SDA) acid is uncommon in plants. Seed oil produced from Echium plantagineum, however, can contain 9 to 16% stearidonic acid according to Berti et al. (2007). Guil-Guerrero et al. (2007) reports 30% saponifiable oil with 13% SDA for Echium plantagineum. Other sources of stearidonic acid include hemp seed (2-3%) (Callaway et al. 1996) and black current seed (2%) (Clough 1993), as well as other Echium species. Guil-Guerrero et al. (2003) report 28.7% saponifiable oil with 13.3% SDA for Echium vulgare, a very similar plant which is also weedy and non-native, but is already present in every province in Canada. Guil-Guerrero et al. (2007) list numerous other plants with similar or even higher levels of SDA in the Boraginaceae and Primulaceae families. Stearidonic acid is also valuable as a health supplement and for its moisturizing and anti-inflammatory action. Echium plantagineum has food use approval in the USA and is awaiting full approval in the EU.
The recent approval of refined echium oil by the FDA in the US has created an increased market opportunity. The approval of echium oil in the UK is still pending.
Potential Trade Impacts
Echium plantagineum is considered a noxious weed in all states and territories of Australia, with the exception of Queensland (Parsons and Cuthbertson 1992). It is regulated as a Category 1 weed in South Africa. In the US, it is considered a quarantine weed for the state of Oregon (USDA, NRCS 2007). If Echium plantagineum was allowed to be cultivated as a crop in Canada and became established, exports of Canada's major crops and other agricultural commodities would potentially be subject to quarantine measures by these or other countries.
Prohibition of Echium plantagineum from Canada would have minimal trade implications. A Phytosanitary Certificate is currently required for all grain imports from off-continent and for all imports of barley, wheat, triticale and rye from the continental US therefore the proposed measure will not require any additional documentation for these crops. Persons responsible for issuing a Phytosanitary Certificate for imports of grain into Canada would need to be confident that the shipment was free from Echium plantagineum as is the case now for three other invasive plant genera: Cuscuta, Orobanche and Striga. A table of major cereal grain imports into Canada for the past few years from countries and states known to support populations of Echium plantagineum has been included (Table 4). The countries which are known to support populations of Echium plantagineum are listed in the pest risk assessment (Castro 2007).
| Barley | Oats | Rye | Wheat | Sorghum | Corn | |
|---|---|---|---|---|---|---|
| US-North Dakota | 8,454,224 | 294,006 | 31,627 | 528,614 | 20,762 | 64,776,228 |
| US-Maine | 1,435,753 | 1,772,408 | 26,934 | / | / | 1024 |
| US-Oregon | 5 | 27,901 | 82 | 422,496 | 289 | 236 |
| US-California | 5,043 | / | / | 8,701 | 251 | 93,583 |
| United Kingdom | 143 | 24 | / | 27,372 | / | 12 |
| Chile | / | / | / | 15,583 | / | 13,179,209 |
| New Zealand | 45 | 411 | / | 7,598 | / | 84 |
| Russia | 316 | 15,282 | / | 4,890 | / | 2,108 |
| Italy | 356 | / | / | 285 | / | / |
| Türkiye | / | / | / | 196 | / | 44,558 |
| Argentina | / | / | / | / | / | 442,435 |
| France | / | / | / | 45 | / | 660,738 |
| Australia | 1 | / | / | 1 | / | 1 |
| Ukraine | / | / | / | / | / | / |
| Spain | / | / | / | / | / | 22,156 |
| Brazil | / | / | / | / | / | 33,725 |
| Portugal | / | / | / | / | / | / |
Source: Trade Data Online (2008)
Note: Items highlighted in bold indicate higher level of risk based on quantity and cereal type. Figures are for 2007 only and may not be indicative of future trends.
The distribution of Echium plantagineum in the US is limited. It is known from two counties in Oregon and is under official control. Field trials were conducted in North Dakota, however they were limited. A 3 acre field trial occurred last year in Maine, but has been discontinued. A Phytosanitary Certificate is currently not required for soybean, corn and oats from Maine, however the risk of contamination of Echium plantagineum in corn and soybean is considered relatively low due to the large seed size of the commodity and harvesting height. California has the most significant infestation of Echium plantagineum in North America. However, in terms of cereal crops, Canada imports mainly corn from California.
Echium plantagineum is also grown and marketed in some parts of the world as a garden species because of its attractive flowers; however, the plant is rarely if ever sold in Canada. It has been identified as a component of three imported flower mixtures since 2007 (CFIA 2010, internal data). The average value of flower seeds, including seed mixtures, imported to Canada in the past five years is about $17,000 every year (Statistics Canada, 2010). Echium plantagineum can be avoided or replaced in flower mixtures by similar flower species without trade impacts.
Existing Domestic, Provincial or other Programs
Echium plantagineum has only recently been considered for use as a new, alternative crop in Canada. The species is not known to occur in Canada outside of cultivation. As a result, there are no existing Canadian programs related to its management.
Appendix 8A: Pest Risk Assessment Summary for Microstegium vimineum (Japanese stiltgrass)
- Identity of Organism
- Organism Status
- Current Regulatory Status
- Probability of Entry
- Probability of Establishment
- Probability of Spread
- Potential Economic Consequences
- Potential Environmental and Social Consequences
- Uncertainty
- Conclusion
- Technical Issues for Consideration
Identity of Organism
Name: Microstegium vimineum
Synonyms: Andropogon vimineum, Eulalia viminea, Eulalia viminea, (USDA-NRCS 2008). Microstegium imberbe, Microstegium willdenovianum, Pollinia imberbis, Pollinia viminea, Pollinia willdenoviana (Tu 2000).
English common names: Japanese stiltgrass, Japanese grass, Nepalese browntop, Nepalgrass (USDA-NRCS 2008).
French common names: None found
Description: Microstegium vimineum is an annual grass with a sprawling habit. It germinates in spring and grows slowly through the summer months, ultimately reaching heights of 0.6-1 m with the reclining stems growing up to 1 metre long. Slender stalks of tiny flowers are produced in August through to September or early October. In late fall they fade to pale greenish-yellow or turns purple in colour (Swearing 2004, Mehrhoff et al. 2003).
Microstegium vimineum grass has become a significant problem in forests in many eastern and Midwestern states. It spreads rapidly due to high seed production and rooting at nodes along the stem and is able to out-compete native vegetation. It often establishes in locations where moist soils are scoured such as along streambanks, floodplains, ditches and trails. Typical habitats include river corridors, forested wetlands, moist woodlands, old fields and thickets, utility rights-of-way, roadsides and lawns (Tu 2000).
Organism Status
Microstegium vimineum is not reported to occur in Canada (CFIA 2008), and no evidence was found that it is cultivated as an ornamental in Canada (CNLA 2009). Based on this information, Microstegium vimineum is considered absent from the PRA area.
Current Regulatory Status
Microtegium vimineum is not currently regulated in Canada. It is not regulated federally in the United States but is regulated in the following states: Alabama, Connecticut, and Massachusetts (USDA-NRCS 2008). It is not known to be regulated in other countries.
Probability of Entry
Microstegium vimineumis native to India, Nepal, China, and Japan and was introduced to North America in 1919 in Tennessee. By 1960 had it had spread (probably in hay and soil) to Ohio and Pennsylvania, and all Atlantic coastal states from Florida to New Jersey (see figure 1) (Howard 2005).
The most likely pathway for entry however is attached to the fur of animals or on human clothes or in mud on tires or hiking boots. Mehrhoff (2000) postulates that some populations along popular hiking trails in the US were likely introduced by way of fruits that adhered to people's clothing, shoes, hiking equipment or dogs.
| Type of pathway | Specific Pathways |
|---|---|
| Natural dispersal | Microstegium vimineum has high seed production which is its natural means of dispersal. It is also able to root at the nodes, which helps it to spread locally. Seeds are able to be dispersed in surface runoff and floodwaters, rivers and ditches are primary corridors for population expansion (Howard 2005) Natural spread is an unlikely pathway into Canada due to the distances between established populations and the Canadian border. |
| Intentional Introduction | Microstegium vimineum has not been documented as being intentionally planted as an ornamental, for erosion control or for forage (Howard 2005; EPPO 2008). No intentional pathways were identified. |
| Unintentional Introduction | According to an EPPO alert, Japanese stiltgrass can be introduced involuntarily as a contaminant of bird feed, soil, nursery stock and hay. No North American references were found which list birdseed as a pathway for spread. CFIA Seed Lab data show no reports of the species in marketplace monitoring samples of imported seed since 2000. It was widely used as a packing material for porcelain from China which was likely its pathway of introduction into the U.S. (Tu 2000). It is not clear whether this pathway still exists It also attaches readily to the fur of animals, human clothes and boots, and in mud on vehicle tires. It can be spread by hikers (Mehrhoff2000). |
Probability of Establishment
Microstegium vimineum is hardy to NAPPFAST zone 6 and also appears to be establishing populations in zone 5 in the United States. This gives it a potential range in Canada of parts of Atlantic Canada, southern Ontario and coastal and southern BC (see figure 2). In the New England States, Microstegium vimineum has invaded floodplain forests, early and late successional forests, abandoned fields, roadsides and other habitats (Mehrhoff et al. 2003. Microstegium vimineum also does well in many disturbed areas. These habitats are abundant within the endangered areas within the PRA area.
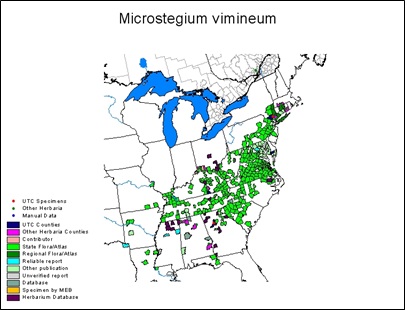
Description of Figure 1:
This image is contains a map of eastern North America distinguishing the range of Microstegium vimineum (Japanese stiltgrass) through the use of colours designated to a specific method. Red dots signify a UTC specimen; green dots equate to other herbaria; blue dots represents manual data; dark blue denotes UTC counties; magenta indicates other herbaria counties; light pink represents contributors; light green, the most predominant colour on the map, signifies state flora/atlas; dark green represents regional fora/atlas; light blue stands for reliable reports; turquoise indicates other publication on Microstegium vimineum while grey shows unverified reports; dark green signifies a database; and mustard yellow denotes specimen by MEB while dark purple represents herbarium database. While small light green pieces are predominant, they are heavily condensed in the following states and their surrounding areas: Virginia, New Jersey, Washington DC; parts of Pennsylvania, South Carolina, Kentucky, Tennessee and West Virginia. Moreover, they also have the presences of other colours, such as magenta, light blue, dark green and dark purple, though none of them pervasive.
Source: Barkworth 2006
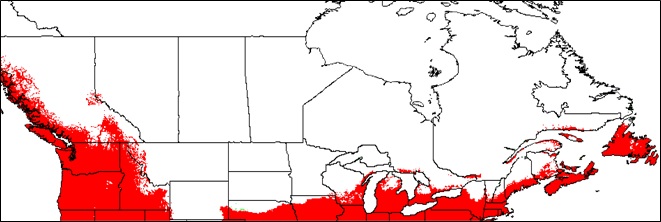
Description of Figure 2:
This image shows the potential range of Microstegium vimineum in Canada and northern America through the use of a map. Red is used to indicate the regions in which Microstegium vimineum could survive according to the Canadian Plant Hardiness Zones map, in this case NAPPFAST Hardiness Zones 5 and 6. In Canada, the potential range of Microstegium vimineum includes parts of Atlantic Canada, southern Ontario and coastal and southern BC. Most of the red can be found in the U.S., this includes the greater parts of the east and west coasts extending inwards reaching most of the central states as well as areas located near the Great Lakes extending downwards beyond the image
Source: Barkworth 2006
Probability of Spread
Although often associated with forested and wetland areas, Microstegium vimineum spreads rapidly into disturbed areas (Weber 2003). Its seeds can be spread by hikers (Mehrhoff et al. 2003), and are dispersed by surface runoff, floodwaters, on human and animal feet, and are carried in contaminated hay, soil or potted plants (Swearingen 2004). Ditches and rivers are primary corridors for population expansion (Howard 2005). It is unpalatable to grazing animals but they have been reported to spread the seeds (Swearingen 2004). By avoiding it and grazing other, more palatable, species wildlife may be indirectly contributing to its spread (Howard 2005). In suitable habitats it quickly spreads by rooting from its prostrate culms, forming dense, monospecific stands (Barkworth 2006).
Potential Economic Consequences
Microstegium vimineum has not been documented as being intentionally planted as an ornamental, for erosion control, or for forage (Tu 2000). It is mainly reported as a weed of natural areas, wetlands and forest understories. There have, however, been reports of it encroaching into crop production areas, landscape plantings, and turfgrass (Judge et al. 2008).
In 2008, a value of $24 million of field crops possibly intended for birdfeed in Canada was imported from areas where Microstegium vimineum is present as well as a value of $746,000 of hay and straw.
Potential Environmental and Social Consequences
Microstegium vimineum can form dense monotypic stands that replace natural communities and can dominate entire habitats, including forest understories and wetland areas. It can tolerate low light conditions allowing it to threaten forest vegetation as well as edge habitats. Once established, it is able to crowd out native herbaceous vegetation in wetlands and forests within three to five years (EPPO 2008).
Populations of Microstegium vimineum disrupt the quality of nesting habitats for birds (e.g. quails). However, it creates an excellent habitat for rats, such as cotton rats, a predator of birds. Microstegium vimineum also may be responsible for altering natural soil conditions, creating an inhospitable environment for many native species. Kourtev et al. (1998) reported that in areas that have been invaded by Microstegium vimineum, both litter and organic soil horizons were thinner than in uninvaded areas, and the pH of such soils was significantly higher (Tu 2000).
Following canopy disturbance Microstegium vimineum responds with a sudden increase in biomass allowing it to rapidly invade forests and impede regeneration of native woody species. Rapid increases in growth of Microstegium vimineum lead to the development of "mats'' on the forest floor that negatively affect native woody species regeneration in multiple ways: directly, through competition for sunlight, nutrients, and water and indirectly, by reducing the likelihood of successful seed to soil contact for germination (Oswalt et al. 2007).
Uncertainty
The climatic tolerance of Microstegium vimineum is uncertain. It is becoming increasingly widespread in the Eastern U.S. in NAPPFAST zone 6, however, there also appear to be populations in zone 5. Its native range is mainly zone 6 but there are some areas of zone 5 included in its distribution. For the purposes of this risk assessment it was assumed that it would be able to establish in at least parts of zone 5.
Conclusion
Based on the outcome of this pest risk assessment, Microstegium vimineum is likely to establish and become invasive in parts of Canada including southern and coastal BC, southern Ontario and parts of Atlantic Canada if it is introduced to these areas. This plant should be considered for regulation under the Plant Protection Act. It should be noted that one of the potential pathways into Canada is as a "hitch-hiker" on human clothing, muddy vehicles or pet fur, which may be difficult to regulate.
Technical Issues for Consideration
- The best strategy for control of Microstegium vimineum is repeated annual efforts to prevent flowering and seed set. Chemical control approved for use in Canada can be effective and may be necessary for larger infestations, although it may have negative impacts on desirable vegetation (Tu 2000).
- Microstegium vimineum is easily confused with Leersia virginica, a native grass species. Confusion arises in the field as these two species often are found growing together and they have similar leaf sizes and shapes and are similar in overall appearances (Mehrhoff 2000). However, the seeds are readily distinguishable as the plants belong to different tribes with very different floral morphologies (L. virginica is in the Oryzeae while M. vimineum is in the Andropogoneae). Close examination reveals differences in both glumes and lemmas. Furthermore, spikelets are paired in Microstegium and 1-flowered in Leersia. The lemmas of M. vimineum can be awned or awnless whereas they are always awnless in Leersia (Mehrhoff 2000).
- Adams (2009) states that human management ultimately may be easier than resource management for controlling the spread of species such as Microstegium vimineum. Suggested strategies which may have the potential to affect the rate of spread of Microstegium vimineum include: outreach and education programs about the spread of invasive species for visitors of natural areas, brushing off bootlaces with a wire brush to remove seeds, avoiding patches of invasive species when they are in seed, and wearing footwear which minimizes the risk of carrying seeds away from the infested site (Adams and Engelhardt 2009).
Appendix 8B: Risk Management Considerations for Microstegium vimineum (Japanese stiltgrass)
- Values at Risk
- Potential Mitigation Measures for Natural Means of Dispersal
- Potential Mitigation Measures for Intentional Introduction Pathways
- Potential Mitigation Measures for Non-intentional Introduction Pathways
Values at Risk
Microstegium vimineum prefers wetlands and forested areas. It is difficult to assign a value to the biodiversity of our Canadian landscape
Potential Mitigation Measures for Natural Means of Dispersal
No potential mitigation measures for natural means of dispersal are highlighted herein for Microstegium vimineum. Microstegium vimineum has disturbed many areas of the New England States and is approaching the Canadian border. Regulating the dispersal by surface water runoff and the migration of wild animals is nearly impossible to control.
Potential Mitigation Measures for Intentional Introduction Pathways
No intentional pathways were identified
Potential Mitigation Measures for Non-intentional Introduction Pathways
Field Crops Not Intended for Propagation
Previous imports
- Based on the information available in the CFIA's Import Permit System, Import Retrieval System, and information compiled at the CFIA's Import Service Centres, Microstegium vimineum has not been recently imported into Canada.
- Birdfeed has been reported in EPPO (2008) as a possible pathway for the introduction of Microstegium vimineum, although no such North American references were found. Birdseed can be imported as mixes or as lots of pure grain. Lots of pure grain are imported to Canada, then mixed, packaged and sold on the domestic retail market. Lots for domestic or wild birds are generally composed of several kinds of grains, whole or broken. More than 30 crop species are used for birdseed mixes, some of the major birdseed component crops include millet, sorghum and niger.
Potential risk mitigation measures
Regulate Microstegium vimineum as a quarantine pest under the Plant Protection Act. Add this species to the List of Pests Regulated by Canada (CFIA 2009) in order to:
- prevent the importation, movement, and cultivation of this species in Canada.
- enable inspectors to take appropriate action for the purposes of eradicating the pest or preventing its spread Footnote 1.
Regulatory actions under the Plant Protection Act could include one or more of the following:
- Exporters could be required to provide a Phytosanitary Certificate with or without an Additional Declaration stating freedom from Microstegium vimineum.
- Recognition of Pest Free Areas – if Microstegium vimineum can be shown to be absent in the country/state/region from which the field crop product was propagated, then risk is negligible and additional phytosanitary requirements may be waived.
- A Permit to Import indicating specific import requirements and conditions for the pest status, handling and use of the commodity could be required. It is the importer's responsibility to apply for and obtain the permit.
- Provisions for importation of grain contaminated with Microstegium vimineum for research, processing, industrial or educational uses under a section 43 permit on a case-by-case basis.
- Phytosanitary measures may be waived for those commodities that have been treated or processed such that the risk of introduction of Microstegium vimineum has been reduced to an acceptable level. End uses impact risk and may therefore impact required risk mitigation measures. For example, field crop commodities containing Microstegium vimineum imported for milling (grinding) into flour would have lower risk than that coming in for direct use as pastured/rangeland animal feed. For milling, risk mitigation measures would include: the milling itself and proper disposal (incineration, grinding or deep burial) of anything removed from the commodity prior to the milling process. The same is true of field crop commodities imported for processing into feed (pelleting, crushing, etc.). Commodities imported for human consumption or direct feeding to livestock would have greater risk and could require treatment to render the grain unviable. Treatments, where applicable, such as heat (i.e. where heat would not compromise quality of grain for intended end use), grinding, pelletizing could be used prior to import or post-entry.
- All risk mitigation measures for field crop commodities containing Microstegium vimineum must be taken with consideration for requirements/measures for pests other than plants (e.g. pathogens and insects).
- Combine harvesting equipment will likely remove the majority of weed seeds out of the grain during harvest. The seeds are unlikely to enter Canada in field crops not intended for propagation.
Trade implications
- Exporting countries may have to devote resources towards inspection and issuance of phytosanitary certificates. Laboratories in exporting countries will need to be able to identify seeds of Microstegium vimineum. Exporters will need to ensure freedom of Microstegium vimineum in field crop shipments, otherwise CFIA can refuse import.
- Potential for reduction and/or loss of some import markets for Canada should exporting countries be unable to meet proposed phytosanitary requirements.
- Not controlling import and spread of the species may result in infestations of field crops for export; if the importing country prohibits that species, market access for Canadian commodities could be compromised.
Cost-effectiveness and Feasibility
- Only a low proportion of imported grain is likely to contain Microstegium vimineum seeds, which reduces the impact of regulating this pathway. This measure is already in place for other pests regulated by Canada. Minor costs will be incurred for identification training for inspectors.
- Microstegium vimineum is easily confused with Leersia virginica Willd., a native grass species. Confusion arises in the field as these two species often are found growing together, have similar leaf sizes and shapes and are similar in overall appearances.
- The seeds of these two species are readily distinguishable as the plants belong to different tribes with very different floral morphologies. This mitigation measure should be feasible and minor costs will be incurred for identification training for inspectors.
Hay and Straw
Previous imports
- The total value of hay and straw imports was approximately $13 million in 2008 for "Swedes, Mangolds, Fodder Roots, Hay, Clover, Sainfoin, Forage Kale, Pellets" (HS 121490) and "Cereal Straw and Husks, Unprepared" (HS 121300). Ninety-seven percent of this value originated from the United States (Industry Canada 2009).
- Between 2006 and 2008 and using the above HS codes, the approximate value of hay and straw imports was $2.2 million from the United States, India, China and Japan, where Microstegium vimineum is present (Industry Canada 2009).
- Between 2004 and 2008 and using the above HS codes, the majority of hay and straw (approximate total of $44 million) was imported from Washington where Microstegium vimineum is not present. This represents the majority of hay and straw imported into Canada (Industry Canada, 2009).
- The level of risk associated with imports of hay and straw into Canada is relatively low since they originate primarily from areas where Microstegium vimineum is not present (i.e., Washington state).
Potential risk mitigation measures
Regulate Microstegium vimineum under the Plant Protection Act as a quarantine pest by placing it on the List of Pests Regulated by Canada. This will:
- Enable inspectors to take appropriate action for the purposes of eradicating the pest or preventing its spread Footnote 2.
- Require importers to obtain a Phytosanitary Certificate with or without an Additional Declaration stating freedom from Microstegium vimineum.
- A Permit to Import indicating specific import requirements and conditions for the pest status, handling and use of the commodity could be required. It is the mporter's responsibility to apply for and obtain the permit.
- For hay or straw intended for use as a biofuel stock, heat treatment or pelletization could be required.
Trade implications
Exporting countries will need to devote resources towards inspection of hay and straw commodities and issuance of phytosanitary certificates. Inspectors in exporting countries need to be able to identify seeds of Microstegium vimineum that could be associated with hay and straw. Exporters need to ensure freedom of Microstegium vimineum in hay and straw, otherwise CFIA can refuse import.
Cost-effectiveness and Feasibility
This measure will facilitate trade of hay and straw with the State of Alabama, Connecticut and Massachusetts, where Microstegium vimineum is regulated. This measure is already in place for other pests regulated by Canada. Minor costs will be incurred for training of inspectors
Vehicles and Used Farm Machinery
Previous imports
- A considerable volume of vehicles cross the U.S.-Canada border every year.
- Information is not available on the volume of imports of used farm machinery.
Potential Risk mitigation measures
Enforcement of Directive 95-26: "Phytosanitary requirements for soil and related matter, alone or in association with plants" (CFIA 2008).
In 2003, the Canada Border Services Agency (CBSA) assumed responsibility for the initial import inspection services in respect to the Acts and Regulations administered by the CFIA to the extent that they are applicable at Canadian border points. The inspection of goods that may be contaminated with soil are among the responsibilities that were transferred to the CBSA in 2003. The Food, Plant and Animals Programs Product of the CBSA is currently finalizing its Standard Operating Procedures (SOP) concerning the "Inspection of Imported Goods Potentially Contaminated with Soil." This SOP provides the CBSA's Border Services Officers with formal procedures for the inspection and disposition of goods that may be contaminated with soil, including used agricultural machinery and vehicles.
Nursery Stock with soil
Previous imports
The total value of imports of nursery stock was $173 million in 2008 (Industry Canada 2009) with approximately 80% of that value originating from the United States (Industry Canada, 2009; based on data using HS 0602 – Other Live Plants (incl. Roots), Cuttings and Slips; Mushroom Spawn). Approximately 40% of the imports from the United States originate from states where Microstegium vimineum is present (e.g., Florida, Pennsylvania, Tennessee, New York, Ohio)
Potential Risk mitigation measures
- Phytosanitary measures, such as nursery certification programs and "pest-free places of production" may be considered if warranted.
- Revision of D-95-26 (CFIA 2008b)to include Microstegium vimineum within its scope (i.e., sections 3.0 and 6.0).
Trade implications
- Soil and related matter is already a highly regulated commodity, and as such trade implications should be relatively minimal. However, the addition of a requirement for declaration of freedom from Microstegium vimineum from those states where it is present would increase the requirements already in place.
Cost-effectiveness and Feasibility
Nursery Stock from infested areas where Microstegium vimineum is present may be imported under the condition that the plants are bare root. Alternatively, using soil free growing media may be a condition of entry imposed on these imports. This mitigation measure should be feasible and minor costs will be incurred.
Used Recreational Equipment and Clothing
Previous imports
- Numerous travelers cross the U.S.-Canada border all year round or fly into Canada from various countries, but foreign travelers usually bring clean clothes with them into Canada. Only Canadians coming back into Canada from a site where Microstegium vimineum is prevalent could potentially carry seed or propagules attached on their clothes. The risk depends on the species' habitats and if these sites at risk are usually visited by travelers.
- Travelers entering Canada by land are more likely to travel with their pet and recreational equipment. The risk will depend on the presence of the species along the Canadian border.
Risk mitigation measures
Increase public awareness of the regulation and the risk posed by this plant and distribute awareness material at border crossings to the public
- Not considered effective by itself if the risk is high, but is a viable action if the level of risk is low.
- Monitor for the presence of Microstegium vimineum in Canada where there are suitable conditions for its establishment.
Trade implications
There are no trade implications anticipated.
Cost-effectiveness and Feasibility
- The most likely pathway for entry for Microstegium vimineum is via attachment to the fur of animals or on human clothes or in mud on tires or hiking boots. The insidious nature of these main pathways could undermine phytosanitary measures that would be put in place to prevent the entry of the species through other pathways.
- Certain states in which Microstegium vimineum is found are adjacent to the Canadian border, in particular Ohio, Pennsylvania and New York. However, with the exception of presence noted in Pennsylvania in a county proximate to Lake Erie, reports from the said states are from counties not adjacent to the border (USDA Plants Database).
- Given the challenge in determining movement of individuals and wildlife from the United States, then within Canada, monitoring for Microstegium vimineum would need to be conducted at key locations not necessarily restricted to border locations. Some training of survey staff would be required.
- A combination of public awareness campaigns and monitoring efforts could serve, respectively, to lower the risk that Microstegium vimineum is introduced through these pathways in question, and to verify the effectiveness of any phytosanitary measures that would be put in place.
Appendix 8C: Import Data for field crops used as birdfeed
Import data for field crops not intended for propagation originating from countries where Microstegium vimineum is present (2006-2008).
Value in Canadian Dollars ($)
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| US-Pennsylvania | 0 | 0 | 1,527 |
| US-Maryland | 0 | 20 | 0 |
| India | 6,041 | 17,386 | 22,618 |
| China, P. Rep. | 0 | 395 | 1,271 |
| Subtotal: | 6,041 | 17,801 | 25,416 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| US-New York | 142,096 | 38,700 | 66,060 |
| US-New Jersey | 21,744 | 30,566 | 59,891 |
| US-Georgia | 19,680 | 0 | 0 |
| US-Tennessee | 16,363 | 1,199 | 21,046 |
| US-Pennsylvania | 0 | 7,041 | 29,210 |
| US-Maryland | 2,322 | 2,569 | 9,884 |
| US-Massachusetts | 1,188 | 4,213 | 7,262 |
| US-Connecticut | 0 | 5,346 | 1,663 |
| US-Kentucky | 348 | 1,823 | 0 |
| US-North Carolina | 4,743 | 229 | 0 |
| US-Virginia | 404 | 0 | 0 |
| US-Indiana | 188 | 0 | 0 |
| China, P. Rep. | 277,389 | 307,425 | 503,778 |
| India | 287,150 | 181,626 | 150,134 |
| Japan | 5,380 | 1,158 | 8,798 |
| Subtotal: | 778,995 | 581,895 | 857,726 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| US-Tennessee | 184,372 | 229,795 | 334,187 |
| US-Georgia | 114,666 | 62,724 | 0 |
| US-Indiana | 124,175 | 133,945 | 190,411 |
| US-New York | 13,093 | 15,987 | 55,456 |
| US-Maryland | 224 | 209 | 326 |
| US-New Jersey | 1,116 | 70,876 | 7,013 |
| US-West Virginia | 0 | 0 | 74 |
| US-North Carolina | 0 | 71 | 13,325 |
| US-Pennsylvania | 1,925 | 183 | 943 |
| US-Kentucky | 0 | 328 | 72 |
| US-Connecticut | 60 | 2,042 | 0 |
| US-Massachusetts | 0 | 505 | 0 |
| US-Virginia | 0 | 0 | 0 |
| China, P. Rep. | 1,520,668 | 1,431,938 | 1,889,796 |
| Japan | 52,639 | 52,063 | 64,184 |
| India | 0 | 45,464 | 119,947 |
| Subtotal: | 2,012,938 | 2,046,130 | 2,675,734 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| US-New York | 662,960 | 671,273 | 552,490 |
| US-Maryland | 252,972 | 214,538 | 396,836 |
| US-Tennessee | 60 | 94,530 | 612,252 |
| US-New Jersey | 140,242 | 65,932 | 114,648 |
| US-Georgia | 45,166 | 75,071 | 68 |
| US-North Carolina | 100,672 | 60,710 | 26,164 |
| US-Virginia | 33,574 | 9,241 | 9,863 |
| US-Indiana | 6 | 29,429 | 12,653 |
| US-Kentucky | 5,672 | 19,292 | 4,163 |
| US-Pennsylvania | 1 | 13,318 | 7,123 |
| US-Connecticut | 14,940 | 0 | 0 |
| US-Massachusetts | 1,883 | 2,549 | 0 |
| US-West Virginia | 0 | 8 | 342 |
| India | 7,933,564 | 8,574,285 | 14,632,712 |
| China P. Rep. | 2,727,886 | 3,808,928 | 4,020,699 |
| Japan | 62,372 | 110,871 | 155,928 |
| Subtotal: | 11,981,970 | 13,749,975 | 20,545,941 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| Total: | 14,779,944 | 16,395,801 | 24,104,817 |
Quantity in metric tonnes (TNE)
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| US-Pennsylvania | 0 | 0 | 5 |
| US-Maryland | 0 | 0 | 0 |
| India | 22 | 57 | 71 |
| China, P. Rep. | 0 | 1 | 5 |
| Subtotal: | 22 | 58 | 81 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| US-New York | 14,635 | 489 | 7,855 |
| US-New Jersey | 5,359 | 12,053 | 34,715 |
| US-Georgia | 90,682 | 0 | 0 |
| US-Tennessee | 65,191 | 4,534 | 90,872 |
| US-Pennsylvania | 0 | 14,632 | 55,086 |
| US-Maryland | 441 | 572 | 3,158 |
| US-Massachusetts | 1,947 | 2,582 | 4,672 |
| US-Connecticut | 0 | 4,848 | 1,167 |
| US-Kentucky | 454 | 1,814 | 0 |
| US-North Carolina | 4,170 | 113 | 0 |
| US-Virginia | 114 | 0 | 0 |
| US-Indiana | 37 | 0 | 0 |
| China, P. Rep. | 213,063 | 409,492 | 540,074 |
| India | 226,297 | 271,519 | 145,113 |
| Japan | 1,062 | 82 | 2,486 |
| Subtotal: | 623,452 | 722,730 | 885,198 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| US-Tennessee | 527 | 512 | 472 |
| US-Georgia | 368 | 140 | 0 |
| US-Indiana | 24 | 27 | 30 |
| US-New York | 13 | 9 | 25 |
| US-Maryland | 0 | 0 | 0 |
| US-New Jersey | 1 | 43 | 3 |
| US-West Virginia | 0 | 0 | 0 |
| US-North Carolina | 0 | 0 | 10 |
| US-Pennsylvania | 1 | 0 | 1 |
| US-Kentucky | 0 | 0 | 0 |
| US-Connecticut | 0 | 1 | 0 |
| US-Massachusetts | 0 | 1 | 0 |
| US-Virginia | 0 | 0 | 0 |
| China, P. Rep. | 1,335 | 933 | 928 |
| Japan | 8 | 0 | 6 |
| India | 0 | 37 | 55 |
| Subtotal: | 2,277 | 1,703 | 1,530 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| US-New York | 2,250 | 1,920 | 1,307 |
| US-Maryland | 101 | 73 | 91 |
| US-Tennessee | 0 | 327 | 1,627 |
| US-New Jersey | 103 | 35 | 51 |
| US-Georgia | 102 | 164 | 0 |
| US-North Carolina | 473 | 249 | 63 |
| US-Virginia | 145 | 37 | 22 |
| US-Indiana | 0 | 2 | 4 |
| US-Kentucky | 2 | 2 | 1 |
| US-Pennsylvania | 0 | 6 | 3 |
| US-Connecticut | 5 | 0 | 0 |
| US-Massachusetts | 0 | 1 | 0 |
| US-West Virginia | 0 | 0 | 0 |
| India | 7,094 | 6,538 | 7,282 |
| China P. Rep. | 1,179 | 1,578 | 1,255 |
| Japan | 13 | 36 | 36 |
| Subtotal: | 11,467 | 10,968 | 11,742 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| Total: | 637,218 | 735,459 | 898,551 |
Source: Statistics Canada 2009.
Note: Countries with an export value of less than $1,000 per year are grouped together and a summed value for all countries is shown.
Appendix 8D: Import Data for Hay and Straw
Import data for hay and straw originating from countries where Microstegium vimineum is present (2006-2008).
Import Data for Hay and Straw, Value in Canadian Dollars ($)
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| US-North Carolina | 272,458 | 316,202 | 212,617 |
| US-New York | 8,984 | 10,260 | 16,288 |
| US-Maryland | 3,151 | 7,596 | 8,908 |
| US-Massachusetts | 0 | 0 | 483 |
| US-New Jersey | 1,917 | 266 | 106 |
| US-Virginia | 0 | 0 | 6 |
| US-Georgia | 0 | 60,000 | 0 |
| US-Pennsylvania | 530 | 3,141 | 0 |
| US-Connecticut | 571 | 249 | 0 |
| US-Indiana | 5,682 | 0 | 0 |
| India | 284,039 | 258,851 | 302,025 |
| China | 263 | 0 | 5,744 |
| Sub-Total: | 577,595 | 656,565 | 546,177 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| US- Pennsylvania | 22,699 | 20,272 | 101,167 |
| US-New Jersey | 44,162 | 37,891 | 48,900 |
| US-Kentucky | 619 | 30 | 15,743 |
| US-New York | 24,965 | 1,808 | 228 |
| US-Maryland | 277 | 190 | 220 |
| US-Virginia | 67 | 20 | 20 |
| US-Indiana | 1,546 | 8 | 1 |
| US-Massachusetts | -- | 10 | -- |
| US-Georgia | -- | 10 | -- |
| China P. Rep. | 13,007 | 12,363 | 32,527 |
| Japan | 4,223 | 1,051 | 1,076 |
| India | 2,933 | 28,297 | 76 |
| Sub-Total: | 114,498 | 101,950 | 199,958 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| Total: | 692,093 | 758,515 | 746,135 |
Source: Statistics Canada 2009.
Note: Countries with an export value of less than $1,000 per year are grouped together and a summed value for all countries is shown.
Appendix 9A: Pest Risk Assessment Summary for Nassella trichotoma (serrated tussock)
- Identity of Organism
- Organism Status
- Current Regulatory Status
- Probability of Entry
- Probability of Establishment
- Probability of Spread
- Potential Economic Consequences
- Potential Environmental and Social Consequences
- Uncertainty
- Conclusion
- Technical Issues for Consideration
Identity of Organism
Name: Nassella trichotoma (family Poaceae, subfamily Pooideae, tribe Stipeae) (USDA-NRCS 2009).
Synonyms: Stipa trichotoma (1829), Urachne trichotoma (1834), Agrostis trichotoma (1841), Urachne macrathera (1853), Piptochaetium trichotomum (1879), Piptatherum macrantherum (1894), Stipa macrathera (1901), nom. illeg., non Stipa macrathera (1896), Oryzopsis trichotoma (1910).
English common names: Serrated tussock, serrated tussock grass, nassella tussock, Yass tussock, Yass River tussock, tumbleweed.
French common names: Stipe à feuilles dentées
Description: Microstegium vimineum is an annual grass with a sprawling habit. It germinates in spring and grows slowly through the summer months, ultimately reaching heights of 0.6-1 m with the reclining stems growing up to 1 metre long. Slender stalks of tiny flowers are produced in August through to September or early October. In late fall they fade to pale greenish-yellow or turns purple in colour (Swearing 2004, Mehrhoff et al. 2003).
Nassella trichotoma is a tall (at least 60 cm), tussock-forming, perennial grass of the tribe Stipeae (family Poaceae), native to the pampas of South America and introduced into dry, warm-temperate regions in other parts of the world, primarily in the southern hemisphere. In Australia and New Zealand it is a serious, invasive weed in pasture lands and is considered one of their most serious weed problems. In pasture, it reduces the carrying capacity due to its low nutritive value and poor palatability to livestock.
Organism Status
Nassella trichotoma is not reported to occur in Canada, and no evidence was found that it iscultivated here (CFIA 2008, Scoggan 1979). Based on this information, it is considered to be absent from the PRA area.
Nassella trichotomahas not yet become established in North America:
- Kartesz (1999) does not list this species for North America, including Hawaii and Puerto Rico.
- The Manual of Grasses for North America (Barkworth 2006) and the Plants Database (USDA-NRCS 2006) map records for Nassella trichotoma in Illinois, Kentucky, Missouri, North Carolina and South Carolina, but this seems to be based on Westbrooks and Cross (1993), which is a report of Nassella-contaminated fescue seed being distributed in those states in 1988. In spite of follow-up surveys in the affected counties, no plants were ever found growing in the field. No reports have been found to indicate that any plants became established as a result of this distribution.
- The CABI Crop Protection Compendium (CABI 2005) lists established populations in Arizona, Hawaii and Florida. However, for this information, they refer to the Plants Database (USDA-NRCS 2004) which does not currently include these states in their distribution map (USDA-NRCS 2006).
- Nassella trichotoma is not listed in Hawaii by Rotar (1968).
- No references to specimen-supported records for North America could be found in the literature.
Current Regulatory Status
Canada: Listed as Class 1 "Prohibited Noxious Weed", under the Weed Seeds Order (Seeds Act). All imported and domestic seed must be free of prohibited noxious weed seeds.
United States: Listed in the USDA-APHIS Footnote 1 Federal Noxious Weed List under the Noxious Weed Regulation (Federal Noxious Weed Act). No person may move a federal noxious weed into or through the United States, or interstate, unless this person obtains a permit for such movement (Noxious Weed Regulation).
European Plant Protection Organization (EPPO): Nassella trichotoma is not regulated in Europe, however, it was added to the EPPO Alert List because it presents a risk to the EPPO region. The objective of the EPPO Alert List is to provide early warning and eventually to propose candidates which may be subjected to a PRA (EPPO 2009).
Probability of Entry
The main pathway through which Nassella trichotoma is likely to be introduced into Canada is in seed lots of forage grasses for planting. However, the species does not seem to be a common weed in seed production fields in either its native range or where introduced and it has only rarely been intercepted in imported lots. Nassella trichotoma has been listed as a prohibited noxious weed seed on the Weed Seeds Order: this should greatly reduce the likelihood of it entering Canada as a seed contaminant. A second pathway is available through the sale of plants and seeds for planting in home gardens. See table 1, Summary of Pathways for Nassella trichotoma (serrated tussock).
| Type of pathway | Specific Pathways |
|---|---|
| Natural dispersal | Seed: the wind distributes seeds over long distances. |
| Intentional Introduction |
|
| Unintentional Introduction |
|
Probability of Establishment
The potential range of Nassella trichotoma in Canada would be very limited. Its range in South America and Australia suggests that it would be confined to Canadian Plant Hardiness Zone 8, possibly extending into zone 7. Even if zone 7 is included, the potential range would be limited to coastal British Columbia where there is a limited amount of suitable habitat (Figure 1).
Climate modelling, based on Nassella trichotoma's current worldwide range, limits the potential distribution even further to extreme Southwestern BC
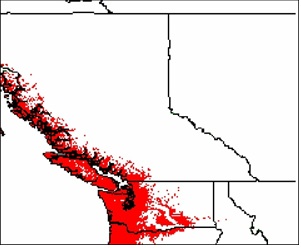
Description of Figure 1
This map shows the potential range of Nassella trichotoma (serrated tussock) in Canada, more specifically in British Columbia. Red is used to indicate the regions in which Nassella trichotoma could survive according to the Canadian Plant Hardiness Zones map, in this case NAPPFAST Hardiness Zone 8 and possibly zone 7. In Canada, the potential range of Nassella trichotoma is very limited to only coastal British Columbia. This image is narrowed to include the lower coastal regions of BC as well as Washington and Oregon which all have a scattered presence of red.
NAPPFAST zones 7-9
Probability of Spread
Natural spread potential is high. Nassella trichotoma spreads by seed, which may be produced in large quantities. The large panicles break off and blow in the wind, like tumbleweeds, distributing seeds over long distances in open habitats. Under suitable conditions, the plant can spread very rapidly. The seeds can persist for many years in the soil. Since Nassella trichotoma does not usually grow in cultivated fields, this reduces the likelihood of the seeds being moved around on machinery.
Potential Economic Consequences
Potential economic impact is not significant. The primary economic impact of Nassella trichotoma is a reduction in carrying capacity of range and pasture. As most of the ranching areas in Canada are outside of the potential range, the impact would be limited. In badly-infested areas in Australia land can become unusable for pasture, greatly reducing land values. A similar level of impact is unlikely to occur in Canada.
Potential Environmental and Social Consequences
Potential environmental impact is high. Nassella trichotoma has demonstrated an ability to out-compete native and introduced grasses on grazed lands where it has been introduced in Australia, New Zealand and South Africa. It has the potential to become established in native grasslands and lightly forested terrain, including the Garry oak meadows of Southwestern BC. While this area is very limited (see Figure 2), these meadows represent a critically endangered ecosystem within Canada, which provides habitat for about 54 species of vascular plants and three butterfly taxa that are listed as endangered or threatened in Canada.
There are no reports of toxicity to livestock or humans, but domestic animals have died from heavy feeding on Nassella trichotoma due to the indigestible material forming large balls in the stomach. Heavy infestations of conservation lands with Nassella trichotoma decrease their biological diversity, as well as their aesthetic value.
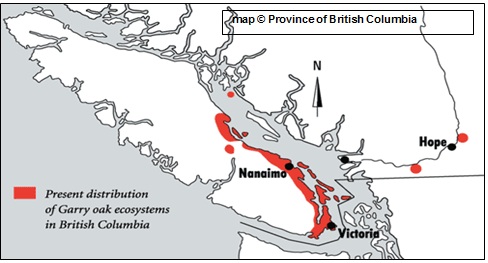
Description of Figure 2:
This map shows the distribution of Garry oak ecosystems in British Columbia through the use of the colour red. It is present, predominantly, on the coastal region of Southern Vancouver Island surrounding the Victoria and Nanaimo regions extending slightly upwards. The distribution of the Garry Oak ecosystem can be found on mainland BC as well which is signified by the two red circles in the vicinity of Hope. There is a black and white arrow on the mainland of the province pointing upwards to identify the direction of north.
Source: GOERT 2009
Uncertainty
This assessment is based on the current understanding of the species' range and climatic tolerances. There are no records of the plant becoming established beyond the equivalent of a Canadian hardiness zone 8. Any evidence to show that it could survive in zone 6 or beyond would dramatically affect this assessment. Establishment potential and economic impact, in particular, would have to be re-assessed. If it was determined that the species was not a threat to endangered species in the Garry oak meadows of Vancouver Island, the environmental impact could be down-graded.
Conclusion
The overall risk associated with Nassella trichotoma is moderate. This indicates that specific phytosanitary measures are necessary. In 2005, the species was listed as a prohibited noxious weed seed. This should greatly reduce the likelihood of Nassella trichotoma being introduced into Canada. The importation and sale of plants for the public should also be regulated. The combination of these two measures would provide adequate phytosanitary protection to Canada against this species. It is recommended that Nassella trichotoma be considered for inclusion on the List of Pests Regulated by Canada.
Technical Issues for Consideration
None
Appendix 9B: Risk Management Considerations for Nassella trichotoma (serrated tussock)
- Values at Risk
- Potential Mitigation Measures for Natural Means of Dispersal
- Potential Mitigation Measures for Intentional Introduction Pathways
- Potential Mitigation Measures for Non-intentional Introduction Pathways
Values at Risk
Seed Industry
In 2007, 162,200 tons of Canadian seed were exported to the U.S., for an approximate value of $197.6 million. If Nassella trichotoma becomes established in Canada, it could complicate seed trade between the U.S. and Canada (Statistics Canada in Agriculture Canada 2008). On several occasions forage grass seed lots of South American origin have been found to be contaminated with seeds of this weed. This has caused problems for the Canadian seed trade when seed lots containing serrated tussock seeds were re-exported to the U.S. It was largely to address these trade concerns that the species was added to the Weed Seed Order as a prohibited noxious weed seed in July 2005.
The potential range of Nassella trichotoma in Canada is limited to the West Coast of British Columbia, where there is almost no forage seed production (Statistics Canada 2007). Its direct impact on seed trade is thus relatively low and is limited to re-exportation to the U.S (Table 2).
Garry Oak Ecosystem
The Garry oak ecosystem is rare and irreplaceable because of its historical and ecological significance. This landscape contributes both to the sense of place and regional identity of the inhabitants of Vancouver Island.
The British Columbia Ministry of Environment, Lands and Parks has two Ecological Reserves that primarily focus on Garry oak ecosystems. The Garry Oak Meadow Preservation Society, the British Columbia Conservation Data Centre and the Canadian Forest Service have initiated programs to promote the preservation of Garry oak ecosystems in Canada.
| Forage crops | 2004 | 2005 | 2006 | 2007 |
|---|---|---|---|---|
| Alfalfa | 8,635 | 69,818 | 69,691 | 8,699 |
| Clover | 9,286 | 16,358 | 28,458 | 41,981 |
| Fescue | 0 | 0 | 0 | 0 |
| Rye grass | 47,926 | 37,046 | 0 | 0 |
| Timothy grass | 0 | 0 | 0 | 0 |
| Other forage plants | 10,497 | 180,974 | 88,191 | 253,822 |
| Total: | 78,348 | 306,201 | 188,346 | 306,509 |
Source: Statistics Canada in Industry Canada 2009.
Cost of control
If Nassella trichotoma becomes established in natural ecosystems, including the Garry oak ecosystem of BC, it could be arduous and costly to eradicate.
The most effective control method in New Zealand, chipping combined with herbicide application, is expensive both in labour costs and cost of chemicals. Control costs vary with the degree of infestation and land use (arable versus non-arable) (Vere and Campbell 1984). Costs for control on moderately to heavily-infested land were estimated at $98.50 to $107.35Footnote 1 per hectare (Vere and Campbell 1984). The same authors explain that continued treatment of heavily-infested land must be continued for up to 22 years before an economic benefit can be expected.
Glyphosate seems to be the only pesticide that is currently registered in Canada that has been successful in controlling Nassella trichotoma. Glyphosate, a non-selective herbicide, kills a broad spectrum of plants. Its use would affect native species within the treated area, including endangered species (Allison 2006).
Potential Mitigation Measures for Natural Means of Dispersal
No measures are required. Nassella trichotoma is not present along the Canadian border. It is not likely to enter Canada by natural means.
Potential Mitigation Measures for Intentional Introduction Pathways
Plants for Planting excluding Seed
Previous imports
The CFIA considers Nassella trichotoma as not cultivated in Canada.
- Based on available information, no plant in the Nassella genus has been recently imported into Canada (CFIA internal data).
- There are anecdotal records of Nassella trichotoma being sold in Vancouver, British Columbia (UBC Botanical Garden 2006, UBC Botanical Garden 2008). This plant is not otherwise available in the Canadian market (CNLA 2009b).
Potential risk mitigation measures
Non-regulatory measures
- 1. Use sterile cultivars if they exist.
- 2. Encourage voluntary cessation of the sale of Nassella trichotoma. Voluntary cessation is not effective by itself, but could support other measures.
- 3. Increase public awareness of the risk posed by this plant, publish a factsheet online, and distribute awareness material to garden centers, botanical gardens, gardener associations, horticulture industry groups, etc. This measure is not effective by itself, but could support other measures.
Regulatory measures
- 4. Allow sale of Nassella trichotoma with special conditions such as not to be grown close to natural areas and mandatory control of adventives. This is not considered effective because once the plant is grown in private gardens; CFIA does not have adequate resources for monitoring populations.
- 5. Regulate Nassella trichotoma under the Plant Protection Act as a quarantine pest:
- Refuse to issue Permits to Import for plants of Nassella trichotomaFootnote 2;
- Will require that importers of material from regions other than the continental U.S. specify the scientific name of any species of Nassella when applying for a Permit to Import plants and propagative material;
- Require that the scientific name of any Nassella species be provided on the Phytosanitary Certificate for all plants exported to Canada from the continental U.S. However, imports from the U.S. are very unlikely to occur because: (1) Nassella trichotoma is a Federal Noxious Weed in the U.S. and its cultivationand movement is prohibited; and (2) Nassella trichotoma is not commonly sold as an ornamental plant.
Trade implications
- The loss of market is expected to be very low because (1) Nassella trichotoma is a Federal Noxious Weed in U.S. and most imports of plants for planting to Canada originate in the U.S.; and (2) Nassella trichotoma is not a popular ornamental plant and alternative ornamental grasses are readily available.
- Foreign exporters would need to obtain phytosanitary certificates for any Nassella species. National Plant Protection Organizations of the exporting country should be able to distinguish Nassella species.
Cost-effectiveness and Feasibility
- No additional resources would be required at the CFIA Plant Health and Biosecurity Permit Office. Applications for a Permit to Import are reviewed by officers on a regular basis.
- An important increase in demand for plant and seed identification is not expected on the CFIA laboratory. Plants and seeds of Nassella trichotoma are readily identifiable.
Seed of Nassella trichotoma
Previous imports
Unknown. No figures are available specifically for Nassella trichotoma.
Potential risk mitigation measures
- 1. Maintain regulation of Nassella trichotoma as a Class 1, prohibited noxious weed, under the Weed Seeds Order of the Seeds ActFootnote 3.
- This species meets the definitions for Class 1Footnote 4 species under the Weed Seeds Order.
- The importation and sale of Class 1 seeds are prohibited in Canada. Imported seed lots require a certificate of analysis stating Nassella trichotoma is absent from the seed lot before it can be imported.
- 2. Regulate as a quarantine pest under the Plant Protection Act and add this species to the List of Pests Regulated by Canada (CFIA 2009) in order to:
- Prevent the importation, movement, and cultivation of this species in Canada. Currently, seed of many new crop species, such as field crops, can be imported without a Permit to Import or a Phytosanitary Certificate from throughout the world.
- Enable inspectors to take appropriate action for the purposes of eradicating the pest or preventing its spreadFootnote 5.
- 3. Regulatory actions under the Plant Protection Act could include the following:
- Prohibit importation of Nassella trichotoma seed and refuse to issue Permits to Import for seed of Nassella trichotomaFootnote 6. Require importers of plant material from the non-continental U.S. to apply for a Permit to Import with scientific name specified.
- Seed of horticultural plants is not within the scope of CFIA's draft directive D-08-04 on plants for planting (CFIA 2008). Therefore phytosanitary requirements will be specified under a new regulatory policy directive or D-08-04 will be revised.
Trade implications
The loss of market is expected to be very low (see Section V, c. Trade Implications).
Cost-effectiveness and Feasibility
The CFIA Seed Program is already in place to prevent the entry of prohibited noxious weeds. Verification of compliance is carried out through the Seed Marketplace Monitoring ProgramFootnote 7.
Potential Mitigation Measures for Non-intentional Introduction Pathways
Seed of Forage Grasses
Previous imports
- The total value of forage grass seedFootnote 8 imported in 2008 was around $26 million (Industry Canada 2009).
- The total value of forage grass seeds imported in 2008 from countries where Nassella trichotoma is present was 12% of total imports, for a value of $3.1 million (see Figure 3 and Appendix 9C).
Potential risk mitigation measures
- Maintain the regulation of Nassella trichotoma as a prohibited noxious weed (Class 1) under the Weed Seeds Order of the Seeds Act.
- This species meets the definitions for Class 1 species under the Weed Seeds Order.
- All imported and domestic seed lots must be free of prohibited noxious weed seeds. Imported seed lots would require a certificate of analysis stating Nassella trichotoma is absent from the seed lot before it can be imported.
- Regulate Nassella trichotoma as a quarantine pest under the Plant Protection Act and add this species to the List of Pests Regulated by Canada (CFIA 2009).
- This could prevent the importation, movement, and cultivation of this species in Canada.
- Exporters could be required to obtain a phytosanitary certificate stating freedom from Nassella trichotomaFootnote 9.
- Phytosanitary agreements could be negotiated to certify imports from pest free areas and/or recognize noxious weed certification in countries or states of origin.
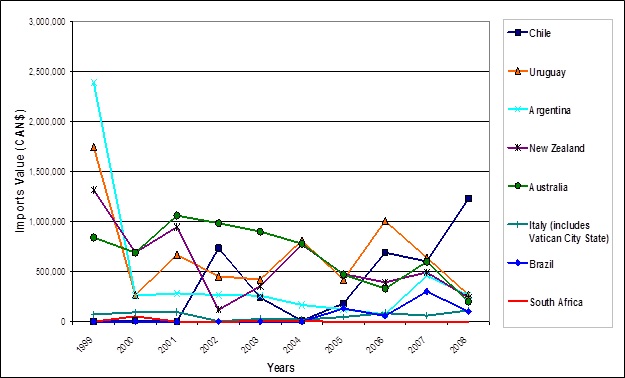
Description of Figure 3
This figure depicts the imports of forage seed into Canada from countries where serrated tussock is present from 1999 to 2008. These include Chile, Uruguay Argentina, New Zealand, Australia, Italy, Brazil and South Africa. In 1999, Canada received the most imports from Argentina (approximately $2,400,00 CAN worth) and in 2008 Canada received the most imports from Chile (approximately $1,250,000 CAN worth).
Trade implications
- This measure facilitates seed trade with the U.S. where Nassella trichotoma is listed as a Federal Noxious Weed. It is also listed as a noxious weed in 24 states.
- NPPOs of exporting countries and seed regulatory agencies will need to devote resources towards inspection of seed lots and issuance of phytosanitary certificates. Laboratories in exporting countries will need to be able to identify seeds of Nassella trichotoma within a seed sample. NPPOs of exporting countries and seed regulatory agencies need to ensure freedom of Nassella trichotoma in seed lots, otherwise CFIA can refuse import.
Cost-effectiveness and Feasibility
- Regulation of Nassella trichotoma as a Class 1 prohibited noxious weed is already in place (see Section V, c. Trade Implications).
- The seeds of Nassella trichotoma are easily identified.
Hay and Straw
Previous imports
- The total value of hay and straw imports was around $13 million in 2008; 98% of this value came from the U.S. (Industry Canada 2009).
- Between 1999 and 2004, limited quantities (less than 0.2%) of hay and straw were imported from Spain, where Nassella trichotoma is present. Since 2004, hay and straw has not been imported from countries where Nassella trichotoma is present (Industry Canada 2009).
Potential risk mitigation measures
No measures required. The level of risk associated with imports of hay and straw into Canada is relatively low because (1) they originate primarily from areas where Nassella trichotoma is not present, and (2) Nassella trichotoma rarely occurs in cultivated crops,such as cultivated grassland.
Live Animals
Previous imports
Between 1999 and 2008, no significant numbers of live sheep or goats for reproduction have been imported into Canada from countries where Nassella trichotoma is present (CFIA internal data).
Potential risk mitigation measures
No measures are required.
Raw Wool
Previous imports
The total value of raw wool imports was around $3.1 million in 2008; 70% of this value came from Australia, New Zealand and Argentina, where Nassella trichotoma is present (Industry Canada 2009).
Potential risk mitigation measures
No measures are required.
- The vegetative matter is removed from commercial wool by carbonizationFootnote 10.
- Raw wool and raw hides imported into Canada from all countries require CFIA inspection. They must be free from feces, blood, ectoparasites and dirt (plant and plant parts are considered to be dirt) or it will be refused entry (Health of Animals Act, Directive AHPD-DSAE-2001-1-1 (CFIA 1990)).
Appendix 9C: Import Data forage seeds for sowing a originating
Import data for forage seeds for sowing a originating from countries where Nassella trichotoma is present (2006-2008)
Value in Canadian Dollars ($)
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| Australia | 199,963 | 136,582 | 358,370 |
| Italy | 19,969 | 16,963 | 69,519 |
| Argentina | 71.872 | 3,122 | 0 |
| Uruguay | 1,049 | 1,475 | 0 |
| Subtotal: | 292,853 | 158,142 | 427,889 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| Chile | 405,808 | 521,345 | 1,287,309 |
| Uruguay | 0 | 0 | 307,293 |
| New Zealand | 285,837 | 507,900 | 215,955 |
| Argentina | 0 | 43,769 | 175,996 |
| Italy | 0 | 56 | 0 |
| Australia | 254,698 | 404,798 | 0 |
| Subtotal: | 946,343 | 1,477868 | 1,986,553 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| Italy | 1,843 | 0 | 42,100 |
| Subtotal: | 1,843 | 0 | 42,100 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| Argentina | 36,015 | 175,826 | 196,704 |
| Uruguay | 223,473 | 1,081 875 | 172,939 |
| New Zealand | 183 | 0 | 2,605 |
| Italy | 1,839 | 0 | 1,432 |
| Chile | 961 | 17 | 1,134 |
| Australia | 1,463 | 1 | 0 |
| Subtotal: | 263,934 | 1,257,719 | 374,814 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| Total: | 1,504 973 | 2,893 729 | 2,831,356 |
Quantity in metric tonnes (TNE)
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| Australia | 58,405 | 48,416 | 60,025 |
| Italy | 4,990 | 3,019 | 21,428 |
| Argentina | 20,286 | 513 | 0 |
| Uruguay | 150 | 175 | 0 |
| Subtotal: | 83,831 | 52,123 | 81,453 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| Chile | 152,528 | 191,132 | 488,676 |
| Uruguay | 0 | 0 | 87,500 |
| New Zealand | 61,951 | 157,840 | 62,952 |
| Argentina | 0 | 17,900 | 54,600 |
| Italy | 0 | 23 | 0 |
| Australia | 60,474 | 116,831 | 0 |
| Subtotal: | 274,953 | 484,526 | 693,728 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| Italy | 1,550 | 0 | 24,465 |
| Subtotal: | 1,550 | 0 | 24,465 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| Argentina | 8,057 | 39,585 | 60,348 |
| Uruguay | 60,000 | 219,768 | 45,000 |
| New Zealand | 33 | 0 | 615 |
| Italy | 373 | 0 | 125 |
| Chile | 4,496 | 8 | 103 |
| Australia | 272 | 0 | 0 |
| Subtotal: | 73,231 | 259,361 | 106,191 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| Total | 433,565 | 796,010 | 905,837 |
Appendix 10A: Pest Risk Assessment Summary for Paspalum dilatatum (Dallis grass)
- Identity of Organism
- Organism Status
- Current Regulatory Status
- Probability of Entry
- Probability of Establishment
- Probability of Spread
- Potential Economic Consequences
- Potential Environmental and Social Consequences
- Uncertainty
- Conclusion
- Technical Issues for Consideration
Identity of Organism
Name: Paspalum dilatatum (family Poaceae, subfamily Panicoideae, tribe Paniceae) (USDA-ARS 2009)
Synonyms: Digitaria dilatata, Panicum platense, Paspalum eriophorum, Paspalum lanatum, Paspalum ovatum, Paspalum pedunculare, Paspalum platense, Paspalum selloi, Paspalum velutinum (Tropicos 2009)
Common names: Dallis grass, Dallisgrass, water grass, paspalum, CAB International 2007; Hitchcock and Chase 1950; USDA-ARS 2009)
French common names: Herbe de Dallis (CAB International 2007; Hitchcock and Chase, 1950; USDA-ARS 2009)
Description: Dallis grass is a perennial, cespitose, C4 grass. The plants have rhizomes, but they are so short (less than 1 cm) that the stems form tufts. The stems are 50-175 cm tall, and erect. The leaf blades are up to 35 cm long, 2-16.5 mm wide, flat, mostly glabrous, with a few long hairs near the base on the upper surface. The panicles are terminal, with 2-7 racemose, divergent branches. The spikelets are 2.3-4 mm long and 1.7-2.5 mm wide. They are paired, and appressed to the branch axes. The lower glumes are absent; the upper glumes and lower lemmas are 5-7-veined, with pilose margins. The caryopses are 2-2.3 mm and white to brown (Barkworth 2006).
Dallis grass is considered a weed of 14 crops in 28 countries (Holm et al. 1977).
Organism Status
Dallis grass is not reported to occur in Canada (CFIA 2008), and no evidence was found that it is cultivated in Canada (CNLA 2009). Based on this information, it is considered absent from the PRA area.
Current Regulatory Status
- Canada:
- Paspalum dilatatum was not regulated in Canada.
- United States:
- It is not regulated as a federal or state noxious weed.
- Other countries:
- In 28 countries it is listed as a serious or principal weed
Probability of Entry
Dallis grass was deliberately introduced into the USA as a forage grass species in the mid-1800's (Hitchcock and Chase 1950). Deliberate introduction seems to have been the main pathway for long distance spread around the world.
In parts of the southern USA, dallis grass is a perennial weed problem on lawns, golf courses and other turf areas. Control in turf is very difficult (Breeden and Brosnan 2009). This suggests that seeds have been introduced as contaminants in grass seed in that region at least. This is probably the most likely pathway for introduction into Canada, as much of the lawn and turf grass seed sold in Canada is imported from the USA (Table 1). The species has also reportedly been used as a turf grass (Barkworth 2006).
| Type of pathway | Specific Pathways |
|---|---|
| Natural dispersal | Paspalum dilatatum is dispersed by means of seed. It is unlikely that natural dispersal will allow the species to enter Canada, as established populations are in the southern states. |
| Intentional Introduction | Deliberate introduction as a forage plant seems to have been the main pathway for long distance spread around the world. This is not a likely pathway for entry of Paspalum dilatatum into Canada, since it is grown intentionally only in warmer climates. |
| Unintentional Introduction | Seeds could be carried as contaminants in seed lots, especially turf grasses. However, no seeds of any Paspalum species have been reported from samples analyzed at the CFIA Seed Laboratory in the past nine years. This could change if more turf grass seed was imported from infested regions. Seeds could be carried as contaminants in grain lots. Paspalum dilatatum is a weed of rice, so it could be introduced into Canada as a contaminant in rice grain lots. This is a likely pathway for entry, but is unlikely to introduce seeds into suitable habitats. Seeds could be carried as contaminants in commodities. Paspalum dilatatum is a weed of tropical fruit crops, including pineapples. Imported pineapples have been reported to be contaminated with weed seeds such as itchgrass, so it could be possible for Paspalum dilatatum to enter by this pathway. This is a likely pathway for entry, but less likely to introduce seeds into suitable habitats. |
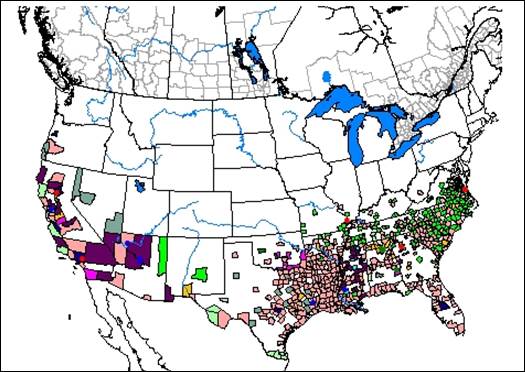
Description of Figure 1:
This image is a map of North America distinguishing the range of Paspalum dilatatum (Dallis grass) through the use of colours designated to a specific method. Red dots signify a UTC specimen; green dots equate to other herbaria; blue dots represents manual data; dark blue denotes UTC counties; magenta indicates other herbaria counties; light pink , the most predominant colour on the map, represents contributors; light green signifies state flora/atlas; dark green represents regional fora/atlas; light blue stands for reliable reports; turquoise indicates other publication on Paspalum dilatatum while grey shows unverified reports; dark green signifies a database; and mustard yellow denotes specimen by MEB, while dark purple represents herbarium database. Large amounts of light pink are condensed in southeast states from West Virginia extending down to Texas. Light green and other colours such as yellow and red are present in the coastal states of Virginia, North Carolina and South Carolina. Magenta, blue and purple as well as turquoise are apparent along the west coastal states in California and parts of Arizona as well as in New Mexico although more spread out in nature than the eastern representation.
Source: Barkworth 2006
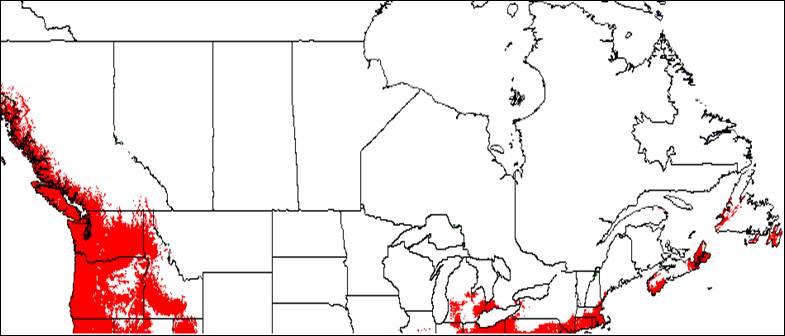
Description of Figure 2:
This image shows the potential range of Paspalum dilatatum in Canada and northern America through the use of a map. Red is used to indicate the regions in which Paspalum dilatatum could survive according to the Canadian Plant Hardiness Zones map, in this case NAPPFAST Hardiness Zones 6-9. In Canada, the potential range of Paspalum dilatatum is limited to coastal and southwestern BC, and, possibly, southern Ontario and parts of the Maritime Provinces if it is introduced to these areas. Most of the red can be found in the U.S., this includes the greater parts of the east and west coasts as well as areas located near the Great Lakes extending downwards beyond the image.
NAPPFAST zones 6-9
Probability of Establishment
Paspalum dilatatum is thought to be native to Bolivia, Brazil, Chile, Paraguay, Uruguay and Argentina (USDA-ARS 2009). The exact native range is now obscure because of the species' pantropic distribution (Weber 2003). It is now well established across the southern U.S., including Hawaii, as a weed in waste places (Barkworth 2006). Paspalum dilatatum is also naturalized in s. Europe, tropical and s. Africa, Asia, Australia, New Zealand, Macaronesia, the Mascarenes, Melanesia, and Polynesia (USDA-ARS 2009).
Based on its current range in the U.S., Paspalum dilatatum will survive to NAPPFAST zone 6. This would suggest a potential range in Canada that includes coastal and southern British Colombia, extreme southwestern Ontario and coastal parts of Nova Scotia and Newfoundland (see figure 2).
In its native range, Paspalum dilatatum is found in sandy or muddy soil near the seashore or in saline soil and swamps. In its introduced range it invades heaths, shrubland, riparian habitats and freshwater wetlands (Weber 2003). In Western Australia it is a serious weed in disturbed and natural clay pans, as well as swamps, lawns, roadsides and pastures (Hussey et al. 2007).
There would be no shortage of suitable habitats in the limited potential Canadian range.
Probability of Spread
Locally, Paspalum dilatatum plant fragments can be moved around by farm equipment, but seed is the main mode of spread. Large quantities of seed are produced, often by apomixis (Holm et al. 1977). Seeds are spread by attaching to animals and people (Holding and Bowcher 2007).
Potential Economic Consequences
Paspalum dilatatum is listed as a serious or principal weed in Australia, Philippines, Brazil, Columbia, the former Soviet Union and Taiwan (Holm et al. 1991). It can be a problem when it encroaches on irrigation ditches (Holm et al. 1977). It is particularly a weed problem in bananas, papayas, pineapples and rice in the Philippines, sugarcane, bananas, orchards and vineyards in Australia, in bananas, pineapples, papayas and sugarcane in Hawaii, tea and citrus in the former Soviet Union, rice in Brazil and India and potatoes, vegetables and pasture in New Zealand. In the United States it is a pasture weed (Holm et al. 1977). In Tennessee, Paspalum dilatatum is now a weed problem throughout the state on lawns, golf courses and other turf areas where it is very difficult to control (Breeden and Brosnan 2009). It is listed as a serious turf weed on websites in a number of other southeastern states and researchers are looking for cost-effective control measures (Henry and Yelverton 2005).
Potential Environmental and Social Consequences
The dense growth habit smothers other low-growing plants and prevents recruitment of native woody species in Western Australia (Hussey et al. 2007).
Uncertainty
It is quite uncertain that Paspalum dilatatum will actually survive to NAPPFAST zone 6. It is certainly widespread in zone 7 in the United States, but if it could only survive to zone 7, its potential range in Canada would be limited to coastal and southwestern British Colombia.
Conclusion
Based on the outcome of this pest risk assessment, Paspalum dilatatum is likely to establish and become invasive in parts of Canada including southern and coastal British Colombia, and, possibly, southern Ontario and parts of the Maritime Provinces if it is introduced to these areas. This plant should be considered for regulation under Plant Protection Act and Seeds Act. It is recommended that the pest risk analysis process continue for this plant with the completion of a Risk Management Document.
Technical Issues for Consideration
There are 320 species of Paspalum in the warm regions of the world (Watson and Dallwitz 1992 onwards), so there will always be identification issues. However, dallis grass is identifiable by trained personnel.
Appendix 10B: Risk Management Considerations for Paspalum dilatatum (Dallis grass)
- Potential Mitigation Measures for Natural Means of Dispersal
- Potential Mitigation Measures for Intentional Introduction Pathways
- Potential Mitigation Measures for Non-intentional Introduction Pathways
Potential Mitigation Measures for Natural Means of Dispersal
According to Allison (2009) it is unlikely that natural dispersal will allow the species to enter Canada, as established populations are in the southern states.
Potential Mitigation Measures for Intentional Introduction Pathways
Plants for Planting excluding Seed
Paspalum dilatatum is not reported to occur in Canada, and no evidence was found that it is cultivated in Canada. Paspalum dilatatum, as an ornamental plant, is not available in Canada (CNLA 2009).
Previous imports
The CFIA requires a Permit to Import, with the scientific and common names, for all plants and propagative material from origins other than the continental U.S. (CFIA 1997; CFIA 2009). Some plants from the continental U.S. also require a Permit to Import as listed in CFIA (2009). The CFIA has not issued a Permit to Import for Paspalum dilatatum plants in the past three years (CFIA internal data).
Potential risk mitigation measures
Non-regulatory measures
Currently, no specific non-regulatory measures are required.
Regulatory measures
Regulate Paspalum dilatatum as a quarantine pest under the Plant Protection Act and add this species to the List of Pests Regulated by Canada.
- This will require that importers of material from regions other than the continental U.S. specify the scientific name when applying for a Permit to Import plant and propagative material;
- This will require that the scientific name be provided on the Phytosanitary Certificate for all plants and plant products exported to Canada from the continental U.S.
- Refuse to issue Permits to Import for plants of Paspalum dilatatum Footnote 1.
Trade Implications
As Paspalum dilatatum is mainly used for pasture and soil retention in tropical and subtropical regions and is not cultivated in or imported into Canada as plants or seed, the regulation of Paspalum dilatatum is not expected to have a significant impact on trade.
Cost-effectiveness and Feasibility
CFIA plant health programs are already in place to prevent the entry of prohibited plant materials. Measures under the Plant Protection Act are already in place for other pests regulated by Canada (CFIA 2009).
Seed
Previous imports
Seed of new crop species, such as field crops, can be imported without a Permit to Import or a Phytosanitary Certificate. There is no regulatory policy or application process in place to adequately control these imports.
Potential risk mitigation measures
Regulate Paspalum dilatatum as a Class 1 prohibited noxious weed under the Weed Seeds Order of the Seeds Act Footnote 2.
- This species meets the definition for a Class 1Footnote 3 species under the Weed Seeds Order.
- All imported and domestic seed lots must be free of prohibited noxious weed seeds. Imported seed lots require a certificate of analysis stating Paspalum dilatatum is absent from the seed lot before it can be imported.
Regulate as a quarantine pest under the Plant Protection Act. Add this species to the List of Pests Regulated by Canada(CFIA 2009) in order to:
- Prevent the importation, movement, and cultivation of this species in Canada. Currently, seed of many new crop species, such as field crops, can be imported without a Permit to Import or a Phytosanitary CertificateFootnote 4.
- Enable inspectors to take appropriate action for the purposes of eradicating the pest or preventing its spread Footnote 5.
Regulatory actions under the Plant Protection Act could include the following:
- Prohibit importation of Paspalum dilatatum seed and refuse to issue Permits to Import for seed of Paspalum dilatatum. The only exceptions would be for the importation of devitalized seed and the importation of seed or preserved specimens for scientific research purposes by recognized herbaria and research facilities Footnote 6.
Require importers of plant material from the non-continental U.S. to apply for a Permit to Import with scientific name specified.
- Seed of horticultural plants is not within the scope of CFIA's unpublished, draft directive on plants for planting (D-08-04). Therefore phytosanitary requirements must be specified either in a revision of D-08-04 (CFIA 2008) or in a new regulatory policy directive.
Sufficient information about the use of Paspalum dilatatum as a potential crop does not exist. If the proponent, located in Canada, needs to collect more information about the plant (e.g. to generate data for a determination of environmental safety), then confined research trials under Part V of the Seeds Regulations could be authorized by the Plant Biosafety Office.
Trade Implications
- The regulation of Paspalum dilatatum, is not expected to have a significant impact on trade.
- Regulation may facilitate seed trade with countries where Paspalum dilatatum is listed as a serious or principal weed (Australia, Philippines, Brazil, Columbia, the former Soviet Union and Taiwan (Holm et al. 1991))
Cost-effectiveness and Feasibility
The CFIA Seed Program is already in place to prevent the entry of prohibited noxious weeds.
Field Crops Not Intended for Propagation
Previous imports
Based on the information available in the CFIA's Import Permit System, two import permits have been issued, in 2003 and 2007, for various dried plant specimens from the genus Paspalum. Import permits were issued under section 43 and were for research purposes only.
Risk mitigation measures
Regulate Paspalum dilatatum as a quarantine pest under the Plant Protection Act and add this species to the List of Pests Regulated by Canada in order to:
- Prevent the importation, movement, and cultivation of this species in Canada.
- Enable inspectors to take appropriate action for the purposes of eradicating the pest or preventing its spread Footnote 7.
Regulatory requirements under the Plant Protection Act could include one or more of the following:
- A Permit to Import indicating specific import requirements and conditions for the handling and use of the commodity. It is the importer's responsibility to apply for and obtain the permit.
- End uses would have an impact on risk mitigation measures. For example, Paspalum dilatatum imported for milling (grinding) into flour would have lower risk than that coming in for direct use as pastured/rangeland animal feed. For milling, risk mitigation measures would include: proper disposal of anything removed from Paspalum dilatatum prior to the milling process and the milling itself. The same is true of Papalum dilatatum imported for processing into feed (pelleting, crushing, etc.). Papalum dilatatum imported for human consumption or direct feeding to livestock would have greater risk and would require treatment to render the grain unviable. Treatments, where applicable, such as heat (i.e. where heat would not compromise quality of grain for intended end use), grinding, pelletizing could be used prior to import or post-entry.
- All risk mitigation measures for Paspalum dilatatum must also take into consideration requirements/measures for pests other than plants (e.g. pathogens and insects).
Trade implications
As there is no identified usage of Paspalum dilatatum as as non-propagative product (food, ornamental, decorative) there will be no impact on trade.
Cost-effectiveness and Feasibility
CFIA plant health programs are already in place to prevent the entry of prohibited plant materials. Measures under the Plant Protection Act are already in place for other pests regulated by Canada (CFIA 2009).
Potential Mitigation Measures for Non-intentional Introduction Pathways
Field Crops Not Intended for Propagation
Previous imports
- The total value of grain commodities (corn, soybean, rice, canola, cereals) imported in 2008 from states where Paspalum dilatatum is present was over $1 billion (Industry Canada, 2009).
- Sixty-three percent of U.S. corn imported into Canada came from states where Paspalum dilatatum is present.
- The total value of soybeans imported from the U.S. in 2009 was $170.9 million. Approximately $5.5 million (3%) came from states where Paspalum dilatatum is present.
Potential risk mitigation measures
Regulate Paspalum dilatatum as a quarantine pest under the Plant Protection Act and add this species to the List of Pests Regulated by Canada (CFIA 2009) in order to:
- Prevent the importation, movement, and cultivation of this species in Canada.
- Enable inspectors to take appropriate action for the purposes of eradicating the pest or preventing its spread Footnote 8.
Regulatory actions under the Plant Protection Act could include one or more of the following:
- Exporters could be required to provide a Phytosanitary Certificate with or without an Additional Declaration stating freedom from Paspalum dilatatum.
- Recognition of Pest Free Areas – if Paspalum dilatatum can be shown to be absent in the country/state/region from which the field crop product was propagated, then risk is negligible and additional phytosanitary requirements may be waived.
- A Permit to Import indicating specific import requirements and conditions for the pest status, handling and use of the commodity could be required. It is the importer's responsibility to apply for and obtain the permit Footnote 9.
- Provisions for importation of grain contaminated with Paspalum dilatatum for research, processing, industrial or educational uses under a section 43 permit on a case-by-case basis.
- Phytosanitary measures may be waived for those commodities that have been treated or processed such that the risk of introduction of Paspalum dilatatum has been reduced to an acceptable level. End uses impact risk and may therefore impact required risk mitigation measures. For example, field crop commodities containing Paspalum dilatatum imported for milling (grinding) into flour would have lower risk than that coming in for direct use as pastured/rangeland animal feed. For milling, risk mitigation measures would include: the milling itself and proper disposal (incineration, grinding or deep burial) of anything removed from the commodity prior to the milling process. The same is true of field crop commodities imported for processing into feed (pelleting, crushing, etc.). Commodities imported for human consumption or direct feeding to livestock would have greater risk and could require treatment to render the grain unviable. Treatments, where applicable, such as heat (i.e. where heat would not compromise quality of grain for intended end use), grinding, pelletizing could be used prior to import or post-entry.
- All risk mitigation measures for field crop commodities containing Paspalum dilatatum must be taken with consideration for requirements/measures for pests other than plants (e.g. pathogens and insects).
Trade implications
- Not controlling import and spread of the species may result in infestations of field crops for export; market access for Canadian commodities could be compromised.
- Exporting countries may have to devote resources towards inspection and issuance of Phytosanitary Certificates. Laboratories in foreign countries will need to be able to identify seeds of Paspalum dilatatum. Exporters will need to ensure freedom of Paspalum dilatatum in field crop shipments, otherwise CFIA can refuse import.
Cost-effectiveness and Feasibility
- Measures under the Plant Protection Act are already in place for other pests regulated by Canada (CFIA 2009).
- The seeds of Paspalum dilatatum can be identified by trained analysts.
Seed
Previous imports
In 2008 the total value of seeds of forage plants imported from the U.S. was $6.68 million. Sixty-three percent of those imports ($4.23 million) came from states where Paspalum dilatatum is present (Industry Canada 2009). See appendix 10C. The probability of the weed seeds contaminating seed lots is unknown. The seed has not been reported as a contaminant in the marketplace of imported seed lots coming into Canada. There is risk associated with Paspalum dilatatum because it is commonly used as a forage crop, for pasture in cultivated subtropical areas, and it is also a serious weed in turf and golf courses in the southern states.
Potential risk mitigation measures
Regulate Paspalum dilatatum as a prohibited noxious weed (Class 1) under the Weed Seeds Order of the Seeds Act Footnote 10.
- All imported and domestic seed lots must be free of prohibited noxious weed seeds. Imported seed lots would require a certificate of analysis stating Paspalum dilatatum is absent from the seed lot before it can be imported
- This species meets the definitions for Class 1 species under the Weed Seeds Order.
Regulate Paspalum dilatatum as a quarantine pest under the Plant Protection Act and add this species to the List of Pests Regulated by Canada (CFIA 2009) in order to:
- Prevent the importation, movement, and cultivation of this species in Canada.
- Enable inspectors to take appropriate action for the purposes of eradicating the pest or preventing its spread Footnote 11.
Regulatory actions under the Plant Protection Act could include one or more of the following:
- Exporters could be required to obtain a Phytosanitary Certificate stating freedom from Paspalum dilatatum.
- Exceptions may be made for the importation of preserved specimens for scientific research purposes by recognized herbaria and research facilities and the importation of seed for research in containment facilities.
- Could negotiate phytosanitary agreements to certify imports from pest free areas and/or recognize noxious weed certification in countries or states of origin.
Trade implications
Exporting countries currently devote resources towards inspection of seed lots and issuance of Phytosanitary Certificates when required. Laboratories in foreign countries will need to be able to identify seeds of Paspalum dilatatum within a seed sample. Exporters need to ensure freedom of Paspalum dilatatum in seed lots, otherwise CFIA can refuse import.
Cost-effectiveness and Feasibility
- The CFIA Seed Program is already in place to prevent the entry of prohibited noxious weeds. The CFIA monitors compliance with the Canadian standards through the Marketplace Monitoring Program Footnote 12.
- There are 320 species of Paspalum in the warm regions of the world (Watson and Dallwitz, 1992 onwards), so there will always be identification issues. However, Paspalum dilatatum is identifiable by trained personnel. Training will be necessary for laboratory analysts.
Hay and Straw
Previous imports
- The total value of hay and straw imports was approximately $5.5 million in 2008; 98% of this value came from the U.S. (Industry Canada 2009).
- Between 2004 and 2008, about 8% of hay and straw imports came from states where Paspalum dilatatum is present.
- The level of risk associated with imports of hay and straw into Canada is relatively low as the volume of importation from states where Paspalum dilatatum is present is low.
Potential risk mitigation measures
Regulate Paspalum dilatatum under the Plant Protection Act as a quarantine pest by placing it on the List of Pests Regulated by Canada in order to:
- prevent the importation, movement, and cultivation of this species in Canada.
- enable inspectors to take appropriate action for the purposes of eradicating the pest or preventing its spread Footnote 13.
Regulatory actions under the Plant Protection Act could include:
- Require importers to obtain a Phytosanitary Certificate with or without an Additional Declaration stating freedom from Paspalum dilatatum.
- A Permit to Import indicating specific import requirements and conditions for the pest status, handling and use of the commodity could be required. It is the importer's responsibility to apply for and obtain the permit.
- For hay or straw intended for use as a biofuel stock, heat treatment or pelletization could be required.
Trade implications
- The impact of the obligation to obtain an Import Permit and Phytosanitary Certificate is expected to be low, as only 8% of imported hay and straw comes from states where Paspalum dilatatum is present.
- Exporting countries will need to devote resources towards inspection of hay and straw commodities and issuance of Phytosanitary Certificates. Inspectors in foreign countries need to be able to identify seeds of Paspalum dilatatum that could be associated with hay and straw. Exporters need to ensure freedom of Paspalum dilatatum in hay and straw, otherwise CFIA can refuse import.
Cost-effectiveness and Feasibility
This measure is already in place for other pests regulated by Canada. Minor costs will be incurred for identification training for inspectors.
Appendix 10C: Import Data forage seeds for sowing
| Year | States where Paspalum dilatatum is present ($) | Total US ($) | % of importation from states where Paspalum dilatatum is present |
|---|---|---|---|
| 2004 | 4,079,093 | 5,520,520 | 73,9% |
| 2005 | 3,990,010 | 5,453,038 | 73,2% |
| 2006 | 3,590,987 | 5,514,533 | 65,1% |
| 2007 | 3,358,093 | 5,340,241 | 62,9% |
| 2008 | 4,233,860 | 6,678,002 | 63,4% |
Source: Statistics Canada 2009
Appendix 11A: Pest Risk Assessment Summary for Persicaria perfoliata (mile-a-minute weed)
- Identity of Organism
- Organism Status
- Current Regulatory Status
- Probability of Entry
- Probability of Establishment
- Probability of Spread
- Potential Economic Consequences
- Potential Environmental and Social Consequences
- Uncertainty
- Conclusion of Weed Risk Assessment
Identity of Organism
Name: Persicaria perfoliata (mile-a-minute weed)
Synonym(s): Ampelygonum perfoliatum, Chylocalyx perfoliatus, Echinocaulon perfoliatum, Fagopyrum perfoliatum, Polygonum perfoliatum, Tracaulon perfoliatum, Truellum perfoliatum (Park 1986).
English common names: Mile-a-minute weed, devil's-tail tearthumb, giant climbing tearthumb, mile-a-minute weed, mile-a-minute-vine, minuteweed, tearthumb, Asiatic tearthumb (Hinds and Freeman 1993; USDA-ARS 2008; USDA-NRCS 2008).
French common names: None.
Mile-a-minute weed was formerly included in the large genus Polygonum, as P. perfoliatum. However, more recently most botanists in North America have agreed that this plant should be placed in the genus Persicaria (Hinds and Freeman 2005).
Persicaria perfoliata is a rapidly growing herbaceous annual vine of the buckwheat family (Polygonaceae). It is a distinctive plant with triangular leaves, backward-curved barbs on the stems and lower leaf blades, and metallic blue, berry-like fruits. Also characteristic of this plant are cup-shaped leafy structures called ocreae encircling the nodes (Okay et al. 1997).
Persicaria perfoliata is native to temperate and tropical Asia. In the United States, it is introduced and invasive in disturbed sites, hedges, and riparian sites of 13 states, including New York (Hinds and Freeman 1993). This aggressive plant forms tangled mats that smother native herbs, shrubs, and understory trees (Weber 2003).
Organism Status
Persicaria perfoliata is not reported to occur in Canada (CFIA 2008). Based on this information, for the PRA area, P. perfoliata is considered absent with no pest records.
Current Regulatory Status
Prior to this RMD decision, Persicaria perfoliata was not regulated in Canada. It is not currently regulated at the federal level in the United States, Mexico, Peru, Brazil, or China. It is currently regulated at the state level in Alabama, Connecticut, Massachusetts, North Carolina, Ohio, Pennsylvania and South Carolina (USDA-NRCS 2009).
Probability of Entry
The main pathways of entry into Canada are unintentional, human-mediated dispersal and natural dispersal (Table 1). Based on previous experience from the United States, the most likely human-mediated pathway is the unintentional movement of seed or plants associated with nursery stock. Additional potential human-mediated pathways include contaminated vehicles, equipment, hay, mulch, soil, shoes, clothing, baggage and ornamental seed. Intentional introduction is also possible but the plant has low ornamental value. Due to the presence of P. perfoliata in nearby states, such as New York and Pennsylvania, natural range expansion is another potential pathway of introduction for this species.
Probability of Establishment
It is native to cool temperate regions of eastern Asia, where it is widely distributed (Johnson Jr. 1996; Steward 1930). It is most prominent in Japan, Korea and parts of China (Johnson Jr. 1996). It extends as far south as the Philippines and as far west as India, where it is generally found in cooler or mountainous regions (Johnson Jr. 1996).
In addition to its native range, it was introduced and established in the United States. The current distribution of P. perfoliata in North America is illustrated in Figure 1 and Figure 2. It has also been detected in Türkiye (Güner 1984) and the Caribbean (Weber 2003) but its status (casual, naturalized, or invasive) is unknown. It was introduced into New Zealand, but has since been eradicated (EPPO 2008). In Canada, P. perfoliata was collected once along a roadside near Pitt Meadows, British Columbia in 1954, but did not persist (Douglas et al. 1990; Hinds and Freeman 1993).
It is considered to be hardy to United States Department of Agriculture's (USDA) Plant Hardiness Zone 6. Within Canada plant hardiness zones 6 to 8 are located along the coast of British Columbia and Vancouver Island, in extreme southern Ontario, and areas in the Maritime Provinces (Figure 3). Suitable habitats for P. perfoliata include a variety of disturbed areas and riparian areas. It prefers sunny, moist sites but can tolerate shady and dry sites as well. Within areas of climatic tolerance, availability of suitable habitats would not be a limiting factor for the establishment of P. perfoliata in Canada.
| Type of Introduction | Specific Pathways |
|---|---|
| Natural Means of Dispersal | Natural dispersal is reported to occur by water, birds, mammals and ants (Okay et al. 1997). Of these natural means of dispersal, birds are probably the most significant long-distant disperser, followed by water, since the fruits can remain buoyant for at least a week (Okay et al. 1997). Rate of spread in the U.S. has been approximately 500 km in several directions over 55 years (EPPO 2008), or approximately 9 km per year. Persicaria perfoliata is present in several states adjacent to Canada, including Ohio, Pennsylvania and New York. If the current trend continues, and includes northward expansion, the range of Persicaria perfoliata could expand into Canada within several decades. The probability of entry of Persicaria perfoliata into Canada by natural means of dispersal appears to be high. |
| Intentional Introduction | Intentional introduction is possible but the plant has low ornamental value. |
| Unintentional Introduction | Unintentional introduction pathways for Persicaria perfoliata include contaminated vehicles, equipment, hay, mulch, soil, shoes, clothing and baggage, seed (mainly ornamental) or grain contamination, and movement of seed or plants associated with nursery stock. The probability of entry of Persicaria perfoliata into Canada association with contaminated seed and/or nursery stock, which were the means of introduction of this species into the U.S. by unintentional means appears to be high, especially in |
Note: States shaded green have at least one occurrence point.
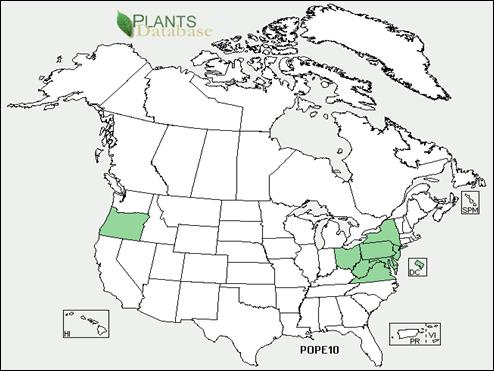
Description for Figure 1:
This map shows the range of Persicaria perfoliata in North America indicating its presence through the use of the colour green. The states that have at least one occurrence point are shaded green they are: Oregon, New York, Pennsylvania, Ohio, West Virginia, Virginia, Maryland, New Jersey, and Delaware.
Source: USDA-NRCS 2009.
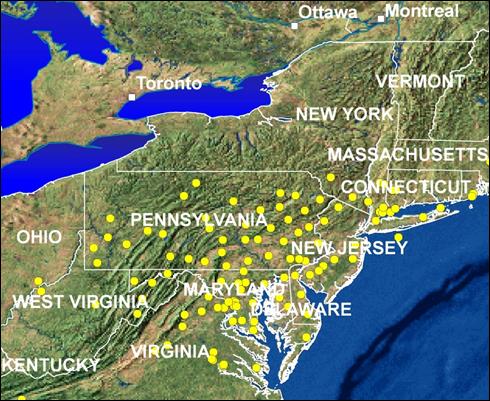
Description for Figure 2:
This map shows the northeastern region of North America demonstrating a county-level distribution Persicaria perfoliata (mile-a-minute weed). Yellow circles are used to distinguish the regions in which distribution is prevalent which is predominantly situated in the Connecticut, New Jersey, Pennsylvania, Maryland and Delaware regions. A scattered and minimal presence can be seen in New York and Massachusetts as well as Virginia and West Virginia.
Source: adapted from Hough-Goldstein et al. 2008.
Note: Mile-a-minute weed is considered to be hardy to USDA Plant Hardiness Zone 6. Within Canada, it may establish in shaded areas along the coast of British Columbia and Vancouver Island, in southern Ontario, and areas in the Maritime Provinces.
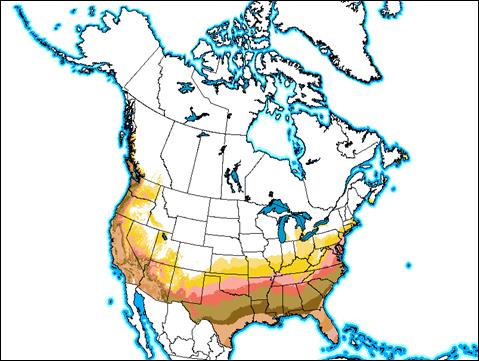
Description for Figure 3:
This is a map of North America showing the Plant Hardiness Zones 1 to 11 in different colours.
Probability of Spread
Persicaria perfoliata reproduces by seed. Individual plants can produce 50 to 100 seeds each. Vegetative growth is remarkably rapid, and can reach 15 cm per day. In addition to a variety of human-mediated means of dispersal, P. perfoliata is dispersed naturally by water, birds, ants, other small animals and deer. In the United States, it has spread rapidly over the past seven decades, and is now present in eleven states. The possibility of it spreading coast to coast has been raised. While some speculate that it could assume a perennial growth habit in Florida, other authors suggest the requirement of a cold stratification treatment for germination might limit the southward spread of this species. Overall, it has a moderate reproductive potential and a moderate to high seed mobility.
Potential Economic Consequences
Persicaria perfoliata is known to cause direct negative economic consequences on industries involved in the production of trees and shrubs, such as orchards, nurseries, Christmas tree plantations and regeneration sites. Plants germinate early and can quickly form a dense, prickly carpet of vegetation over seedlings and shrubs, causing defoliation, mechanical damage, and mortality. Potential indirect economic impacts include increased costs of control to a variety of sectors (e.g. transportation industry, parks and recreation, home gardeners), seed or grain contamination, decreased hay quality, and losses to the hunting and tourism industries.
Potential Environmental and Social Consequences
Persicaria perfoliata can cause reduced plant diversity by out competing and killing native understory plants including wildflowers, grasses, shrubs and saplings. Although P. perfoliata is often found in disturbed areas, it has the potential to cause species reductions in sensitive riparian areas as well. Furthermore, it may degrade food and habitat for wildlife, restrict wildlife movement, and reduce the aesthetic value of properties and public areas. The prickly vines, which can form dense thickets, are a nuisance to people, pets and wildlife.
Uncertainty
Areas of uncertainty that were identified in the risk assessment related to the potential for P. perfoliata to establish and spread in colder climates than it has previously, and the scarcity of quantitative information on the economic impacts of this species.
Conclusion of Weed Risk Assessment
Persicaria perfoliata is an aggressive invasive plant that can cause significant economic and environmental harm. Potential mitigation measures should be considered for this plant, recognizing that over time spread by natural means from the northeastern United States into Canada might occur.
Appendix 11B: Risk Management Considerations for Persicaria perfoliata (mile-a-minute weed)
- Pest Management
- Pest Risk Assessments from Other Countries
- Existing Domestic, Provincial or other Programs
- Canadian Christmas Tree Industry
- Potential Mitigation Measures for Natural Dispersal Pathways
- Recommended Mitigation Measures for Intentional and Unintentional Introduction Pathways
Pest Management
Persicaria perfoliata can be effectively controlled with herbicides either preemergence or postemergence (McCormick and Hartwig 1995). Hand-pulling, cultivation, and herbicides can control P. perfoliata on small scales; however, these methods are not feasible for large infestations or infestations in environmentally sensitive areas (Mountain 1989; Hough-Goldstein and Lake 2008; Price et al. 2003). Mechanical control is ineffective (Lehtonen 1994). The seed remains viable in the in the soil for six years, so managing P. perfoliata successfully depends on yearly treatments (Hough-Goldstein et al. 2008). A classical biological control program for P. perfoliata began in the U.S. in 1996 (Hough-Goldstein et al. 2008) using the weevil Rhinoncomimus latipes, a natural enemy introduced from China which is specific to the vine; with releases beginning in 2004 (Hough-Goldstein and Lake 2008; Hough-Goldstein 2008; Colpetzer et al. 2004).
Pest Risk Assessments from Other Countries
A pest risk assessment has been conducted by USDA-APHIS (Lehtonen 1994). The overall pest risk potential rating in the USDA-APHIS assessment was "high", which was based on "high" ratings for spread probability and environmental impact potential, and a "medium" rating for economic consequences of establishment. It was determined that the species no longer met the definition of noxious weed as required for listing under the U.S. Federal Noxious Weed Act. Recommendations included investigation of biological control for the species and cooperation in integrated management with other government agencies willing to share costs of control.
A Report of a Pest Risk Analysis on Polygonum perfoliatum is available from EPPO (EPPO 2007). The overall conclusion of pest risk assessment portion of the analysis was that the risk is unacceptable and management measures should be considered; it also reported that the plant is capable of causing significant damage to freshwater ecosystems and to commercial forest areas and forest regeneration. The report identified plants for planting with growing media, soil as a commodity, and imports of Meliosma seeds as the main introduction pathways.
New York State (U.S.) completed a non-native plant invasiveness ranking form for P. perfoliata concluding an invasiveness rank of "very high".
Existing Domestic, Provincial or other Programs
There are no existing Canadian programs related to the management or control of P. perfoliata . A Mile-A-Minute Control Management Plan has been developed by the Pennsylvania Department of Agriculture where the plant is a State Noxious Weed; it is a joint control program between the USDA-APHIS PPQ and the Pennsylvania Department of Agriculture (Bravo 2008). Persicaria perfoliata is considered widely distributed in the U.S. so does not meet the definition of a noxious weed as required for listing under the Federal Noxious Weed Act; eradication is no longer feasible (Lehtonen 1994). At the U.S. State level, it is listed as a Class A noxious weed in North Carolina, a prohibited noxious weed in Ohio, and a noxious weed in Pennsylvania and Alabama (Kumar and Ditommaso 2005).
Persicaria perfoliata can unintentionally contaminate hay (Castro 2008). Weed free forage programs can help prevent the spread of invasive plants. The Government of Alberta, Agriculture and Rural Development, administers the Alberta Certified Weed Free Hay Program. The objectives of the program are to provide a premium product that is recognized as marketable and transportable, to prevent the spread of restricted and noxious weeds, and to protect private and public lands from non-native, invasive plant species. Persicaria perfoliata is not currently designated as an undesirable plant on this list.
Canadian Christmas Tree Industry
Persicaria perfoliata is known to have deleterious economic consequences on the production of Christmas trees (Castro 2008). It grows rapidly and may out-compete Christmas tree seedlings. In 2007, Canada exported 2.5 million Christmas trees worth $29.8 million (Table 1). Areas in the Maritime Provinces that export Christmas trees, and that have suitable climate conditions for P. perfoliata (see Appendix 11A, Figure 3), are at risk if P. perfoliata establishes in Canada.
Potential Mitigation Measures for Natural Dispersal Pathways
Natural range expansion is a potential pathway of introduction. Persicaria perfoliata produces numerous fruits and has a high rate of seed set that contributes to natural dispersal (Oliver 1996). Once established, it can spread into surrounding habitats and watersheds by birds, animals and water (Lehtonen 1994). Birds and rodents disperse seeds as dorivers and streams, especially after flooding events (Mountain 1989; Gerlach Okay et. al 2007). Birds are also recognized as long-distance dispersal agents and ant species transport seed short distances (Kumar and Ditommaso 2005; Gerlach Okay et. al 2007). Seeds can pass through avian digestive tracts and remain viable (Okay 1997 in Kumar and Ditommaso 2005). Persicaria perfoliata seed dispersal and germination can also be facilitated by white-tailed deer (Odocoileus virginianus) (Hough-Goldstein et al. 2008). Once established in a new location, new populations can establish as P. perfoliata can reproduce without cross-pollination (Kumar and Ditommaso 2005).
Risk management of natural dispersal events is difficult. However, regulation will require surveys that would detect any new incursions established through natural dispersal pathways. Education and outreach programs, as well as pest identification training for inspectors, would also support the early detection and rapid response to new pest incursions that may result from natural dispersal events.
Table 2: Summary of Christmas trees exported by Canadian provinc and year
| Province | 1998 | 1999 | 2000 | 2001 | 2002 | 2003 | 2004 | 2005 | 2006 | 2007 |
|---|---|---|---|---|---|---|---|---|---|---|
| Nova Scotia | 9.4 | 9.0 | 9.6 | 11.0 | 11.1 | 9.9 | 10.8 | 10.9 | 10.6 | 8.5 |
| New Brunswick | 5.2 | 4.6 | 5.7 | 6.3 | 6.4 | 5.7 | 5.3 | 4.8 | 4.8 | 4.8 |
| Québec | 19.6 | 20.6 | 22.6 | 25.7 | 25.5 | 21.9 | 19.4 | 18.1 | 18.7 | 15.7 |
| Other provinces | 2.0 | 0.8 | 0.7 | 0.8 | 0.9 | 0.6 | 0.7 | 1.0 | 0.8 | 0.8 |
| Canada | 36.2 | 35.0 | 38.6 | 43.8 | 43.9 | 38.0 | 36.2 | 34.8 | 34.9 | 29.8 |
Source: AAFC 2008
Recommended Mitigation Measures for Intentional and Unintentional Introduction Pathways
Recommended risk mitigation measures
Regulatory measures under the Plant Protection Act
It is recommended that Canadaregulate P. perfloliata as a quarantine pest under the Plant Protection Act (1990, c.22)Footnote 1, and add this species to the List of Pests Regulated by Canada (CFIA 2009). This would prevent the intentional importation, movement, or cultivation of this species in Canada. The CFIA would not approve Permit to Import applications for P. perfoliata.
The CFIA would need to rescind any Permits to Import issued for Persicaria perfoliata. Any plants of Persicaria perfoliata established either outside of cultivation or in cultivation would need to be placed under official control.
To support regulation, a general requirement would be that importers would be required to provide full scientific names on all applications for Permits to Import as there are many species in the Persicaria genus.
Regulatory exemptions for the import of live or dried Persicaria perfoliata plant parts for special purposes such as research, food or medicine would be considered on a case by case basis by the CFIA.
Regulatory measures under the Seeds Act
It is recommended that Canada regulate P. perfoliata as a Class 1 Prohibited Noxious weed under the Weeds Seeds Order (SOR/2005-220) of the Seeds Act (R.S., 1985, c. S-8) Footnote 2. This species meets the definition for Class 1Footnote 3 species under the Weeds Seeds Order. All imported and domestic seed lots must be free of prohibited noxious weed seeds. Imported seed lots would require a certificate of analysis stating P.. perfoliata is absent from the seed lot before it can be imported.
The Weed Seeds Orderis a Ministerial Order made under the Seeds Act. The Weed Seeds Order allows the Minister of Agriculture and Agri-Food to regulate the seeds of the species of plants set out in the schedule that are deemed to be weed seeds for the purpose of establishing grades under the Seeds Act.
Non-regulatory measures
Education and awareness programs that foster awareness and support regulation of P. perfoliata are recommended. These would support regulatory measures, as well as prevention and early detection activities. Industry could also implement voluntary measures to end the sale and distribution of known invasive plants through mail order catalogs or internet sales.
Trade Implications
Plant Protection Act
The regulation of P. perfoliata under the Plant Protection Act will have trade impacts as commodities will need to be certified as free from the pest and subjected to new plant import requirements for import to Canada. International trade as a plant for planting is limited as it is not known to have any economic value in Canada and most countries recognize P. perfoliata as an invasive plant (Oliver 1996).
Seeds Act
The regulation of P. perfoliata under the Seeds Act will impact trade as exporting countries will need to devote resources towards inspection of seed lots and issuance of foreign Phytosanitary Certificates under the Plant Protection Act. Laboratories in exporting countries will need to be able to identify seeds of P. perfoliata within a seed sample. Exporters would need to ensure freedom of P. perfoliata in seed lots, otherwise the CFIA will refuse entry.
Cost-effectiveness and Feasibility
Additional resources would be required so that the CFIA could "trace-back" all existing permits issued for Persicariaspp. to determine if the species, P. perfoliata, has been intentionally imported. If it has been imported, an official control program would be required.
Resources would be required to expand the CFIA survey efforts. Minor costs may be incurred for pest identification training for inspectors and survey staff.
Resources would be required for an education and awareness program to foster awareness of P. perfoliata as an invasive plant and potential quarantine pest, targeting the general public, gardeners and the horticultural industry.
The CFIA currently requires import declarations and certificates of analysis for seed imported into CanadaFootnote 4. Additional resources required to regulate P. perfoliata via the seed pathway would be minimal.
Resources will be required to expand CFIA seed sampling efforts and minor costs would be incurred for seed identification training for inspectors. Seeds consist of spherical, shiny-black achenes, covered by a white or pink perianth, which becomes blue and fleshy when mature. They form blueberry-like "fruits", each 5 mm in diameter, arranged in clusters (Wu et al. 2002). Inspectors will need to be able to detect the seeds in any imported commodities.
The horticultural industry would need to be trained to recognize the seed and plants of P. perfoliata, as part of a preventive approach. The CFIA Seed Program is already in place to prevent the entry of prohibited noxious weeds. The CFIA monitors compliance with the Canadian standards through the Marketplace Monitoring ProgramFootnote 5.
Exporting countries will need to devote resources towards inspection of hay and straw commodities and for the issuance of Phytosanitary Certificates. Inspectors in foreign countries need to be able to identify seeds and plants of P. perfoliata that could attach themselves to hay and straw or that might become tangled up in the hay or straw with viable seeds attached. Exporting countries would need to ensure freedom of P. perfoliata in hay and straw, otherwise the CFIA will refuse entry.
Inspection staff at Canada Border Services Agency (CBSA) would need to be trained on how to identify P. perfoliata seed on vehicles or other equipment entering Canada. Inspections should focus on any vehicles or equipment entering Canada from infested north eastern U.S. States (see see Appendix 11A, Figure 2).
Implicated Pathways
Intentional Introduction Pathways
- Plants for Planting
- Plants for planting is recognized as a potential pathway of entry for P. perfoliata into Canada, however, the plant has low ornamental value (Castro 2008). Persicariaspp.have been imported intentionally into Canada. Between 2006 and 2009, the CFIA issued 25 Permits to Import for Persicariaspp., and another 11 Permits to Import permits between 2004 and 2005. However, it is not known if P. perfoliata has specifically been imported into Canada as species level data was not specified on the import permits. Permits to import Persicaria spp. have been issued to importers in Quebec (imports from the Netherlands, France, Japan, and United Kingdom), Ontario (imports from the Netherlands, United Kingdom, United States), Manitoba (imports from the Netherlands), Alberta (imports from the Netherlands) and British Columbia (imports from the Netherlands).
- Seed
- It is unlikely that importers would import pure lots of P. perfoliata seed. However, persons wishing to intentionally import the species may choose the seed pathway. Persicaria perfoliata could also enter Canada undetected in packets of ornamental seed, such as in wildflower mixes (Castro 2008).
Unintentional Introduction Pathways
- Movement of seed or plants associated with nursery stock
- A potential unintentional pathway is the importation of nursery stock or plants contaminated with seeds or plant material of P. perfoliata . The plant is well adapted for dispersal from nursery to nursery or nursery to planting-bed (Hill et al. 1981). Seeds are likely to survive along the nursery stock pathway (Castro 2008). Persicaria perfoliata seed or plants associated with nursery stock rooted in soil is a potential pathway only from the U.S. (Castro 2008). Introduction into Canada through contaminated seed commodities is a possibility (Castro 2008). Persicaria perfoliata has been reported to be unintentionally introduced by seed associated with Meliosma seed imported into the U.S. from China, and in Ilex seeds from Japan (Lehtonen 1994) as well as being reported as a contaminant from rhododendron nursery stock imported from eastern Asia (Hill et al. 1981). Seeds can remain viable for up to seven years and withstand a fairly wide range of temperatures (Castro 2008).
- It is not known if nursery stock contaminated by seed or plants of P. perfoliata has been imported into Canada. The Seed Science and Technology Section of the CFIA Saskatoon laboratory maintains a record of contaminants found in domestic and imported seed. Persicaria perfoliata has not previously been detected as a seed contaminant.
- Persicaria perfoliata seed may contaminate imported nursery stock present either in the soil or adhering to plants (e.g., Persicaria perfoliata can wrap around the nursery stock and fruits are known to remain on the plants long after the plant senesces) (Kumar and Di Tommaso 2005). The import of plants with soil and related matter is prohibited with the exception of shipments from the continental United States which require a Phytosanitary Certificate (see CFIA Policy Directive D-02-02 Plant protection import requirements for rooted, or unrooted plants, plant parts, and plants in vitrofor planting). Shipments of plants rooted in soil or related matter, or without soil, would be required to be free of P. perfoliata.
- Hay and Straw
- Hay is a potential pathway for the introduction of P. perfoliata , although there is little information about it in the literature (Castro 2008). The forage industry is economically significant in Canada (Table 2a & 2b). It is not known if P. perfoliata has been unintentionally imported into Canada as a contaminant in hay or straw (i.e., forage crops).
Summary of forage imports and exports into and out of Canada between 2003 and 2007
| Year | Quantity ('000) Tonnes |
Value ('000 Cdn$) |
|---|---|---|
| 2007 | 0.253 | 5,710 |
| 2006 | 0.268 | 5,723 |
| 2005 | 0.402 | 7,814 |
| 2004 | 0.457 | 8,349 |
| 2003 | 1.168 | 9,836 |
| Year | Quantity ('000) Tonnes |
Value ('000 Cdn$) |
|---|---|---|
| 2007 | 746 | 198,381 |
| 2006 | 677 | 172,826 |
| 2005 | 616 | 162,790 |
| 2004 | 679 | 171,515 |
| 2003 | 450 | 136,272 |
Source: Statistics Canada 2008
Vehicles and Equipment - Persicaria perfoliata may unintentionally be introduced into Canada as a contaminant on railroad cars, vehicles, and similar equipment (e.g. mowing equipment) entering Canada from infested areas. For example, P. perfoliata may have been introduced into Virginia (U.S.) as a contaminant of railroad cars or in mud on gas well drilling equipment (Lehtonen 1994). It can also be dispersed by logging equipment (McCormick and Hartwig 1995) and roadside mowers (Oliver 1996).
In 2003, CBSA assumed responsibility for the initial import inspection services in respect of the Acts and Regulations administered by the CFIA to the extent that they are applicable at Canadian border points. The inspections of goods that may be contaminated with soil are among the responsibilities that were transferred to the CBSA in 2003. The Food, Plant and Animals Programs Section of the CBSA is currently finalizing its Standard Operating Procedures (SOP) concerning the "Inspection of Imported Goods Potentially Contaminated with Soil." This SOP provides the CBSA's Border Services Officers with formal procedures for the inspection and disposition of goods that may be contaminated with soil, including used agricultural machinery and vehicles.
Regulatory measures would include enforcement of CFIA's Policy Directive 95-26 (CFIA 2008) that specifies the phytosanitary requirements for the import and domestic movement of soil and related matter. It includes requirements for soil and related matter individually or in association with plants, plant material, and other things such as vehicles, equipment, seed, hay, and containers. This directive also specifies the standards by which the Canadian Food Inspection Agency or the Canada Border Services Agency may inspect, certify or release these articles. If added to the List of Pests Regulated by Canada (CFIA 2009), things such as vehicles, equipment, and containers may be refused entry into Canada if contaminated with Persicaria perfoliata.
Appendix 12A: Pest Risk Assessment Summary for Senecio inaequidens (South African ragwort)
- Identity of Organism
- Organism Status
- Current Regulatory Status
- Probability of Entry
- Probability of Establishment
- Probability of Spread
- Potential Economic Consequences
- Potential Environmental and Social Consequences
- Uncertainty
- Conclusion
- Technical Issues for Consideration
Identity of Organism
Name: Senecio inaequidens (Asteraceae) (USDA-ARS 2009)
Synonyms: Senecio burchellii (CAB International 2007)
English common names: South African ragwort, narrow-leaved ragwort (CAB International 2007), fireweed, variable groundsel (Mondragón Pichardo and Vibrans 2005)
French common names: Séneçon du Cap (CAB International 2007)
Taxonomic note: There has been considerable taxonomic confusion regarding Senecio inaequidens, which is owed to morphological variation within the species and superficial similarity between two groups of species (Senecio madagascariensis complex and Senecio lautus complex) (CAB International 2007). Traditionally, a distinction has been made between Senecio inaequidens and Senecio madagascariensis, although recent research suggests that they are conspecific and differ in ploidy level only (CAB International 2007; Lafuma et al. 2003). It was found that a tetraploid form was introduced in Europe, and a diploid form in Australia, Argentina, Mexico and possibly the U.S. (Hawaii), although both forms originated in South Africa (Lafuma et al. 2003). The tetraploid form is referred to as Senecio inaequidens, and the diploid form may actually be what has been referred to as Senecio madagascariensis.
Overall, the distinction between Senecio inaequidens and Senecio madagascariensis is still 'controversial' (CAB International 2007). For the purposes of risk assessment, they are considered to be distinct taxa in accordance with the USDA Germplasm Resources Information Network (GRIN) (USDA-ARS 2009), although some of the references consulted treated the two together. It should be noted that this distinction between taxa may disappear in the future, which would require an update to the taxonomy presented in this risk assessment. At this time, since Senecio madagascariensis possesses many of the same invasive qualities as Senecio inaequidens (Lafuma et al. 2003), the plant health risk posed by the two taxa should probably be considered concurrently. Senecio inaequidens and Senecio madagascariensis are both listed on the U.S. Federal Noxious Weed List (USDA-APHIS 2006).
Description: Senecio inaequidens is a broadleaved, herbaceous short-lived perennial shrub that reaches 100 cm in height (CAB International 2007). It has erect stems that often branch from the base, bright green alternate leaves that usually clasp the stem, and yellow flowers composed of 7 to 13 female ray florets and numerous perfect disc florets (CAB International 2007). The seeds are achenes, 2 to 2.5 mm long, with a white pappus, 2 to 3 times as long (EPPO 2006a). The specific epithet inaequidens means unequal teeth, likely referring to variation in the degree of dissection and the width of the lobes of the leaves (CAB International 2007).
Organism Status
It is not reported to occur in Canada (CAB International 2007; CFIA 2008; Scoggan 1979), with the exception of Heger and Böhmer (2006), who refer to Garcia-Serrano et al. (2004). However, there is no statement in the latter reference (Garcia-Serrano et al. 2004) indicating that Senecio inaequidens is present in Canada, so the reference appears to have been used in error (possibly because it was published in the Canadian Journal of Botany). No evidence of its cultivation in Canada was found either (CNLA 2008; Isaacson and Allen 2007). Based on this information, for the PRA area, Senecio inaequidens is considered absent.
Current Regulatory Status
It is not currently regulated in Canada. It is regulated as a federal noxious weed in the US but not regulated in any of the states.Summary of pathways for Senecio inaequidens (South African ragwort). It is not currently regulated by any European country (EPPO 2006), but is on the EPPO List of Invasive Alien Plants and is considered to impose an important threat to plant health, the environment and biodiversity in the EPPO region. Senecio madagascariensis, a very closely related taxon, is listed as a noxious weed in Hawaii (HEAR 2003), Australia (i.e., New South Wales, Western Australia, Queensland and the Australian Capital Territory (Australian Weeds Committee 2008; EPPO 2006)) and in Mexico.
Probability of Entry
Although natural dispersal is an important pathway of introduction in its current range, natural dispersal is unlikely to be means of introduction for into Canada (Table 1). However, there are a number of potential unintentional pathways for introduction of Senecio inaequidens into Canada including wool imports, contaminated hay or grain, and as a generalist contaminant on travelers or on surfaces.
| Type of pathway | Specific Pathways |
|---|---|
| Natural dispersal |
|
| Intentional Introduction | None identified |
| Unintentional Introduction |
These unintentional pathways are low risk. |
Probability of Establishment
It native to southern Africa (Lesotho, South Africa, Swaziland, Mozambique, Botswana, and Namibia) and is naturalized elsewhere (CAB International 2007) (Figure 1 ). Its reported range is variable among sources due to the taxonomic confusion mentioned above. It is currently found in many European countries, from Spain to Poland, and including the northern countries of Norway and Sweden (CAB International 2007; Heger and Böhmer 2006). Although it has been reported in Finland, it does not appear to have established there (Heger and Böhmer 2006). It was also recently discovered in Taiwan (Jung et al. 2005). Reports of Senecio inaequidens from Mexico, Argentina (Figure 1), and possibly Colombia, may refer to S. madagascariensis (Lafuma et al. 2003).
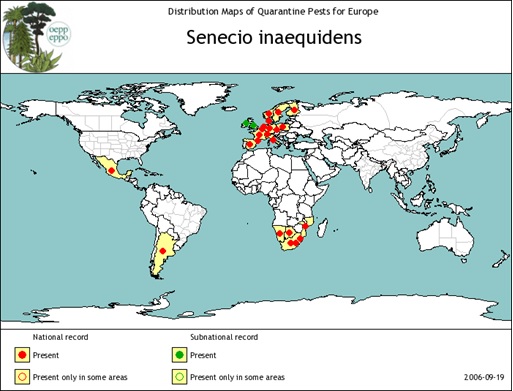
Description of Figure 1:
This image shows the distribution map of Senecio inaequidens (South African ragwort). You can see a map of the world under which there is a legend that is subdivided into two sections: national records and subnational records. Under national records there is a yellow square with a filled in red circle inside it symbolizing that records are present. Secondly, there is a yellow square inside which there is a red outline of a circle signifying that it is present only in some areas. Subnational record also has two symbols: a yellow square with a green filled in circle denoting present records on the subnational level and a yellow square with an outline of a green circle inside it signifying that they are only present in some areas. Southern Africa is covered in yellow containing filled in red circles in regions such as: Lesotho, South Africa, Swaziland, Mozambique, Botswana, and Namibia. Moreover, a great many European countries ranging from Spain to Poland extending to Norway and Sweden are covered in yellow while most contain complete red circles, Ireland and the United Kingdom have green full circles. Argentina and Mexico also contain complete red circles.
Source: EPPO 2008
In southern Africa, Senecio inaequidens grows in grasslands at high elevations (1400-2800 m), along the margins of periodically flooded streams, and in ruderal habitats (CAB International 2007). In Europe, this species establishes in a wide variety of habitats and on a wide range of soils, although it prefers warm, dry disturbed sites with well-drained soils (CAB International 2007). Habitats include railway lines, roadsides, the dividing strips of highways, river ports, flat roofs, flower tubs, logging areas, storm-damaged forests, burnt land industrial sites, disused quarries, rocky sites, coastal dunes, crops and pastures from sea level to approximately 600 m (EPPO 2006; Heger and Böhmer 2006).
There is potential for Senecio inaequidens to establish in Canada due to its opportunistic nature and its ability to establish in temperate climates. An EPPO datasheet on Senecio inaequidens describes it as hardy and well adapted to zone 7 (EPPO 2006) (likely referring to USDA Plant Hardiness Zone 7). Based on the results of the NAPPFAST System, it appears capable of establishing in zone 6 as well, based on records from Norway, Sweden (GBIF 2008) and Switzerland (Heger and Böhmer 2006). In Canada, zone 6 and above includes coastal British Columbia and Vancouver Island, a small area in extreme southern Ontario, and some of the coastal areas of Nova Scotia and Newfoundland (Figure 2).
Note: As predicted by the NAPPFAST system. The red shading corresponds to areas at risk which represent global plant hardiness zones 6 and above.
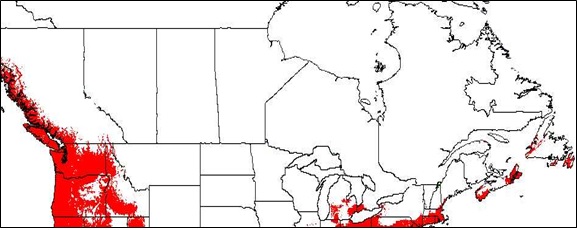
Description of Figure 2:
This image shows the potential range of Senecio inaequidens (South African ragwort) in Canada and northern America through the use of a map. Red is used to indicate the regions in which Senecio inaequidens could survive in according to the Canadian Plant Hardiness Zones map, in this case NAPPFAST Hardiness Zones 6. In Canada, the potential range of Senecio inaequidens includes coastal British Columbia and Vancouver Island, a small area in extreme southern Ontario, and some of the coastal areas of Nova Scotia and Newfoundland. Most of the red can be found in the U.S., this includes the greater parts of the east and west coasts as well as areas located near the Great Lakes extending downwards beyond the image.
Probability of Spread
It is a prolific seed producer that is dispersed naturally by wind and unintentionally by humans in association with clothes, shoes, soil movement, road and rail vehicles, building materials, hay, grain, ornamental plants, livestock, and wool (CAB International 2007; EPPO 2006; Heger and Böhmer 2006). In Europe, its initial introduction occurred in association with sheep wool imports and then it spread rapidly along linear anthropogenic structures, especially railway tracks and highways (Heger and Böhmer 2006). It grows vigorously and is readily adaptable to a wide range of environments (EPPO 2006).
Potential Economic Consequences
From various foci of introduction in Europe, Senecio inaequidens began to spread and by the 1970s was considered invasive in many European countries (EPPO 2006). It continues to expand its range today, and tends to form very high infestation levels (EPPO 2006). Senecio inaequidens is known to colonize vineyards and to reduce the value of pastures (EPPO 2006). The plants contain pyrrolizidine alkaloids, which are toxic to livestock and humans (EPPO 2006) and may affect consumer demand for milk and honey (EPPO 2006).
Control or eradication in Europe is considered very difficult and costly (EPPO 2006). Negative economic consequences associated with herbicide resistance of Senecio inaequidens have been reported from German railways (100,000 Euros per year) (Heger and Böhmer 2006; Reinhardt et al. 2003). The EPPO Pest Risk Analysis for this species indicates that economic risks for this species are medium to high (EPPO 2006). [Note that there is a discrepancy between this source and CAB International (2007), which considers economic impacts to be minimal.] The introduction of Senecio inaequidens in the PRA areacould also threaten Canada's ability to export commodities to the United States (where it is a designated federal noxious weed) and potentially elsewhere. The achenes are similar in size to those of Senecio vulgaris L. (common groundsel), a known contaminant of forages and grasses in Canada.
Potential Environmental and Social Consequences
It is known to invade natural habitats (e.g., dunes, cliffs, temporary ponds) and may threaten biodiversity (EPPO 2006). It is considered capable of modifying landscapes and is also an "colonizer of wasteland" (EPPO 2006). The EPPO Pest Risk Analysis for this species indicates that environmental risks for this species are medium to high and social risks are low to medium (EPPO 2006). Note again that there is a discrepancy between this source and CAB International (2007), which considers environmental impacts to be minimal.
Uncertainty
It may only be marginally hardy to NAPPFAST zone 6. It is certainly widespread in zone 7 in Europe, but if it could only survive to zone 7, its potential range in Canada would be limited to coastal and southwestern BC.
Although Senecio inaequidens is considered to have negative impacts on crops and pastures, information on the severity of these impacts, or on what crop species it invades (other than vineyards) is scarce.
Conclusion
The pest risk assessment concluded that Senecio inaequidens would likely become weedy or invasive in parts of Canada, including southern and coastal British Columbia, extreme southern Ontario and parts of the Maritimes, if introduced.
Technical Issues for Consideration
Senecio taxa may be distinguished based on shorter achenes (1.5 mm to 2.0 mm) and hairs confined to the achene grooves in Senecio madagascariensis as compared to longer achenes (2.5 mm) and completely hairy achenes in Senecio inaequidens. These differences should be treated with caution as they are based on limited sampling (CAB International 2007; Radford et al. 2000).
Appendix 12B: Risk Management Considerations for Senecio inaequidens (South African ragwort)
Values at Risk
- Canadian Livestock Industry:
- Senecio inaequidens contains 4 different pyrrolizidine alkaloids, two of which are known hepatotoxins (Dimande et al. 2007). These chemicals are poisonous to grazing animals, including cattle and sheep, which have resulted in death (Scherber 2007, Dimande et al. 2007). The total export value of cattle, sheep and horse products originating from the Canadian provinces where Senecio inaequidens is likely to invade was approximately $450 million in 2008 (Industry Canada 2009). In addition to directly affecting livestock, pastureland yields could decrease if the weed is introduced into Canada.
- Canadian Wine Industry:
- This species has the potential to colonize vineyards affecting the Lake Erie North Shore and Pelee Island wine regions of Ontario. In 2007, approximately $5 million worth of grapes Footnote 1 were produced in the counties of Ontario where this species could become established (OMAFRA 2009). The coastal wine-growing region of B.C. could also be affected by Senecio inaequidens affecting $1.6 million.
- Biological Diversity:
- There is evidence that Senecio inaequidens threatens biological diversity.
- Animal Health:
- The plant contains pyrrolizidine alkaloids which are toxic to livestock and humans and may affect consumer demand for milk and honey.
Pathways of Introduction
Natural Dispersal
The major pathways for the natural dispersal of Senecio inaequidens are wind, animals, and water. The risk of introduction into Canada from these natural dispersion mechanisms is low as there are no populations present near Canadian borders.
Intentional Introduction
There are no identified intentional introduction pathways for the entry.
Non-intentional Introduction
- Field crops not intended for propagation. The level of risk associated with the import of cereal grain into Canada is very low since imports originate primarily from areas where Senecio inaequidens is absent.
- Hay and Straw. The level of risk associated with imports of hay and straw into Canada is relatively low since they originate primarily from areas where Senecio inaequidens is absent.
- Livestock. The level of risk associated with the importation of livestock is low because of the small number of animals imported into Canada from countries where Senecio inaequidens is present.
- Vehicles and Used Farm Machinery. Information is not available on the volume of imports of used farm machinery. The lik lihood of Senecio inaequidens being introdued into Canada from seeds adhering to tires is low.
- Nursery Stock with Soil. Not present in United-States so it is not likely to be introduced to Canada through nursery stock with soil.
- Travelers and Their Effects. Travelers are not likely to bring Senecio inaequidens back into Canada unintentionally.
- Packaging Material. Container surfaces are a potential pathway of introduction for Senecio inaequidens. The achenes are more likely to attach to rough surfaces (e.g.. wood) rather than smooth ones, due to the nature of the seed. This pathway is considered low risk.
Appendix 12C: Assessment of Measures for Senecio inaequidens (South African ragwort)
- Field Crops Not Intended for Propagation
- Hay and Straw
- Livestock
- Vehicles and Used Farm Machinery
- Nursery Stock with Soil
- Travelers and Their Effects
- Packaging Material
Field Crops Not Intended for Propagation
Previous imports
- The total value of cereal grain commodities (wheat and meslin, rye, barley, oats, maize, buckwheat, millet, and other unmilled cereals) imported in 2008 was approximately $568 million or 11.6 million metric tonnes. Of this amount, 0.5% (approximately $2.9 million or 255 thousand tonnes; Table 2 ) of imports originated from countries where Senecio inaequidens is present (Statistics Canada 2009).
- The level of risk associated with the import of grain into Canada is unknown due to the lack of information available regarding grain as an introduction pathway. However, the amount of grain imports from countries where Senecio inaequidens is present is small in comparison to total grain imports (0.5%) and most imported grain is destined for consumption. Based on this information, grain as a pathway of introduction for Senecio inaequidens is low risk.
Import data for cereal grains originating from countries where Senecio inaequidens (South African ragwort) is present
Value in Canadian Dollars ($)
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| Argentina | 0 | 0 | 250,050 |
| Netherlands | 0 | 0 | 1,765 |
| United Kingdom (U.K.) | 457 | 27,372 | 212 |
| Germany | 483 | 4,383 | 90 |
| Denmark, Italy, South Africa, France | 46 | 330 | 231 |
| Subtotal: | 986 | 32,085 | 252,348 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| Germany | 0 | 9 | 2,050,723 |
| Poland | 18 | 364 | 0 |
| Subtotal: | 18 | 373 | 2,050,723 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| Switzerland | 0 | 0 | 1,118 |
| United Kingdom | 1,376 | 143 | 9 |
| Finland, Netherlands, Sweden, Poland, Germany, Italy | 704 | 1,095 | 928 |
| Subtotal: | 2,080 | 1,238 | 2,055 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| Switzerland, Poland, Sweden, South Africa | 161 | 412 | 332 |
| United Kingdom | 9,842 | 24 | 149 |
| Subtotal: | 10,003 | 436 | 481 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| Argentina | 2,982 | 96,519 | 2,562 |
| Netherlands, U.K., Poland, Finland, South Africa | 419 | 12 | 234 |
| Spain | 0 | 22,156 | 0 |
| France | 46,000 | 0 | 0 |
| Subtotal: | 49,401 | 118,687 | 2,796 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| Switzerland | 18,739 | 289,206 | 291,267 |
| Netherlands | 40,728 | 320,144 | 227,418 |
| Poland | 11,370 | 20,133 | 49,375 |
| South Africa | 0 | 0 | 24,311 |
| France | 64,279 | 31,242 | 23,686 |
| Italy | 7,116 | 15,760 | 6,877 |
| United Kingdom | 781 | 5,502 | 5,253 |
| Germany | 2,858 | 19,220 | 52 |
| Spain, Argentina, Hungary | 475 | 1,227 | 0 |
| Austria | 47,221 | 0 | 0 |
| Finland | 3,706 | 0 | 0 |
| Subtotal: | 197,273 | 702,434 | 628,239 |
| All Exporting Countries | 2006 | 2007 | 2008 |
|---|---|---|---|
| Total: | 259,761 | 855,253 | 2,936,642 |
Quantity (metric tonnes, TNE)
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| Argentina | 0 | 0 | 320 |
| Netherlands | 0 | 0 | 4 |
| United Kingdom (U.K.) | 0 | 17 | 0 |
| Germany | 0 | 3 | 0 |
| Denmark, Italy, South Africa, France | 0 | 0 | 0 |
| Subtotal: | 0 | 20 | 324 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| Germany | 0 | 0 | 4,015 |
| Poland | 0 | 3 | 0 |
| Subtotal: | 0 | 3 | 4,015 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| Switzerland | 0 | 0 | 1 |
| United Kingdom | 7 | 0 | 0 |
| Finland, Netherlands, Sweden, Poland, Germany, Italy | 1 | 0 | 3 |
| Subtotal: | 8 | 0 | 4 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| Switzerland, Poland, Sweden, South Africa | 1 | 2 | 2 |
| United Kingdom | 20 | 1 | 0 |
| Subtotal: | 21 | 3 | 2 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| Argentina | 42 | 329 | 119 |
| Netherlands, U.K., Poland, Finland, South Africa | 2 | 0 | 144 |
| Spain | 0 | 119 | 0 |
| France | 3,878 | 89 | 365 |
| Subtotal: | 3,922 | 537 | 628 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| Switzerland | 3,788 | 69,330 | 57,456 |
| Netherlands | 21,248 | 221,320 | 141,553 |
| Poland | 6,066 | 8,176 | 15,984 |
| South Africa | 0 | 0 | 21,500 |
| France | 17,351 | 8,604 | 6,460 |
| Italy | 2,329 | 4,595 | 3,149 |
| United Kingdom | 487 | 558 | 4,668 |
| Germany | 1,070 | 25,537 | 31 |
| Spain, Argentina, Hungary | 364 | 87 | 0 |
| Austria | 63,170 | 0 | 0 |
| Finland | 22 | 0 | 0 |
| Subtotal: | 115,895 | 338,207 | 250,801 |
| All Exporting Countries | 2006 | 2007 | 2008 |
|---|---|---|---|
| Total: | 119,846 | 338,770 | 255,774 |
Note: Countries with an export value of less than $1,000 per year are grouped together and a summed value for all countries is shown.
Potential risk mitigation measures
Regulate Senecio inaequidens as a quarantine pest under the Plant Protection Act and add this species to the List of Pests Regulated by Canada (CFIA 2009) in order to:
- prevent the importation, movement, and cultivation of this species in Canada.
- enable inspectors to take appropriate action for the purposes of eradicating the pest or preventing its spread Footnote 1.
Regulatory actions under the Plant Protection Act could include one or more of the following:
- exporters could be required to provide a Phytosanitary Certificate with or without an Additional Declaration stating freedom from Senecio inaequidens.
- requirement for a Permit to Import issued by the CFIA indicating specific import requirements and conditions for the pest status, handling and use of the commodity. It is the importer's responsibility to apply for and procure the permit.
- provisions for importation of grain contaminated with Senecio inaequidens for research, processing, industrial or educational uses under a section 43 permit on a case-by-case basis.
- recognition of Pest Free Areas – if Senecio inaequidens can be shown to be absent from the country/state/region from which the field crop product was propagated, then the risk is negligible and additional phytosanitary requirements may be waived.
Note: End uses impact risk and may therefore impact required risk mitigation measures. Regulatory measures may be waived for those commodities that have been treated or processed such that the risk of introduction of Senecio inaequidens has been reduced to an acceptable level.
All risk mitigation measures for field crop commodities containing Senecio inaequidens must be taken with consideration for requirements/measures for pests other than plants (e.g.. pathogens and insects).
Trade implications
- since a small proportion of grain imports are originating from areas where S. inaequidens is present, there will be minimal reduction and or loss of import markets if exporting countries are unable to meet proposed phytosanitary requirements.
- exporting countries may have to devote resources toward the inspection of grain commodities and the issuance of Phytosanitary Certificates.
- not controlling import and spread of Senecio inaequidens may result in infestations of field crops for export; if the importing country prohibits this species, market access for Canadian commodities could be compromised. There is also the potential for additional costs to Canadian producers for weed control in the field and cleaning of grain.
- regulation under the Plant Protection Act will safeguard cereal grain trade with the U.S., where Senecio inaequidens is regulated as a federal noxious weed. Canada exported approximately $1.2 billion worth of cereal grain to the U.S. in 2008 (Industry Canada 2009).
Cost-effectiveness and Feasibility
- resources will be needed by CFIA for marketplace monitoring and sampling, inspector training, compliance, enforcement and communication material development.
- grain contaminated with Senecio inaequidens could be cleaned to remove the contaminant seeds. Senecio inaequidens seeds are identifiable, but may not be readily screened from grain that is similar in size to its seeds (2.5mm long and 0.8mm wide; CFIA Seed Laboratory). This should be done by the exporting country. Field inspections or laboratory testing could be used to ensure freedom from species of quarantine concern.
Hay and Straw
Previous imports
- the total value of hay and straw imports was approximately $13 million in 2008; 98% of this value came from the U.S. Approximately $3,400 in total hay and straw imports came from countries where Senecio inaequidens is present (Table 3; Industry Canada 2009).
- between 1999 and 2004, quantities of hay and straw imported from countries where Senecio inaequidens is present ranged from $20,000 per year to $180,000 per year (Industry Canada 2009).
- between 1999 and 2004, high quantities of hay and straw (an approximate average value of $15.3 million per year) were imported from Washington and Oregon, where Senecio inaequidens is not present. This represents the majority of hay and straw imported into Canada. Since 2004, imports of hay and straw have remained relatively constant (Industry Canada 2009).
- the level of risk associated with imports of hay and straw into Canada is relatively low since they originate primarily from areas where Senecio inaequidens is absent.
Import data for hay and straw originating from countries where Senecio inaequidens is present
Value in Canadian Dollars ($)
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| Germany | 3,079 | 2,453 | 3,013 |
| United Kingdom | 2,518 | 8,773 | 408 |
| Subtotal: | 5,597 | 11,226 | 3,421 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| Germany | 60 | 3,000 | 0 |
| France | 19 | 2014 | 0 |
| Subtotal: | 79 | 5,014 | 0 |
| All Exporting Countries | 2006 | 2007 | 2008 |
|---|---|---|---|
| Total: | $ 5,676 | $ 16,240 | $ 3,421 |
Source: Industry Canada 2009
Potential risk mitigation measures
Regulate Senecio inaequidens under the Plant Protection Act as a quarantine pest by placing it on the List of Pests Regulated by Canada in order to:
- prevent the importation, movement, and cultivation of this species in Canada.
- enable inspectors to take appropriate action for the purposes of eradicating the pest or preventing its spread Footnote 2.
Regulatory actions under the Plant Protection Act could include one or more of the following:
- requirement for exporters to provide a Phytosanitary Certificate with or without an Additional Declaration stating freedom from Senecio inaequidens Requirement for a Permit to Import indicating specific import requirements and conditions for the pest status, handling and use of the commodity could be required. It is the importer's responsibility to apply for and obtain the permit.
- recognition of Pest Free Areas – if Senecio inaequidens can be shown to be absent from the country/state/region where the hay or straw was produced, then risk is negligible and additional phytosanitary requirements may be waived.
- requirement for heat treatment or pelletization for hay or straw intended for use as a biofuel stock.
Trade implications
- regulation of Senecio inaequidens under the Plant Protection Act will potentially safeguard trade of hay and straw with the U.S., where Senecio inaequidens is regulated as a federal noxious weed. In 2008, British Columbia, Ontario, Nova Scotia and Newfoundland exported approximately $15 million worth of hay and straw to the U.S. (Industry Canada 2009).
- exporting countries will need to devote resources towards inspection of hay and straw commodities and issuance of Phytosanitary Certificates. Inspectors in exporting countries need to be able to identify achenes of Senecio inaequidens that could attach themselves to hay and straw, or conduct pre-harvest field inspections. Exporters need to ensure freedom of Senecio inaequidens in hay and straw, otherwise CFIA can refuse import.
- since 98% of hay and straw imports come from the U.S., and a low value of imports come from areas where Senecio inaequidens is invasive, there will not be significant market losses of this commodity if it is found to be contaminated.
Cost-effectiveness and Feasibility
- minor costs will be incurred for identification training for inspectors.
- resources will be needed by the CFIA for marketplace monitoring, compliance and enforcement.
Livestock
Previous imports
- since 2004, 1,116 sheep, goats, bovine animals and horses for reproduction were imported from countries where Senecio inaequidensis present (CFIA, internal data). This represents 0.7% of all livestock imports (for reproduction) that have been imported into Canada since 2004.
- the level of risk associated with the importation of livestock is low because of the small number of animals (for reproduction purposes) coming into Canada from countries where Senecio inaequidens is present.
Potential risk mitigation measures
No mitigation measures are required due to the low number of animals entering Canada that could act as an introduction pathway for Senecio inaequidens.
Previous imports
- the total value of sheep and goat raw wool imports was approximately $3 million in 2008; 7% of this value ($220,000) came from countries where Senecio inaequidens is present (Industry Canada 2009).
- between 1999 and 2004, a total of $2.4 million worth of raw wool was imported from countries where Senecio inaequidensis present (Industry Canada 2009).
Potential risk mitigation measures
No measure is required:
- vegetative matter is removed from commercial wool by carbonization Footnote 3.
- raw wool and raw hides imported into Canada from all countries require CFIA inspection. They must be free from feces, blood, ectoparasites and dirt (plant and plant parts are considered to be dirt) or they will be refused entry (Health of Animals Act, Directive AHPD-DSAE-2001-1-1 (CFIA 1990)).
Vehicles and Used Farm Machinery
Previous imports
- information is not available on the volume of imports of used farm machinery
- the likelihood of Senecio inaequidens being introdued into Canada from seeds adhering to tires is low (Castro 2009)
Potential Risk mitigation measures
Enforcement of the Directive 95-26: "Phytosanitary requirements for soil and related matter, alone or in association with plants" (CFIA 2008).
- in 2003, the Canada Border Services Agency (CBSA) assumed responsibility for the initial import inspection services in respect to the Acts and Regulations administered by the CFIA to the extent that they are applicable at Canadian border points. The inspection of goods that may be contaminated with soil are among the responsibilities that were transferred to the CBSA in 2003. The Food, Plant and Animals Programs Section of the CBSA has developed its Standard Operating Procedures (SOP) concerning the "Inspection of Imported Goods Potentially Contaminated with Soil." This SOP provides the CBSA's Border Services Officers with formal procedures for the inspection and disposition of goods that may be contaminated with soil, including used agricultural machinery and vehicles.
Nursery Stock with Soil
Plants imported from off-continent sources must be free of soil as specified in directive D-02-02: Plant Protection Import Requirements for Rooted, or Unrooted Plants, Plant Parts, and Tissue Cultured Plants for Propagation (CFIA 2006). Soil can be imported with plants from the continental U.S., but since Senecio inaequidens is not present in that country, it is not likely to be introduced to Canada through nursery stock with soil; therefore, no mitigation measures are required.
Travelers and Their Effects
Previous imports
- a considerable number of people cross the Canadian border every year. Around 3 million people traveled to Canada and around 5.4 million Canadians traveled abroad in the 4th quarter of 2008 alone. The majority of this travel was to and from the U.S. (Statistics Canada 2009). Europe was the second most popular destination visited by Canadians in 2007 (Statistics Canada 2008)
- travelers are not likely to bring Senecio inaequidens back into Canada unintentionally. The risk is associated with the achenes attaching to the clothes, equipment and other effects of returning Canadians. The level of risk associated with this pathway is considered low (Castro 2009)
Risk mitigation measures
Increase public awareness of the regulation and the risk posed by Senecio inaequidens and distribute awareness material at border crossings to the public. It is difficult to target travelers for one particular species that could be unintentionally imported. Thus, a general information and awareness campaign about the risks associated with several species may be the best option. While not considered effective by itself if the risk is high, increased public awareness is a viable action if the level of risk is low.
Trade implications
None anticipated.
Cost-effectiveness and Feasibility
- resources will have to be allocated for promotional materials to be distributed at border crossings.
- public awareness campaigns could be an effective way to lower the risk of introduction of Senecio inaequidens into the country. This campaign could also be done in conjunction with other weed species to reduce costs and to inform the public on a more general level about invasive alien plants.
Packaging Material
Previous Imports
- a considerable volume of imported products are shipped in wood packaging material, including dunnage, pallets, spacers, bearers and crating.
- container surfaces are listed as a potential pathway of introduction for Senecio inaequidens. The achenes are more likely to attach to rough surfaces (e.g. wood) rather than smooth ones, due to the nature of the pappus (K. Castro, personal communication). This pathway is considered low risk (Castro 2009).
Risk Mitigation Measures
- wood packaging material is regulated under directive D-98-08: Entry Requirements for Wood Packaging Materials Produced in All Areas Other Than the Continental United States (CFIA 2008). All non-manufactured wood packaging material is permitted for entry into Canada only if it has been officially treated by an approved method (i.e. heat treatment or fumigation using methyl bromide) and if it is affixed with an official seal from the NPPO of the originating country.
- heat treatment or fumigation may make plant material inviable, although species-specific studies need to be conducted in order to determine if the seeds of Senecio inaequidens survive the treatments or not. Fumigation may not be the most effective option, as its efficacy decreases in dry environments (Bond 1984).
- if the above treatments do not effectively make Senecio inaequidens seeds inviable, modifications to directive D-98-08 should be made to include further treatment options that will eliminate viable plant material (e.g. increase the temperature of heat treatment). For Senecio vulgaris, previous studies have shown that a heat treatment of 75& deg;C for 0.5+ days resulted in 0% germination of seeds (Thompson et al. 1997). Additional research into the temperature tolerance of Senecio inaequidens seed is required; the temperature required to kill seeds is species-specific.
Trade Implications
No additional trade implications since countries that do not comply with the requirements set out in D-98-08 may have goods refused at the Canadian border.
Cost-effectiveness and Feasibility
- directive D-98-08 is in place to prevent the entry of other quarantine pests such as emerald ash borer (Agrilus planipennis).
- revising the directive and notifying inspection staff of the change will require minimal resources on the CFIA's behalf.
Appendix 13A: Pest Risk Assessment Summary for Senecio madagascariensis (Madacascar ragwort)
- Identity of Organism
- Organism Status
- Current Regulatory Status
- Probability of Entry
- Probability of Establishment
- Probability of Spread
- Potential Economic Consequences
- Potential Environmental and Social Consequences
- Uncertainty
- Conclusion
- Technical Issues for Consideration
Identity of Organism
Name: Senecio madagascariensis (Asteraceae) (USDA-ARS 2009)
Synonyms: Senecio incognitus Cabrera (USDA-ARS 2009)
English common names: Fireweed, Madagascar ragwort (USDA-ARS 2009), variable groundsel (Starr et al. 1999)
French common names: Séneçon de Madagascar
Taxonomic note: As noted in PRA 2009-20 sein (Senecio inaequidens DC.), the distinction between Senecio inaequidens (2n=40) and Senecio madagascariensis (2n=20) is controversial (International 2007; Lafuma et al. 2003; López et al. 2008). For the purposes of this risk assessment they are considered to be distinct taxa in accordance with the USDA Germplasm Resources Information Network (USDA-ARS 2009). Detailed investigation of populations from the exotic ranges of these species, together with several other closely related Senecio species from southern Africa is needed to understand the genetics, systematics and morphological variation of Senecio madagascariensis (CAB International 2007).
Description: It is a broadleaved, herbaceous, short-lived, branched perennial with golden yellow flowers (CAB International 2007; Starr et al. 1999). The plant has erect stems that reach 60 cm (CAB International 2007). Its leaves are bright green, alternate, occasionally petiolate and variable, reaching 12 cm long and 2.5 cm wide (CAB International 2007). Plants produce 2 to 200 small daisy-like flowers per plant, which appear at the branch tips (Starr et al. 1999). Seeds are achenes measuring approximately one to three mm long (Starr et al. 1999). Each has a white pappus, with hairs two to three times as long as the achenes (CAB International 2007; Starr et al. 1999).
Organism Status
It is not reported to occur in Canada, and no evidence was found that it has been cultivated in Canada (CAB International 2007; CFIA 2008; CNLA 2008; Isaacson and Allen 2007; Scoggan 1979). Based on this information Senecio madagascariensis is considered to be absent in Canada.
Current Regulatory Status
It is not currently regulated in Canada. It is regulated as a Federal Noxious Weed in the United States (USDA-APHIS 2006) and at the state level in Hawaii (HEAR 2003). It is also listed as a noxious weed in Australia, in New South Wales, Western Australia, Queensland and the Australian Capital Territory (Australian Weeds Committee 2008) and in Japan (Government of Japan 2006).
Probability of Entry
Potential pathways for introduction of Senecio madagascariensis into the PRA area are primarily unintentional pathways (Table 1). There is a moderate risk of this species entering the PRA area on travelers, on their clothing, shoes, or personal effects. Additional, minor pathways include imports of grass or pasture seed for planting and imports of livestock from areas where this plant is present. From the year 2000 to present, Senecio madagascariensis has not been detected in samples analysed by the CFIA Seed Lab.
| Type of pathway | Specific Pathways |
|---|---|
| Natural dispersal | Wind is considered to be the major factor for dispersal of seeds over large areas and long distances (i.e. seeds are light and spread easy by wind) The risk of natural dispersal into Canada is low because there are no populations near Canadian borders. |
| Intentional Introduction | None identified |
| Unintentional Introduction |
Contaminated shoes, clothing and personal effects of travellers may be the most likely of these pathways due to the high frequency of human travel between Canada and Hawaii, Australia, Japan, Colombia, Argentina and southern Africa. The remaining pathways are low risk. |
Probability of Establishment
Senecio madagascariensis is native to South Africa (Cape Province, Natal, Transvaal), Swaziland (USDA, ARS, 2009), Mozambique and Madagascar (CAB International 2007) (Figure 1). It is introduced in Kenya, Mauritius, U.S. (Hawaii), Argentina (Buenos Aires, Santa Fe, Entre Rios, Corrientes and Mendoza), Colombia, Australia (New South Wales, Queensland, and Victoria) (CAB International 2007) and Japan (Koike et al. 2006).
Note: a red dot indicates widespread; yellow indicates present but no further details; white indicates present and localized; green indicates present with additional regional information available (CAB International 2007).

Description of Figure 1:
This image is a distribution map of Senecio madagascariensis (Madagascar ragwort) in the world. You can see a map of the world which contains red dots signifying Senecio madagascariensis is widespread, yellow dots indicating that it is present but with no further details, white dots denoting a presence that is localized and green dots signifying that it is present with additional regional information available. South America has two whites dots located in Columbia and Argentina. At the centre of Australia there is a green dot. Asia has a yellow dot in the vicinity of Japan while another yellow dot can be seen in the Hawaiian Islands of North America. Lastly and most pervasively, there are yellow, white and red dots scattered along the lower half of the continent of Africa more specifically in Kenya which has a yellow dot as well as Swaziland and Mauritius which has multiple; Madagascar has a red dot and Mozambique has white one.
Source: CAB International 2007.
In general, this species is opportunistic and able to colonize a wide range of habitats, "from low-elevation arid pastures and coastal plains to higher-elevation, moist pastures, yards, fields and roadsides" (Starr et al. 1999). It often occurs in overgrazed or neglected pastures as well as cultivated or disturbed land (Starr et al. 1999). Although it has been found in both shady and sunny sites, it does not perform well in pastures over 1 m tall (CAB International 2007). Both high and low fertility soils, as well as a range of soil textures, are suitable for its growth. It is tolerant of drought (Plant and Robertson 2007) but does not survive in poorly drained or waterlogged soils (Starr et al. 1999; Watson et al. 1997). Senecio madagascariensis appears to be most successful in humid maritime sub-tropical climates and tends to be restricted to climates with low frost incidence (CAB International 2007; Sindel and Michael 1992). Frost may kill seedlings and reduce plant vigor (Sindel and Michael 1992; Starr et al. 1999).
Based on its current distribution and the results of the North Carolina State APHIS Plant Pest Forecasting (NAPPFAST) System it may adapt to plant hardiness zones 8 and higher. In Canada, zones 8 and above are restricted to coastal British Columbia and Vancouver Island (Figure 2). Within the area at risk, the dominant biogeoclimatic zone is coastal western hemlock, and there are small areas of coastal Douglas-fir as well (BC Ministry of Forests 1999). While unlikely to establish in mature forests, Senecio madagascariensis may be capable of establishing in the warm, humid disturbed areas of these biogeoclimatic zones.

Description of Figure 2:
This image shows the potential distribution of Senecio madagascariensis (Madagascar ragwort) in Canada, more specifically in British Columbia. Red is used to indicate the regions in which Senecio inaequidens could survive according to the Canadian Plant Hardiness Zones map, in this case NAPPFAST Hardiness Zones 8. In Canada, the potential range of Senecio madagascariensis is very limited to include only coastal British Columbia where it has a scattered presence. This image is narrowed to include parts of the U.S. more specifically Washington and Oregon where the red has more of a prevalent presence and is heavily condensed.
Probability of Spread
It is a highly adaptable species with an ability to grow vigorously and spread rapidly after introduction. Seeds are small and light (Plant and Robertson 2007). Seed production, which is extended over a period of weeks, is prolific (100 to 150 seeds per flower, 25,000 to 30,000 per plant) and germination rates are high (Starr et al. 1999). Light infestations can result in 1,000,000 seeds per hectare (Queensland Government 2007). Seeds are capable of germinating immediately after release, and quickly cover areas recently disturbed by fire or cultivation before more desirable species have a chance to emerge (Plant and Robertson 2007). Once present in an area, the species is easily dispersed by wind and other natural and unintentional human-mediated means (see Probably of Entry). Within Australia, contaminated fodder has been identified as one means of spread of Senecio madagascariensis (Bega Valley Fireweed Association no date).
Potential Economic Consequences
It is a serious pest plant in Australia and Hawaii (Starr et al. 1999). Its introduction to Japan has been fairly recent, but it is spreading rapidly there as well (Koike et al. 2006). In Colombia and Argentina, its distribution is currently considered localized (CAB International 2007). The main economic impact of this species is reduced pasture productivity (CAB International 2007). It is a strong competitor with desired pasture species and also has toxic effects on livestock (CAB International 2007). Consumption of large quantities of Senecio madagascariensis has resulted in acute poisoning and death of livestock due to pyrrolizidine alkaloid toxicosis. Consumption of smaller quantities over longer periods results in sublethal liver poisoning. Poultry, pigs, cattle and horses are all sensitive to pyrrolizidine alkaloids. Sheep and goats are fairly resistant, but continuous exposure should be avoided (CAB International 2007). In Australia, over a decade ago, costs due to herbicide use and toxic effects on cattle were estimated to be $11 million (currency not stated) in non-drought years (DRDC 1996, cited in Starr et al. 1999).
The establishment of Senecio madagascariensis into Canada would threaten Canada's ability to export commodities to the United States where it is a designated Federal Noxious Weed, as well as Australia and Japan.
Potential Environmental and Social Consequences
It can have a negative effect on biodiversity by competing with native vegetation such as grass and other low-growing plants (CAB International 2007; Sindel et al. 1998). Changes to the vegetative composition of an area due to this species may also promote soil erosion (CAB International 2007).
Uncertainty
No significant areas of uncertainty identified.
Conclusion
Senecio madagascariensis could become invasive in parts of Canada (coastal British Columbia and Vancouver Island) if it were introduced.
Technical Issues for Consideration
Senecio taxa may be distinguished based on shorter achenes (1.5 mm to 2.0 mm) and hairs confined to the achene grooves in Senecio madagascariensis as compared to longer achenes (2.5 mm) and completely hairy achenes in Senecio inaequidens. These differences should be treated with caution as they are based on limited sampling (CAB International 2007; Radford et al. 2000).
Appendix 13B: Risk Management Considerations for Senecio madagascariensis (Madacascar ragwort)
Values at Risk
- Canadian Livestock Industry
- Senecio madagascariensis contains pyrrolizidine alkaloids that are toxic to livestock (i.e., cattle, sheep, goats, and horses) and causes irreversible liver damage. Total pasture land in BC was approximately 3.4 million acres in 1996 (Statistics Canada 1996), however the majority of farms producing cattle in BC are outside of the potential distribution range (i.e., coastal British Columbia and Vancouver Island) of the weed.
- Animal Health
- It contains pyrrolizidine alkaloids that are toxic to livestock (i.e., cattle, sheep, goats, and horses) and causes irreversible liver damage.
- Biological Diversity
- It can have a negative effect on biodiversity by competing with native vegetation such as grass and other low-growing plants.
Pathways of Introduction
Natural Dispersal
The risk of introduction into Canada by natural dispersal is low as there are no known populations near the Canadian border.
Intentional Introduction
No intentional introduction pathways were identified.
Non-intentional Introduction
- Field crops not intended for propagation: The level of risk associated with the import of cereal grain into Canada is very low since imports originate primarily from areas where Senecio madagascariensis is absent.
- Hay and Straw: The level of risk associated with imports of grass and forage seed into Canada is low since they originate primarily from areas where Senecio madagascariensis is absent.
- Travellers and Their Effects: A considerable number of people cross the Canadian border every year. Approximately 5.4 million Canadians traveled abroad in the 4th quarter of 2008 alone (Statistics Canada 2009). There is a high frequency of travel between Canada and areas where Senecio madagascariensis is present (Hawaii, Australia, Japan, Colombia, Argentina and southern Africa). This pathway is considered the most likely pathway of introduction of Senecio madagascariensis into Canada, but the level of risk is unknown.
Appendix 13C: Assessment of Measures for Senecio madagascariensis (Madacascar ragwort)
- Field Crops Not Intended for Propagation
- Seeds
- Imports of forage and grass seed for sowing from countries where Senecio madagascariensis is present
- Travellers and Their Effects
Field Crops Not Intended for Propagation
Previous imports
- Between 2006 and 2008, an average of approximately $402 million per year of cereals (small grain cereals and corn, excluding rice) were imported into Canada. Of this value, less than 0.1% came from countries where Senecio madagascariensis is present. An average value of $143,000 and an average quantity of 8,102 tonnes per year (between 2006 and 2008) of cereals were imported from countries where the weed is present (Table 2).
- The level of risk associated with the import of cereal grain into Canada is very low since the imports originate primarily from areas where Senecio madagascariensis is absent.
Import data for cereal grains originating from places where Senecio madagascariensis is present.
| Product | Exporting Country Table Note ** | 2006 | 2007 | 2008 |
|---|---|---|---|---|
| HS 1001 - Wheat | Argentina | 0 | 0 | 250,050 |
| HS 1001 - Wheat | U.S. - Hawaii | 0 | 0 | 1,151 |
| HS 100590 - Maize (excluding sweet corn) | Colombia | 8,520 | 5,107 | 3,036 |
| HS 100590 - Maize (excluding sweet corn) | Argentina | 2,982 | 96,519 | 2,562 |
| HS 1008 - Buckwheat, millet, canary seed; other unmilled cereals Table Note * | Colombia | 8,181 | 13,875 | 38,927 |
| Total: | 19,683 | 115,501 | 295,726 | |
| Product | Exporting Country Table Note ** | 2006 | 2007 | 2008 |
|---|---|---|---|---|
| HS 1001 - Wheat | Argentina | 0 | 0 | 320 |
| HS 1001 - Wheat | U.S. - Hawaii | 0 | 0 | 4 |
| HS 100590 - Maize (excluding sweet corn) | Colombia | 39 | 13 | 12 |
| HS 100590 - Maize (excluding sweet corn) | Argentina | 42 | 329 | 119 |
| HS 1008 - Buckwheat, millet, canary seed; other unmilled cereals Table Note * | Colombia | 2,191 | 5,883 | 15,354 |
| Total: | 2,272 | 6,225 | 15,809 | |
Source: Statistics Canada 2009
Table Notes
- Table note star 1
-
Quantity listed as N/A
- Table note star 2
-
Countries with an import value of less than $1,000 are not included.
Potential risk mitigation measures
Regulate Senecio madagascariensis as a quarantine pest under the Plant Protection Act and add this species to the List of Pests Regulated by Canada (CFIA 2009) in order to:
- Prevent the importation, movement, and cultivation of this species in Canada.
- Enable inspectors to take appropriate action for the purposes of eradicating the pest or preventing its spread Footnote 1.
Regulatory actions under the Plant Protection Act could include one or more of the following:
- Requirement for a Phytosanitary Certificate with or without an Additional Declaration stating freedom from Senecio madagascariensis.
- Recognition of Pest Free Areas – if Senecio madagascariensis can be shown to be absent from the country/state/region from which the field crop product was propagated, then risk is negligible and additional phytosanitary requirements may be waived.
- Requirement for an import permit indicating specific import requirements and conditions for the pest status, handling and use of the commodity.
Note: End uses impact risk and may therefore impact required risk mitigation measures. Regulatory measures may be waived for those commodities that have been treated or processed such that the risk of introduction of Senecio madagascariensis has been reduced to an acceptable level.
Trade implications
- Since a small value of grain imports are originating from countries where Senecio madagascariensis is present, reduction and or loss of import markets if exporting countries are unable to meet proposed phytosanitary requirements would be low.
- Lack of Canadian regulation could compromise market access for Canadian commodities should Senecio madagascariensis become established.
- Exporting countries may have to devote resources toward inspection of grain commodities and the issuance of Phytosanitary Certificates.
- Regulation of Senecio madagascariensis under the Plant Protection Act will help to maintain export of grain from BC to the US (approximately $9.3 million in 2008; Industry Canada 2009), where Senecio madagascariensis is regulated as a federal noxious weed.
Feasibility
Resources will be needed by CFIA for marketplace monitoring and sampling, inspector training, and communication material development.
Seeds
Previous imports
- The Seed Science and Technology Section of the CFIA Saskatoon laboratory maintains a record of contaminants through the marketplace monitoring of domestic and imported seed. From the year 2000 to present, Senecio madagascariensis has not been recorded in samples analysed by the Seed Lab.
- The average value of imported forage and grass seed for sowing between 2006 and 2008 was approximately $37 million. Imports from countries where Senecio madagascariensis is present made up 1.8% (approximately $683,000 per year from 2006 to 2008) of this value (Industry Canada 2009). An approximate average of 178,000 kilograms of forage and grass seed was imported into Canada.
Imports of forage and grass seed for sowing from countries where Senecio madagascariensis is present.
Table 3a-3d: Value in Canadian Dollars ($)
| Product | Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|---|
| HS 120921 - Alfalfa | Australia | 208,607 | 234,443 | 198,929 |
| HS 120921 - Alfalfa | Argentina | 56 | 3,122 | 0 |
| Subtotal: | 208,663 | 237,565 | 198,929 | |
| Product | Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|---|
| HS 120922 – Clover | Australia | 119,673 | 364,365 | 0 |
| HS 120922 – Clover | Argentina | 43,769 | 91,082 | 146,970 |
| Subtotal: | 163,442 | 455,447 | 146,970 | |
| Product | Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|---|
| HS 120923 – Fescue | Japan | 0 | 0 | 2,505 |
| HS 120925 - Rye grass | Argentina | 0 | 17,242 | 0 |
| HS 120929 - Seed of forage plants | Argentina | 28,760 | 445,141 | 142,798 |
| HS 120929 - Seed of forage plants | Japan | 0 | 970 | 2,475 |
| Subtotal: | 28,760 | 446,111 | 145,273 | |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| Total: | 400,865 | 1,156,365 | 493,677 |
Table 3a-3d: Quantity in Kilograms (KGM)
| Product | Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|---|
| HS 120921 - Alfalfa | Australia | 69,571 | 41,718 | 31,372 |
| HS 120921 - Alfalfa | Argentina | 11 | 513 | 0 |
| Subtotal: | 69,582 | 42,231 | 31,372 | |
| Product | Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|---|
| HS 120922 – Clover | Australia | 31,500 | 105,331 | 0 |
| HS 120922 – Clover | Argentina | 17,900 | 33,300 | 41,300 |
| Subtotal: | 49,400 | 138,631 | 41,300 | |
| Product | Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|---|
| HS 120923 – Fescue | Japan | 0 | 0 | 1,474 |
| HS 120925 -Rye grass | Argentina | 0 | 24,990 | 0 |
| HS 120929 - Seed of forage plants | Argentina | 6,100 | 98,036 | 32,204 |
| HS 120929 - Seed of forage plants | Japan | 0 | 154 | 903 |
| Subtotal: | 6,100 | 98,190 | 33,107 | |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| Total: | 125,082 | 304,042 | 107,253 |
Source: Statistics Canada 2009
- Countries where the plant is present between 2006 and 2008 (Table 3 )
- The level of risk associated with imports of grass and forage seed into Canada is low since they originate primarily from areas where Senecio madagascariensis is absent.
Potential risk mitigation measures
- Regulate Senecio madagascariensis as a prohibited noxious weed (Class 1) under the Weed Seeds Order of the Seeds ActFootnote 2.
- This species meets the definition for Class 1Footnote 3 species under the Weed Seeds Order.
- All imported and domestic seed lots must be free of prohibited noxious weed seeds. Importation would require a certificate of analysis stating Senecio madagascariensis is absent from the seed lot.
- Regulate Senecio madagascariensis as a quarantine pest under the Plant Protection Act and add this species to the List of Pests Regulated by Canada (CFIA 2009) in order to:
- Prevent the importation, movement, and cultivation of this species in Canada.
- Enable inspectors to take appropriate action for the purposes of eradicating the pest or preventing its spread
- Regulatory actions under the Plant Protection Act could include one or more of the following:
- Requirement for a Phytosanitary Certificate with or without an Additional Declaration stating freedom from Senecio madagascariensis.
- Requirement for an import permit indicating specific import requirements and conditions for the pest status, handling and use of the commodity.
- Requirement for an import permit for the importation of devitalized seed, preserved specimens for scientific research purposes by recognized herbaria and research facilities and the importation of seed for research in containment facilities.
- Recognition of Pest Free Areas – if Senecio madagascariensis can be shown to be absent from the country/state/region from which the seed was harvested, then risk is negligible and additional phytosanitary requirements may be waived
- Recognition of foreign government phytosanitary noxious weed certification in countries or states of origin may allow for additional phytosanitary requirements to be waived.
Trade implications
- Exporting countries currently devote resources towards inspection of seed lots and issuance of Phytosanitary Certificates. Laboratories in exporting countries will need to be able to identify seeds of Senecio madagascariensis within a seed sample. Exporters will need to ensure freedom of Senecio madagascariensis in seed lots, otherwise CFIA can refuse import.
- There is potential for reduction and or loss of some import markets for Canadians if exporting countries are unable to meet proposed phytosanitary requirements. This loss will be very low as a small value of seed imports originate from countries where Senecio madagascariensis is present.
- Lack of Canadian regulation could compromise market access for Canadian commodities to those countries that regulate Senecio madagascariensis, should this species become established in Canada.
- Canadian regulation will sageguard trade with the U.S., Australia, Japan and possibly elsewhere where Senecio madagascariensis is regulated. Canada exported approximately $107 million worth of forage and grass seed to these 3 countries in 2008. Approximately $2.7 million worth of forage and grass seed was exported from British Colombia, where Senecio madagascariensis is most likely to establish.
Cost-effectiveness and Feasibility
- The CFIA Seed Program is already in place to prevent the entry of prohibited noxious weeds. The CFIA monitors compliance with the Canadian standards through the Marketplace Monitoring ProgramFootnote 4.
- The Seed Science and Technology Section at CFIA is currently acquiring a specimen of Senecio madagascariensis to determine the identifiabilty of its seeds.
Travellers and Their Effects
Previous imports
- A considerable number of people cross the Canadian border every year. Approximately 5.4 million Canadians traveled abroad in the 4th quarter of 2008 alone (Statistics Canada 2009b). There is a high frequency of travel between Canada and areas where Senecio madagascariensis is present (Hawaii, Australia, Japan, Colombia, Argentina and southern Africa).
- This pathway is considered the most likely pathway of introduction of Senecio madagascariensis into Canada (Castro 2009), but the level of risk is unknown. Travellers would need to spend enough time in areas (e.g. pastures, fields, roadsides) where the weed is growing for achenes to stick to shoes, clothing or other effects. Contaminated shoes, clothing or other effects would then have to be brought back across the Canadian border and achenes introduced into a suitable climate for establishment.
Risk mitigation measures
Increase public awareness of the regulation and the risk posed by Senecio madagascariensis and distribute awareness material at border crossings to the public. It is difficult to target travelers for one particular species that could be unintentionally imported. Thus, a general information and awareness campaign about the risks associated with several species may be the best option. While not considered effective by itself if the risk is high, increased public awareness is a viable action if the level of risk is low.
Trade implications
None anticipated.
Cost-effectiveness and Feasibility
- Resources will have to be allocated for promotional materials to be distributed at border crossings.
- Public awareness campaigns could be an effective way to lower the risk of introduction of Senecio madagascariensis into the country. This campaign could also be done in conjunction with other weed species to reduce costs and to inform the public on a more general level about invasive alien plants.
Appendix 14A: Pest Risk Assessment Summary for Solanum elaeagnifolium (silverleaf nightshade)
- Identity of Organism
- Organism Status
- Current Regulatory Status
- Probability of Entry
- Probability of Establishment
- Probability of Spread
- Potential Economic Consequences
- Potential Environmental and Social Consequences
- Uncertainty
- Conclusion
- Technical Issues for Consideration
Identity of Organism
Name: Solanum elaeagnifolium (Solanaceae; USDA-ARS, 2009)
Synonyms: None listed (e.g. CAB International, 2007; USDA-ARS, 2009; USDA-NRCS, 2009).
English common names: Bitter apple, bitterleaf nightshade, bull nettle, prairie-berry, silverleaf bitter-apple, silverleaf nightshade, silverleaf-nettle, tomato weed, white horse-nettle (EPPO, 2007; USDA-ARS, 2009).
French common names: Morelle jaune (EPPO, 2007)
Description: Solanum elaeagnifolium is an erect, deep-rooted, shrub-like, perennial herb. Plants are multi-stemmed and highly branched; reaching a height of up to 80 cm. Leaves are alternate, stalked, and lance-shaped with wavy or scalloped edges. Stems and leaves are densely covered with short, fine, star-shaped hairs that give the plant a silvery-white appearance. Stems and main veins of leaves also have numerous slender, yellow to red prickles. Flowers are star-shaped and bright blue to purple/violet or occasionally white, with 5 fused petals and 5 prominent yellow anthers. Flowers give rise to clusters of smooth, globular berries that change in colour as they mature from green with stripes to mottled yellow and orange or brownish when ripe. Each berry contains 60-120 seeds that are flat, light, and closely resemble those of tomatoes (Parsons and Cuthbertson, 2001; CAB International, 2007; EPPO, 2007; Mekki, 2007).
Solanum elaeagnifolium is a weed of crops, pastures, and disturbed areas in its native and introduced ranges worldwide. All parts of the plant, but particularly berries, are poisonous to livestock. Historically, the berries and seeds were used by aboriginal peoples in the south-western U.S. in the preparation of food and clothing (Boyd et al., 1984). More recently, the fruit has been identified as a rich source of steroidal alkaloids (e.g. solasodine), used in the synthesis of contraceptive and corticosteroid drugs. These have been commercially extracted in India and Argentina (Maiti and Mathew, 1967; Parsons and Cuthbertson, 2001; CAB International, 2007).
Organism Status
Solanum elaeagnifolium is not reported to occur in the Canadian flora (Scoggan, 1979; Kartesz, 1999; CFIA, 2008), and no evidence was found that it is cultivated in Canada. Based on this information, Solanum elaeagnifolium is considered absent with no pest records in the PRA area.
Current Regulatory Status
Solanum elaeagnifolium is not currently regulated as a pest in Canada, either federally or provincially (although Solanum spp. are regulated in Manitoba). It is not a Federal Noxious Weed in the U.S., but it is regulated as a noxious weed and/or noxious weed seed in 19 U.S. states (Alabama, Arkansas, Arizona, California, Colorado, Florida, Georgia, Hawaii, Idaho, Kansas, Louisiana, Michigan, New Mexico, Nevada, Oklahoma, Oregon, South Carolina, Texas, and Washington) (Rice, 1997-2008; USDA-ARS, 2009; USDA-NRCS, 2009). Worldwide, it is a quarantine pest in Belarus, Russia, and Ukraine, and is controlled under noxious weed legislation in Australia and South Africa (EPPO, 2007). In Europe, it is included on the European and Mediterranean Plant Protection Organization (EPPO) list of species recommended for regulation as quarantine pests by member countries (EPPO, 2009).
Probability of Entry
The most likely pathway of entry of Solanum elaeagnifolium into the PRA area is unintentional introduction as a contaminant in commodities or conveyances. It has been distributed around the world in contaminated fodder and crop seed, and may also be spread by livestock and manure, agricultural machinery, vehicles, bulldozers and other earth-moving equipment (Boyd et al., 1984; Parsons and Cuthbertson, 2001; CAB International, 2007; EPPO, 2007; Mekki, 2007). Soil, sand, and ornamental plants can also be contaminated by fragments of roots or seeds (EPPO, 2007).
| Type of pathways | Specific pathways |
|---|---|
| Natural dispersal | Natural spread of Solanum elaeagnifolium occurs by seed and vegetatively from cut root sections. Seeds are fleshy berries adapted to dispersal by birds and animals. Studies have shown seed to be viable after passing through the digestive tracts of animals (Parsons and Cuthbertson, 2001). The fruit can also float, and be dispersed along rivers and streams (Boyd and Murray, 1982; EPPO, 2007) and dried berries can be dispersed by strong winds in the winter (Parsons and Cuthbertson, 2001). This is a possible pathway of entry into Canada, as there are populations in the U.S. approaching the Canadian border (e.g. Washington). Natural spread by vegetative fragments would be local, but spread of seeds by birds, animals, wind, or water could cover greater distances. |
| Intentional introduction | Solanum elaeagnifolium is not generally cultivated as an ornamental (Bailey and Bailey, 1976; Isaacson and Allen, 2007). Solanum elaeagnifolium has some limited use as fodder in South Africa (CAB International, 2007), and the steroidal alkaloid solasodine has been commercially extracted in India and Argentina (EPPO, 2007). However, it is unclear to what extent (if any) the plant is cultivated in this regard. This pathway is considered unlikely at this time. |
| Unintentional introduction | Solanum elaeagnifolium is a weed of crops and pastures, and has been distributed around the world in contaminated fodder and crop seed (Parsons and Cuthbertson, 2001). It may also be spread by livestock and manure, agricultural machinery, vehicles, bulldozers and other earth-moving equipment, as well as in soil, sand, and ornamental plant material (EPPO, 2007; Boyd et al., 1984). This is considered the most likely pathway of entry into Canada. |
Probability of Establishment
Solanum elaeagnifolium is native to the south-western U.S. and north-eastern Mexico. It is sometimes considered native to Argentina, although the nature of the herbivorous insect fauna suggests this is a secondary distribution (Boyd et al., 1984; CAB International, 2007; EPPO, 2007). It has been introduced elsewhere in North and South America, and is now widespread in the U.S. in all but the Great Lakes and New England regions (Figure 1; Kartesz, 1999; CAB International, 2007; USDA-NRCS, 2009). It is also introduced in other parts of the world including Australia, India, South Africa and around the Mediterranean basin (Parsons and Cuthbertson, 2001; CAB International, 2007; EPPO, 2007; GBIF, 2008). Most of its range falls in NAPPFAST zones 8-9 and above, but occurrences in the U.S. suggest it can survive in zone 6 and probably 5 (Figure 2).
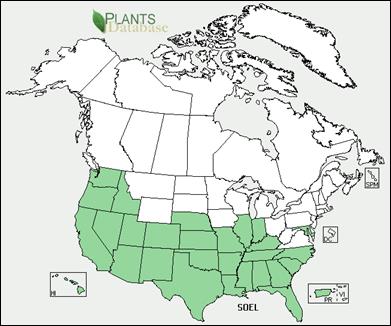
Description for Figure 1:
This map shows the current distribution of in North America indicating its presence through the use of the colour green. Two thirds of the United States is covered in green predominantly the lower western half, none of the states along the north eastern coast contain green except for Maryland.
Source: USDA-NRCS, 2009.
Solanum elaeagnifolium is adapted to a wide range of habitats but appears mostly in warm-temperate regions in areas of relatively low annual rainfall (250-600 m). It thrives on disturbed land and is not confined to any particular soil type. Infestations are more serious in dryland situations, but irrigated crops are also prone to invasion (Parsons and Cuthbertson, 2001; CAB International, 2007). In addition to cultivated and agricultural land, it occurs along roadsides, railways, riverbanks, and canal-sides, and in rangeland, livestock corrals, construction sites, and wastelands (CAB International, 2007; EPPO, 2007; Mekki, 2007).
Based on its native distribution and available records, it appears that Solanum elaeagnifolium may find climatic and ecological conditions suitable for establishment and spread in the PRA area, in the parts of British Columbia, southern Ontario and the Maritimes which comprise zones 6-9 and possibly also zone 5.
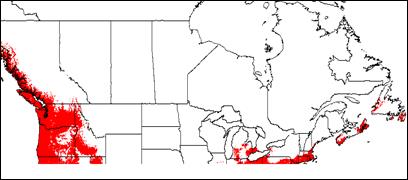
Description for Figure 2-1:
This image shows the potential range of Solanum elaeagnifolium (silverleaf nightshade) in Canada and northern America through the use of a map. Red is used to indicate the regions in which Solanum elaeagnifolium could survive in according to the Canadian Plant Hardiness Zones map, in this case NAPPFAST Hardiness Zones 6-9. In Canada, this includesSolanum elaeagnifolium is likely to become weedy or invasive in parts of Canada, including southern and coastal British Columbia, southern Ontario, and the Maritimes. Most of the red can be found in the U.S., this includes the parts of the east and west coasts as well as areas located near the Great Lakes extending downwards beyond the image.
NAPPFAST zones 6-9.
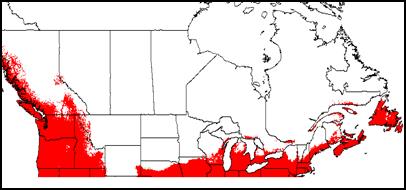
Description for Figure 2-2:
This image shows the potential range of Solanum elaeagnifolium in Canada and northern America through the use of a map. Red is used to indicate the regions in which Solanum elaeagnifolium could survive in according to the Canadian Plant Hardiness Zones map, in this case NAPPFAST Hardiness Zones 5-9. In Canada, the potential range of Solanum elaeagnifolium includes parts of Atlantic Canada, southern Ontario and coastal and southern BC. Most of the red can be found in the U.S., this includes the greater parts of the east and west coasts extending inwards reaching most of the central states as well as areas located near the Great Lakes extending downwards beyond the image.
NAPPFAST zones 5-9.
Probability of Spread
Natural spread of Solanum elaeagnifolium occurs both by seed, and vegetatively from cut root sections. Flowers are cross-pollinated by insects, primarily bees (Hardin et al., 1972; EPPO, 2007), and produce clusters of fleshy berries that may be dispersed by birds, animals, wind and water. A single plant generally produces 40-60 fruits per growing season (up to 200), each containing 60-120 seeds (CAB International, 2007; EPPO, 2007). Dense populations are capable of producing 250 million seeds/ha (Boyd and Murray, 1982). Seeds are highly viable and may also last up to 10 years in soil (EPPO, 2007; Mekki, 2007). Plants are also able to regenerate vegetatively as early as ten days after germination (EPPO 2007). Established plants have extensive root systems, and new shoots are produced from the lateral roots each spring from as deep as 50 cm. All parts of the root are capable of forming shoot buds (Parsons and Cuthbertson 2001); root fragments only 1 cm long retain the ability to sprout, and remain viable for up to 15 months (Boyd et al. 1984; Parsons and Cuthbertson 2001; EPPO 2007). Despite this variety of natural dispersal mechanisms, most long-distance spread has been attributed to human activity.
Potential Economic Consequences
Solanum elaeagnifolium is an agricultural weed that competes for moisture and nutrients with a variety of crops in both dryland and irrigated conditions. Several crops are affected worldwide, the most important of which are cereals (wheat, sorghum, maize), alfalfa, and cotton (Parsons and Cuthbertson, 2001; CAB International, 2007). The most serious crop losses have been recorded in alfalfa (in Australia, South Africa, and U.S.), cotton, sorghum, maize and groundnut (in Morocco, U.S.), wheat (in Australia, U.S.) and cultivated pastures (in Australia, Greece, Morocco, U.S.) (CAB International, 2007). Other crops affected include many vegetables (e.g. potato, asparagus, tomato), grapes, and some fruit trees (e.g. peaches). In addition to yield reductions, presence of the weed in harvested products reduces their quality and sale (CAB International, 2007).
Potential Environmental and Social Consequences
Solanum elaeagnifolium is primarily an agricultural weed that invades croplands, pastures, and disturbed and marginal areas such as roadsides. No reports of environmental impacts or effects on natural areas were found, although it may replace natural vegetation in overgrazed rangeland (EPPO, 2007; Mekki, 2007). Social impacts may result from the loss of value of agricultural land infested with the weed; in Morocco the value of infested fields reportedly decreased by 25%, while in the U.S. entire farms have been abandoned because of the weed (Gmira et al., 1998; EPPO, 2007).
Uncertainty
Further investigation of the distribution of Solanum elaeagnifolium in the U.S. and Europe could help refine the estimated potential range in Canada. No other significant areas of uncertainty were identified.
Conclusion
Based on the outcome of this risk assessment, Solanum elaeagnifolium has the potential to become weedy or invasive in areas in which it can establish. Parts of British Columbia, southern Ontario and the Atlantic provinces are considered to be the areas most at risk. This plant should be considered for regulation according to Canada's Plant Protection Act and Seeds Act.
Technical Issues for Consideration
Various other species of the genus have reportedly been confused with Solanum elaeagnifolium in the field in Australia, South Africa and the U.S. However, it is not generally considered difficult to distinguish or detect as a commodity contaminant (CAB International, 2007).
Appendix 14B: Risk Management Considerations for Solanum elaeagnifolium (silverleaf nightshade)
- Values at Risk
- Potential Mitigation Measures for Natural Means of Dispersal
- Potential Mitigation Measures for Intentional Introduction Pathways
- Potential Mitigation Measures for Non-Intentional Introduction Pathways
Values at Risk
Several crops are affected by Solanum elaeagnifolium worldwide, the most important being cereals (wheat, sorghum and maize), alfalfa (lucerne) and cotton. In Australia and the U.S., wheat production losses have been as high as 50%. A 2007 survey of farmers in Australia showed that Solanum elaeagnifolium has spread across the wheat belt of Victoria, New South Wales and South Australia. On average, the total farm cost for the weed was $1,730 (AUD) per year for control and $7,786 (AUD) per year in production losses (Kidston et al. 2006). Sorghum losses have ranged from 4-10% under optimal moisture conditions (CAB International, 2007; Sforza and Jones 2007).
The amount of land in each province in hardiness zones 5-9 that are planted with the species most affected by Solanum elaeagnifolium. The Province of Ontario has the greatest amount of crop area that could be impacted. In 2008, Ontario counties in hardiness zones 5 and 6 produced approximately 12 million bushels of wheat (OMAFRA, 2009) Maize production was approximately 48 million bushels in 2008 in Ontario counties (OMAFRA, 2009). Sorghum production in the respective provinces is considered marginal. A total of 22 hectares of organic sorghum was produced in Canada in 2005, with 15 hectares originating from provinces where Solanum elaeagnifolium could establish (Macey 2006).
| Type of Commodity (Pathways) |
Introduction | Previous Imports | Potential Risk Mitigation Measures | Trade Implications | Cost Effectiveness & Feasibility |
|---|---|---|---|---|---|
| Seed | Intentional | No data available |
| Market loss for exploring countries would be minimal | Seed Program already in place |
| Seed | Unintentional (as a contaminant) | This species has not previously been recorded in imported and Canadian seeds samples Level of risk is high since maize and forage plants originate primarily from areas where this species is present |
|
|
|
| Field crops not intended for propagation | Unintentional | 300,000 tonnes of wheat, sorghum, maize and soybeans from countries with Solanum eleagnifolium were import in 2008 | Regulate under the PPA | Potential for reduction or loss of some import markets for Canada if physanitary requimrents can not be met. | Resources will be needed for marketplace monitoring |
| Hay and Straw | Unintentional | Hay and straw imports were around $13 million in 2008 (harvested frequently and before weeds produce fruit) | Regulate under the PPA | Regulation of this species will safeguard trade of hay and straw with US, Potential reduction of import markets in Canada | Resources will be needed for CFIA marketplace monitoring |
| Vehicles and Used Farm Machinery | Unintentional | Considerable volume of vehicles cross US and Canadian border Information not available on imports of used farm machinery | Enforcement of Directive 95-96: Phytosanitary requirements for soil and related matter, alone or in association with plants | ||
| Nursery Stock with soil | Unintentional | In 2008, 73% of all nursery stock imports came from countries where this species is present | Regulate under the PPA | Potential reduction in import market for Canada if exporting countries cannot meet requirements | Phytosanitary certificate are currently issued by exporting countries for nursery stock. |
| Manure | Unintentional | Data is unavailable | Bagged, composted manure regulated by Health of Animals Regulations. There are currently no standards for fresh manure. | No trade implications | If heating process makes weed seeds inviable, the n this would be a cost effective measure. |
| Livestock | Unintentional | Risk associated with livestock pathway is relatively low | It is not feasible to implement mitigation measures for this pathway at this time. |
Potential Mitigation Measures for Natural Means of Dispersal
Natural dispersal of Solanum elaeagnifolium seeds by birds, animals or water could cover great distances and this represents a possible pathway of entry into Canada. Currently, populations of the plant in Washington are in counties at the southern margins of the state (USDA-NRCS, 2009). Washington State carries out eradication of Class A weeds as required by law (NWCB, 2009). Oregon and Idaho also have programs to eradicate noxious weeds (ODA, 2009; ISDA, 2005). If these programs are effective, the risk of natural dispersal as a pathway into Canada will decrease.
Since Solanum elaeagnifolium is currently at the southern margins of bordering states, an early detection and rapid response (EDRR) program should be employed such as the framework established in British Columbia. The Invasive Plant Council of BC has approved an EDRR framework that describes the process, steps and stakeholders involved in carrying out an EDRR system in the province (IPCBC, 2007). Early detection can be made possible with the use of atlases already available such as E-flora BC (Klinkenberg 2008) that inventory native, non-native and invasive species.
Potential Mitigation Measures for Intentional Introduction Pathways
Although sources indicate Solanum elaeagnifolium is not currently being sold as an ornamental plant in Canada (CNLA, 2009), further research found two Internet sites with seeds of Solanum elaeagnifolium for sale. While these companies indicate that they will ship to Canada, it is unknown how often, if any, seeds have been brought into Canada through this pathway. The potential for intentional introduction of Solanum elaeagnifolium into Canada is low.
Seed
Previous imports
None known. There is no data available specific to imports of Solanum elaeagnifolium.
Potential risk mitigation measures
Regulate Solanum elaeagnifolium as a prohibited noxious weed (Class 1) under the Weed Seeds Order of the Seeds Act Footnote 1.
- This species meets the definitions for Class 1Footnote 2 species under the Weed Seeds Order.
- All seed lots sold or imported into Canada must be free of prohibited noxious weed seeds. Imported seed lots would require a certificate of analysis stating Solanum elaeagnifolium is absent from the seed lot before it can be imported.
Regulate as a quarantine pest under the Plant Protection Act. Add this species to the List of Pests Regulated by Canada (CFIA, 2009) in order to:
- Prevent the importation, movement, and cultivation of this species in Canada. Currently, seed of many new crop species, such as field crops, can be imported without a Permit to Import or a Phytosanitary Certificate.
- Enable inspectors to take appropriate action for the purposes of eradicating the pest or preventing its spread Footnote 3.
Regulatory actions under the Plant Protection Act could include the following:
- prohibit importation of Solanum elaeagnifolium seed. The only exceptions would be for the importation of devitalized seed and the importation of seed or preserved specimens for scientific research purposes by recognized herbaria and government research facilities Footnote 4.
- Require importers of plant material from the non-continental U.S. to apply for a Permit to Import with scientific name specified.
- Seed of horticultural plants is not within the scope of the CFIA's draft directive D-08-04 on plants for planting (CFIA 2008). Therefore phytosanitary requirements will be specified under a new regulatory directive or D-08-04 will be revised .
Sufficient information about the use of Solanum elaeagnifolium as a potential crop does not exist. If the proponent, located in Canada, needs to collect more information about the plant (e.g. to generate data for a determination of environmental safety), then confined research field trials under Part V of the Seeds Regulations could be authorized by the Plant Biosafety Office.
Trade Implications
- Currently, there is not a large market for Solanum elaeagnifolium seeds in Canada, so market loss for exporting countries will be minimal.
- Lack of Canadian regulation could lead to establishment of Solanum elaeagnifolium in Canada, which could compromise market access for Canadian commodities to those countries that regulate Solanum elaeagnifolium. Worldwide, it is a quarantine pest in Belarus, Russia, and Ukraine, and is controlled under noxious weed legislation in Australia and South Africa (EPPO, 2007). In Europe, it is included on the European and Mediterranean Plant Protection Organization (EPPO) list of species recommended for regulation as quarantine pests by member countries (EPPO, 2009).
Cost-effectiveness and Feasibility
- The CFIA Seed Program is already in place to prevent the entry of prohibited noxious weeds. Verification of compliance is carried out through the Seed Marketplace Monitoring Program Footnote 5.
- No additional staff will be required at the CFIA Plant Health and Biosecurity Permit Office to regulate the species. Applications to import are reviewed by officers on a regular basis.
Potential Mitigation Measures for Non-Intentional Introduction Pathways
Field Crops Not Intended for Propagation
Previous imports
The total volume of wheat, sorghum, maize and soybeans Footnote 6 imported in 2008 from countries where Solanum elaeagnifolium is present was approximately 300,000 metric tonnes (Appendix 14C; Statistics Canada, 2009). The majority of these imports (99.5%) came from infested U.S. states (Industry Canada, 2009).
Potential risk mitigation measures
Regulate Solanum elaeagnifolium as a quarantine pest under the Plant Protection Act and add this species to the List of Pests Regulated by Canada in order to:
- Prevent the importation, movement, and cultivation of this species in Canada.
- Enable inspectors to take appropriate action for the purposes of eradicating the pest or preventing its spread Footnote 7.
Regulatory actions under the Plant Protection Act could include one or more of the following:
- Exporters could be required to provide a Phytosanitary Certificate with or without an Additional Declaration stating freedom from Solanum elaeagnifolium.
- Requirement for an import permit issued by the CFIA indicating specific import requirements and conditions for the pest status, handling and use of the commodity. It is the importer's responsibility to apply for and obtain the permit Footnote 8.
- Recognition of Pest Free Areas– if Solanum elaeagnifolium can be shown to be absent in the country/state/region from which the field crop product was propagated, then risk is negligible and additional phytosanitary requirements may be waived.
End uses have an impact on risk and may, therefore, affect required risk mitigation measures. Phytosanitary measures may be waived for those commodities that have been treated or processed such that the risk of introduction of Solanum elaeagnifolium has been reduced to an acceptable level.
All risk mitigation measures for field crop commodities containing Solanum elaeagnifolium must be taken with consideration for requirements/measures for pests other than plants (e.g. pathogens and insects).
Trade implications
- Exporting countries may have to devote resources towards inspection and issuance of phytosanitary certificates. Laboratories in foreign countries will need to be able to identify seeds of Solanum elaeagnifolium. Exporters will need to ensure freedom of Solanum elaeagnifolium in field crop shipments, otherwise CFIA can refuse import.
- Potential for reduction and or loss of some import markets for Canada should exporting countries be unable to meet proposed phytosanitary requirements.
- Not controlling import and spread of Solanum elaeagnifolium may result in infestations of field crops for export; if the importing country prohibits this species, market access for Canadian commodities could be compromised. Canada exported around $2 billion worth of wheat, sorghum and maize in 2008; approximately $733 million of exports went to states in the U.S. that regulate Solanum elaeagnifolium (Industry Canada, 2009).
- Canadian regulation will facilitate trade with those countries and states which have established regulatory control of Solanum elaeagnifolium. Worldwide, it is a quarantine pest in Belarus, Russia, and Ukraine, and is controlled under noxious weed legislation in Australia and South Africa (EPPO, 2007). In Europe, it is included on the European and Mediterranean Plant Protection Organization (EPPO) list of species recommended for regulation as quarantine pests by member countries (EPPO, 2009).
Cost-effectiveness and Feasibility
- Resources will be needed by CFIA for marketplace monitoring and sampling, inspector training, and communication material development.
- Solanum elaeagnifolium seeds can be readily screened out of grain lots due to the relatively large size (2-3 mm wide and 2-2.5 mm) and distinctly smooth surface and hilum (R. Wang – National Seed Herbarium, personal communication). Prior to export, grain can be cleaned to remove contaminant seeds.
- Field inspections or laboratory testing in the exporting country could be used to ensure freedom from Solanum elaeagnifolium.
Seed
Previous imports
- The Seed Science and Technology Section of the CFIA Saskatoon laboratory maintain a record of contaminants through the marketplace monitoring of domestic and imported seed. Solanum elaeagnifolium has not previously been recorded in imported and Canadian seed samples.
- The total value of seeds of maize and forage plants (alfalfa, clover, fescue, kentucky blue grass, rye grass and timothy grass) imported from countries where Solanum elaeagnifolium is present was approximately $89 million and the total value was approximately 35 million kilograms in 2008 (Appendix 14D; Statistics Canada, 2009). This represents about 54% of the total value of these commodities imported in 2008 (Industry Canada, 2009).
- The level of risk associated with imports of seed of maize and forage plants into Canada is relatively high since they originate primarily from areas where Solanum elaeagnifolium is present. Ninety percent of total imports of these seeds came from the U.S. (including states where Solanum elaeagnifolium is currently absent; Industry Canada, 2009), so the risk of introduction will increase if the range of the plant increases to states where it is currently absent.
Potential risk mitigation measures
Regulate Solanum elaeagnifolium as a prohibited noxious weed (Class 1) under the Weed Seeds Order of the Seeds Act.
- This species meets the definitions for Class 1 species under the Weed Seeds Order.
- All imported or domestic seed lots must be free of probhibited noxious weed seeds. Imported seed lots would require a certificate of analysis stating Solanum elaeagnifolium is absent from the seed lot before it can be imported.
Regulate Solanum elaeagnifolium as a quarantine pest under the Plant Protection Act and add this species to the List of Pests Regulated by Canada in order to:
- Enable inspectors to take appropriate action for the purposes of eradicating the pest or preventing its spread.
- Prevent the importation, movement, and cultivation of this species in Canada.
Regulatory actions under the Plant Protection Act could include one or more of the following:
- Exports could be required to provide a Phytosanitary Certificate with or without an Additional Declaration stating freedom from Solanum elaeagnifolium.
- Requirement for seed from infested areas to be allowed entry into Canada only after the importer obtains a CFIA issued import permit indicating specific import requirements and conditions for handling and use of the commodity. Prior to permit issuance, a facility inspection by CFIA and verification of importers ability to meet permit requirements may be required
- Negotiate phytosanitary agreements to certify imports from pest free areas and/or recognize noxious weed certification in countries or states of origin.
Trade implications
- Regulation of Solanum elaeagnifolium under the Seeds Act and Plant Protection Act will facilitate seed trade with the 19 U.S. states where Solanum elaeagnifolium is regulated as a noxious weed. It will also facilitate trade with Belarus, Russia, China, Ukraine, Australia and South Africa where it is a quarantine pest. Canada exported approximately $61 million worth of alfalfa, clover, fescue, Kentucky blue grass, rye grass and timothy grass seed in 2008 to the states and countries where Solanum elaeagnifolium is regulated.
- Exporting countries will need to devote resources towards inspection of seed lots and issuance of phytosanitary certificates. Laboratories in foreign countries will need to be able to identify seed of Solanum elaeagnifolium within a seed sample. Exporters will need to ensure freedom of Solanum elaeagnifolium in seed lots, otherwise CFIA can refuse import.
- There is potential for reduction and or loss of some import markets for Canada if exporting countries are unable to meet proposed regulatory requirements.
Cost-effectiveness and Feasibility
- The CFIA Seed Program is already in place to prevent the entry of prohibited noxious weeds. The CFIA monitors compliance with the Canadian standards through the Marketplace Monitoring Program Footnote 9.
- Seed of Solanum elaeagnifolium can be easily identified by trained analysts (R. Wang – National Seed Herbarium, personal communication) .
- Prior to export, seed is typically cleaned to remove contaminant seeds. Solanum elaeagnifolium seeds can be readily screened out of seed lots due to the relatively large size (2-3 mm wide and 2-2.5 mm) and distinctly smooth surface and hilum (R. Wang – National Seed Herbarium, personal communication).
Hay and Straw
Previous imports
- The total value of hay and straw imports was around $12 million in 2008; 91% of this value came from countries and states where Solanum elaeagnifolium is present (Appendix 14E; Industry Statistics Canada, 2009).
- Between 1999 and 2004, high quantities (an average of around $10 million per year) of hay and straw were imported from countries and states where Solanum elaeagnifolium is present (Statistics Canada, 2009).
- Between 1999 and 2004, high quantities of hay and straw (an average of $9.5 million per year) were imported from Washington and Oregon, where Solanum elaeagnifolium is present. This represents the majority of hay and straw imported into Canada. Since 2004, imports of hay and straw have remained relatively constant (Industry Canada, 2009).
- Although imports of hay and straw into Canada originate primarily from areas where Solanum elaeagnifolium is present, the level of risk associated with this pathway may be low if harvest occurs frequently and before the weeds produce fruit, which usually occurs from the end of spring until the fall (EPPO 2007).
Potential risk mitigation measures
Regulate Solanum elaeagnifolium under the Plant Protection Act as a quarantine pest by placing it on the List of Pests Regulated by Canada in order to:
- Prevent the importation, movement, and cultivation of this species in Canada.
- Enable inspectors to take appropriate action for the purposes of eradicating the pest or preventing its spread.
Regulatory actions could include one or more of the following:
- Exporters could be required to obtain a Phytosanitary Certification with or without an Additional Declaration stating freedom from Solanum elaeagnifolium.
- Requirement for an import permit indicating specific import requirements and conditions for the pest status, handling and use of the commodity could be required. It is the importer's responsibility to apply for and obtain the permit.
- Recognition of Pest Free Areas – if Solanum elaeagnifolium can be shown to be absent in the country/state/region from which the field crop product was propagated, then risk is negligible and additional phytosanitary requirements may be waived.
- Requirement for heat treatment or pelletization for hay or straw intended for use as a biofuel stock.
Trade implications
- Regulation of Solanum elaeagnifolium will safeguard trade of hay and straw with the 19 states that regulate Solanum elaeagnifolium. Canada exported $4.5 million worth of hay and straw in 2008 to states where the weed is regulated as a noxious weed.
- Exporting countries may have to devote resources towards inspection of hay and straw commodities and issuance of Phytosanitary Certificates. Inspectors in foreign countries need to be able to identify berries and seeds of Solanum elaeagnifolium that could attach themselves to hay and straw. Exporters need to ensure freedom of Solanum elaeagnifolium in hay and straw, otherwise CFIA can refuse import.
- Potential for reduction in and or loss of some import markets for Canada if exporting countries are unable to meet proposed regulatory requirements. Since 98% of hay and straw imports come from the U.S., and the majority from states where Solanum elaeagnifolium is invasive, there could be significant market losses of this commodity if Canadian regulations cannot be met.
Cost-effectiveness and Feasibility
Resources will be needed by CFIA for marketplace monitoring and sampling, inspector training, and communication material development.
Vehicles and Used Farm Machinery
Previous imports
- A considerable volume of vehicles cross the U.S.-Canada border every year.
- Information is not available on the volume of imports of used farm machinery.
Potential Risk mitigation measures
Enforcement of the Directive 95-26: "Phytosanitary requirements for soil and related matter, and for items contaminated with soil and related matter" (Canada, 2008).
- In 2003, the Canada Border Services Agency (CBSA) assumed responsibility for the initial import inspection services in respect to the Acts and Regulations administered by the CFIA to the extent that they are applicable at Canadian border points. The inspection of goods that may be contaminated with soil are among the responsibilities that were transferred to the CBSA in 2003. The Food, Plant and Animals Programs Section of the CBSA is currently finalizing its Standard Operating Procedures (SOP) concerning the "Inspection of Imported Goods Potentially Contaminated with Soil." This SOP provides the CBSA's Border Services Officers with formal procedures for the inspection and disposition of goods that may be contaminated with soil, including used agricultural machinery and vehicles.
- Vehicles and farm machinery should be thoroughly checked for berries and root fragments of Solanum elaeagnifolium, especially if originating from infested areas.
Nursery Stock with Soil
All parts of the root of Solanum elaeagnifolium are capable of forming shoot buds; root fragments only 1 cm long retain the ability to sprout. In addition, sections of taproot can remain viable for up to 15 months (Boyd et al., 1984; Parsons and Cuthbertson, 2001). Root fragments of Solanum elaeagnifolium can contaminate soil in imported nursery stock and establish if planted in suitable growing conditions.
Previous imports
- The total value of imports of nursery stock was around $173 million in 2008 Footnote 10. Approximately 73% of this value came from countries where Solanum elaeagnifolium is present, with 64% coming from the continental U.S. (Industry Canada, 2009). While soil from the non-continental U.S. is prohibited as per Directive D-95-26: Phytosanitary requirements of soil and related matter, alone or in association with plants, soil associated with nursery stock from the continental U.S is permitted in some cases. All but 2 states (of the 28) infested with Solanum elaeagnifolium exported nursery stock to Canada; Florida, Oregon, California and Washington exported the highest value of nursery stock at around $98 million value. It is not possible to determine the amount of nursery stock from the continental U.S. arriving with soil.
- The level of risk associated with this pathway is unknown. One reference (EPPO, 2007) made a statement that ornamental plants are a possible pathway for this species, but it is unknown how often ornamental plants are contaminated with Solanum elaeagnifolium.
Potential Risk mitigation measures
Regulate Solanum elaeagnifolium under the Plant Protection Act as a quarantine pest and add it to the List of Pests Regulated by Canada in order to:
- Prevent the importation, movement, and cultivation of this species in Canada.
- Enable inspectors to take appropriate action for the purposes of eradicating the pest or preventing its spread.
Regulatory actions could include one or more of the following:
- Addition of Solanum elaeagnifolium to D-95-26 Phytosanitary requirements for soil and related matter, alone or in association with plants Appendix 2: Additional declarations required for shipments from the U.S. All soil and related matter in association with plants requires a phytosanitary certificate stating freedom from pests named in this appendix.
- Requirement for importers to obtain a phytosanitary certificate with or without an Additional Declaration stating freedom from Solanum elaeagnifolium.
- Requirement for nursery stock imported from areas infested with Solanum elaeagnifolium to be allowed entry into Canada only after the importer obtains a CFIA issued import permit indicating specific import requirements and conditions for pest status, handling and use of the commodity. Prior to permit issuance, a facility inspection by CFIA and verification of importers ability to meet permit requirements may be required. It is the importer's responsibility to apply for and obtain the permit.
- Recognition of pest free areas; if the exporting country shows Solanum elaeagnifolium to be absent from the country/state/region from which the nursery stock is produced, then risk is negligible.
Trade implications
- Potential for reduction in and or loss of some import markets for Canada should exporting countries be unable to meet proposed regulatory requirements. Since a large proportion of nursery stock comes from infested areas of the U.S., there could be significant market losses of this commodity if Canadian regulations cannot be met.
- The exporting country may have to devote resources toward inspection of pest-free areas of production and the issuance of Phytosanitary Certificates. These measures are currently in place for other pests regulated by Canada.
Cost-effectiveness and Feasibility
Phytosanitary certificates are currently issued by exporting countries for nursery stock and exporting countries currently comply with the phytosanitary requirements set out in D-95-26, including pest-free areas of production.
Manure
Previous imports
Data is unavailable to determine the total values of manure imports.
Potential risk mitigation measures
- Bagged, composted, manure may be imported according to the Health of Animals Regulations (HAR), s. 46.1 and manure may be imported according to the HAR, s. 46.1 if accompanied by an official export certificate from the U.S. and if heated to a minimum of 77 degrees centigrade for at least 7 days. It is possible that this heating process may make any weed seeds inviable at time of entry, in which case, the risk associated with this pathway would be negligible.
- There are currently no standards in place for the import of fresh manure, but it is uncertain whether this is transported across the border. Most manure and fertilizer is allowed entry into the country if deemed acceptable by the CFIA Animal Health Directorate.
- More information is required to determine if the regulations currently in place for manure would prevent viable seeds from entering the country.
Trade implications
No trade implications are expected since regulations are already in place for manure.
Cost-effectiveness and Feasibility
If the heating process does make weed seeds inviable, this provides a cost-effective measure for the manure pathway.
Livestock
Previous imports
- Between 2004 and 2008, approximately 11,400 live horses, asses, bovine animals, swine, sheep and goats for reproduction were imported into Canada from countries where Solanum elaeagnifolium is present. The majority of these animals were horses (CFIA internal data).
- The risk associated with the livestock pathway is relatively low due to the small number of animals for reproduction being imported into Canada from places where Solanum elaeagnifolium is established. In addition, berries of Solanum elaeagnifolium can be toxic to cattle and the plants will not be eaten unless there are no other food sources available (EPPO 2007).
Potential risk mitigation measures
It is not feasible to implement mitigation measures for this pathway at this time.
Appendix 14C: Import Data for selected cereal grains from countries where Solanum elaeagnifolium is present (2006-2008)
Value in Canadian Dollars ($)
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| U.S. - Washington | 433,287 | 2,317,101 | 1,825,056 |
| U.S. - Illinois | 312,720 | 1,003,852 | 1,529,417 |
| Argentina | 0 | 0 | 250,050 |
| U.S. - Arkansas | 0 | 0 | 115,175 |
| U.S. - Indiana | 411,493 | 1,748 | 36,159 |
| Oceania (Pacific) | 4,250 | 7,626 | 6,762 |
| U.S. - California | 4,974 | 8,701 | 5,260 |
| U.S. - Nebraska | 352,212 | 1 | 3,967 |
| U.S. - Oregon | 1,508,006 | 422,496 | 1,707 |
| U.S. - Oklahoma | 769 | 0 | 1,384 |
| U.S. - Hawaii | 0 | 0 | 1,151 |
| U.S. - Colorado, Italy, MacedoniaTable Note * | 784 | 1,191 | 805 |
| U.S. - Missouri | 0 | 38,835 | 467 |
| U.S. - Utah | 4,648 | 5,445 | 30 |
| Chile | 1,513 | 15,635 | 8 |
| Mexico | 0 | 1,172 | 4 |
| U.S. - Idaho | 0 | 279,928 | 0 |
| U.S. - Arizona | 9,004 | 7,032 | 0 |
| India | 0 | 1,438 | 0 |
| U.S. - Georgia | 5,496 | 0 | 0 |
| U.S. - Kentucky | 1,616 | 0 | 0 |
| Subtotal: | 3,050,772 | 4,112,201 | 3,777,402 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| U.S. - Illinois | 28,838,180 | 49,663,060 | 33,903,759 |
| U.S. - Missouri | 5,682,607 | 30,593,710 | 12,789,022 |
| U.S. - Indiana | 2,698,866 | 4,094,695 | 5,844,521 |
| U.S. - Nebraska | 4,577,333 | 2,320,141 | 4,787,771 |
| U.S. - Kansas | 2,060,505 | 6,044,428 | 1,695,519 |
| U.S. - Texas | 665,144 | 729,857 | 941,429 |
| U.S. - Washington | 97,964 | 709,358 | 930,800 |
| U.S. - Maryland | 84,119 | 494,884 | 652,269 |
| U.S. - New Mexico | 195,171 | 137,586 | 181,034 |
| U.S. - Arkansas | 0 | 12 | 127,852 |
| U.S. - Florida | 5,884,350 | 492 | 108,552 |
| U.S. - California | 30,341 | 79,721 | 68,819 |
| U.S. - Kentucky | 77,628 | 56,354 | 64,653 |
| U.S. - Alabama | 35,053 | 78,330 | 42,210 |
| U.S. - North Carolina | 17,857 | 25,978 | 40,763 |
| U.S. - Tennessee | 28,774 | 97,072 | 33,820 |
| Mexico | 28,286 | 43,508 | 18,050 |
| U.S. - Colorado | 14,358 | 5,176 | 16,902 |
| India | 4,944 | 4,144 | 10,536 |
| Oceania (Pacific) | 14,809 | 591 | 8,899 |
| U.S. - Idaho | 10,664 | 10,420 | 4,975 |
| Macedonia | 0 | 9,064 | 3,465 |
| Argentina | 2,982 | 96,519 | 2,562 |
| Puerto Rico | 0 | 611 | 2,125 |
| Croatia | 74 | 92 | 1,325 |
| U.S. - (Mississippi, Arizona, Oregon, Utah), Chile, Guatemala, Paraguay | 1,151 | 1,790 | 2,665 |
| U.S. - Oklahoma | 30,197 | 11 | 56 |
| Spain | 0 | 22,156 | 0 |
| U.S. - Louisiana | 0 | 18,530 | 0 |
| U.S. - Nevada | 3,281 | 84 | 0 |
| France | 46,000 | 0 | 0 |
| Subtotal: | 51,130,638 | 95,338,374 | 62,284,353 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| U.S. - Missouri | 167,283 | 179,995 | 358,776 |
| U.S. - Kansas | 59,304 | 85,207 | 125,015 |
| U.S. - Colorado | 9,977 | 3,017 | 25,814 |
| India | 6,041 | 17,386 | 22,618 |
| U.S. - Nebraska | 45,379 | 97,333 | 4,888 |
| U.S. - California | 1,247 | 251 | 374 |
| Egypt | 0 | 1,444 | 0 |
| U.S. - Oregon | 0 | 289 | 0 |
| U.S. - Texas | 16,919 | 12 | 0 |
| Subtotal: | 306,150 | 384,934 | 537,485 |
| All Exporting Countries | 2006 | 2007 | 2008 |
|---|---|---|---|
| Total: | 54,487,560 | 99,835,509 | 66,599,240 |
Quantity in metric tonnes (TNE)
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| U.S. - Washington | 2,200 | 8,916 | 5,334 |
| U.S. - Illinois | 2,043 | 3,372 | 2,837 |
| Argentina | 0 | 0 | 320 |
| U.S. - Arkansas | 0 | 0 | 162 |
| U.S. - Indiana | 1,243 | 5 | 64 |
| Oceania (Pacific) | - | - | - |
| U.S. - California | 8 | 17 | 10 |
| U.S. - Nebraska | 9,463 | 2,009 | 1 |
| U.S. - Oregon | 3,102 | 0 | 5 |
| U.S. - Oklahoma | 2 | 0 | 3 |
| U.S. - Hawaii | 0 | 0 | 4 |
| U.S. - Colorado, Italy, MacedoniaTable Note * | 4 | 3 | 3 |
| U.S. - Missouri | 0 | 141 | 0 |
| U.S. - Utah | 29 | 20 | 0 |
| Chile | 6 | 11 | 0 |
| Mexico | 0 | 0 | 0 |
| U.S. - Idaho | 0 | 1,225 | 0 |
| U.S. - Arizona | 81 | 27 | 0 |
| India | 0 | 2 | 0 |
| U.S. - Georgia | 141 | 0 | 0 |
| U.S. - Kentucky | 6 | 0 | 0 |
| Subtotal: | 18,328 | 15,748 | 8,743 |
Table Notes
- Table note star
-
Countries or states with an export value of less than $1,000 per year are grouped together and a summed value for all countries and states is shown.
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| U.S. - Illinois | 245,237 | 285,222 | 159,720 |
| U.S. - Missouri | 43,659 | 214,923 | 71,755 |
| U.S. - Indiana | 7,298 | 11,626 | 17,115 |
| U.S. - Nebraska | 41,532 | 15,969 | 22,544 |
| U.S. - Kansas | 17,581 | 38,118 | 9,124 |
| U.S. - Texas | 2,584 | 3,062 | 2,867 |
| U.S. - Washington | 367 | 3,482 | 3,690 |
| U.S. - Maryland | 806 | 3,524 | 3,000 |
| U.S. - New Mexico | 299 | 292 | 521 |
| U.S. - Arkansas | 0 | 0 | 493 |
| U.S. - Florida | 42,794 | 3 | 429 |
| U.S. - California | 207 | 204 | 244 |
| U.S. - Kentucky | 119 | 35 | 33 |
| U.S. - Alabama | 299 | 68 | 35 |
| U.S. - North Carolina | 152 | 192 | 247 |
| U.S. - Tennessee | 93 | 501 | 185 |
| Mexico | 122 | 99 | 105 |
| U.S. - Colorado | 73 | 62 | 58 |
| India | 11 | 16 | 69 |
| Oceania (Pacific) | - | - | - |
| U.S. - Idaho | 102 | 158 | 3 |
| Macedonia | 0 | 10 | 13 |
| Argentina | 42 | 329 | 119 |
| Puerto Rico | 1 | 3 | 11 |
| Croatia | 0 | 0 | 0 |
| U.S. - (Mississippi, Arizona, Oregon, Utah), Chile, Guatemala, Paraguay | 145 | 500 | 146 |
| U.S. - Oklahoma | 181 | 0 | 1 |
| Spain | 0 | 119 | 0 |
| U.S. - Louisiana | 0 | 89 | 0 |
| U.S. - Nevada | 20 | 0 | 0 |
| France | 3,878 | 89 | 365 |
| Subtotal: | 407,602 | 578,695 | 292,892 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| U.S. - Missouri | 674 | 999 | 1,668 |
| U.S. - Kansas | 269 | 384 | 437 |
| U.S. - Colorado | 39 | 9 | 97 |
| India | 22 | 57 | 71 |
| U.S. - Nebraska | 304 | 563 | 24 |
| U.S. - California | 4 | 1 | 0 |
| Egypt | 0 | 5 | 0 |
| U.S. - Oregon | 0 | 1 | 0 |
| U.S. - Texas | 39 | 0 | 0 |
| Subtotal: | 1,351 | 2,019 | 2,297 |
| All Exporting Countries | 2006 | 2007 | 2008 |
|---|---|---|---|
| Total: | 427,281 | 596,462 | 303,932 |
Source: Statistics Canada, 2009
Appendix 14D: Import Data for Maize and Forage seed from countries where Solanum elaeagnifolium is present (2006-2008)
Value in Canadian Dollars ($)
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| U.S. - Missouri | 10,862,986 | 16,151,932 | 23,746,229 |
| U.S. - Indiana | 7,568,819 | 12,419,312 | 17,477,833 |
| U.S. - Illinois | 2,377,380 | 2,247,719 | 5,116,696 |
| U.S. - Nebraska | 259,169 | 289,555 | 789,896 |
| U.S. - Washington | 184,628 | 319,903 | 50,866 |
| U.S. - Idaho | 698,171 | 91,953 | 15,937 |
| U.S. - Colorado | 36,980 | 43,445 | 5,061 |
| U.S. - California | 810 | 13,862 | 536 |
| Chile | 3,458,260 | 13,123,866 | 9,495,021 |
| France | 255,593 | 660,736 | 1,157,153 |
| Argentina | 104,390 | 4,112,304 | 357,611 |
| India | 0 | 0 | 216,397 |
| Subtotal: | 25,807,186 | 49,474,587 | 58,429,236 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| U.S. - Idaho | 4,772,722 | 1,580,344 | 2,799,074 |
| U.S. - Washington | 26,447 | 404,826 | 199,382 |
| U.S. - California | 421,346 | 649,464 | 197,474 |
| U.S. - Tennessee | 7,357 | 62,330 | 19,394 |
| U.S. - Kentucky | 0 | 11,841 | 14,976 |
| U.S. - Missouri | 0 | 1,734 | 2,403 |
| U.S. - Oregon | 50,023 | 1,008 | 1,355 |
| U.S. - North Carolina | 23,119 | 10 | 1,329 |
| Italy | 23,986 | 21,697 | 69,814 |
| France | 27 | 2,529 | 52,220 |
| Subtotal: | 5,325,027 | 2,735,783 | 3,357,421 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| U.S. - Oregon | 1,206,471 | 1,338,669 | 836,195 |
| U.S. - Idaho | 223,065 | 287,751 | 117,948 |
| U.S. - Alabama | 1,862 | 68,736 | 72,247 |
| U.S. - Mississippi | 0 | 0 | 40,485 |
| U.S. - Washington | 44,762 | 8,334 | 25,275 |
| U.S. - California | 6 | 56,460 | 22,911 |
| U.S. - Kentucky | 11,530 | 1,430 | 7,626 |
| Chile | 688,426 | 599,468 | 1,229,059 |
| Uruguay | 0 | 128,656 | 178,637 |
| Argentina | 43,769 | 91,082 | 146,970 |
| Subtotal: | 2,219,891 | 2,580,586 | 2,677,353 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| U.S. - Oregon | 1,925,464 | 1,898,365 | 2,722,623 |
| U.S. - Washington | 7,210 | 26,523 | 46,936 |
| U.S. - Idaho | 19,668 | 88,387 | 33,445 |
| U.S. - Colorado | 0 | 55,503 | 4,549 |
| Italy | 0 | 4,284 | 37,816 |
| France | 0 | 10,613 | 3,606 |
| Subtotal: | 1,952,342 | 2,083,675 | 2,848,975 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| U.S. - Oregon | 4,181,870 | 4,755,449 | 7,836,065 |
| U.S. - Washington | 3,130,349 | 2,808,083 | 4,252,270 |
| U.S. - Idaho | 1,667,490 | 1,532,595 | 2,049,030 |
| U.S. - Illinois | 375,617 | 385,992 | 260,592 |
| Subtotal: | 9,355,326 | 9,482,119 | 14,397,957 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| U.S. - Oregon | 5,256,248 | 3,814,246 | 6,626,765 |
| U.S. - Washington | 2,779 | 2,974 | 85,305 |
| U.S. - Idaho | 54,674 | 31,876 | 77,718 |
| U.S. - Arizona | 33 | 2,242 | 12,125 |
| U.S. - Illinois | 1,595 | 15,119 | 1,116 |
| U.S. - California | 11,092 | 0 | 288 |
| U.S. - Colorado | 0 | 10,474 | 0 |
| France | 0 | 15,020 | 15,044 |
| Argentina | 0 | 17,242 | 0 |
| Uruguay | 71,052 | 55,110 | 0 |
| Italy | 61,300 | 32,132 | 0 |
| Subtotal: | 5,458,773 | 3,996,435 | 6,818,361 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| U.S. - California | 33,579 | 0 | 0 |
| Subtotal: | 33,579 | 0 | 0 |
| All Exporting Countries | 2006 | 2007 | 2008 |
|---|---|---|---|
| Total: | 50,152,124 | 70,353,185 | 88,529,303 |
Quantity in Kilograms (kg)
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| U.S. - Missouri | 6,874,000 | 10,986,000 | 12,136,000 |
| U.S. - Indiana | 2,585,000 | 2,934,000 | 4,025,000 |
| U.S. - Illinois | 604,000 | 403,000 | 922,000 |
| U.S. - Nebraska | 137,000 | 64,000 | 166,000 |
| U.S. - Washington | 54,000 | 100,000 | 36,000 |
| U.S. - Idaho | 62,000 | 158,000 | 3,000 |
| U.S. - Colorado | 14,000 | 56,000 | 1,000 |
| U.S. - California | 0 | 1,000 | 0 |
| Chile | 1,251,000 | 3,320,000 | 3,664,000 |
| France | 3,857,000 | 89,000 | 365,000 |
| Argentina | 38,000 | 87,000 | 117,000 |
| India | 0 | 0 | 52,000 |
| Subtotal: | 15,476,000 | 18,198,000 | 21,487,000 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| U.S. - Idaho | 955,171 | 327,406 | 440,995 |
| U.S. - Washington | 5,457 | 80,165 | 45,398 |
| U.S. - California | 114,149 | 166,799 | 43,479 |
| U.S. - Tennessee | 1,814 | 12,412 | 2,653 |
| U.S. - Kentucky | 0 | 1,999 | 1,950 |
| U.S. - Missouri | 0 | 272 | 465 |
| U.S. - Oregon | 21,900 | 198 | 134 |
| U.S. - North Carolina | 5,936 | 3 | 181 |
| Italy | 6,013 | 3,421 | 21,212 |
| France | 19 | 180 | 6,335 |
| Subtotal: | 1,110,459 | 592,855 | 562,802 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| U.S. - Oregon | 387,648 | 421964 | 238341 |
| U.S. - Idaho | 67,994 | 83663 | 46156 |
| U.S. - Alabama | 532 | 22256 | 20573 |
| U.S. - Mississippi | 0 | 0 | 11036 |
| U.S. - Washington | 8,625 | 2744 | 6806 |
| U.S. - California | 2 | 10343 | 4990 |
| U.S. - Kentucky | 3,087 | 453 | 1951 |
| Chile | 256,960 | 239676 | 374500 |
| Uruguay | 0 | 45000 | 42500 |
| Argentina | 17,900 | 33300 | 41300 |
| Subtotal: | 742,748 | 859,399 | 788,153 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| U.S. - Oregon | 975900 | 921911 | 1270884 |
| U.S. - Washington | 2583 | 9628 | 14786 |
| U.S. - Idaho | 7257 | 28563 | 12435 |
| U.S. - Colorado | 0 | 19894 | 1091 |
| Italy | 0 | 1800 | 22665 |
| France | 0 | 5209 | 1515 |
| Subtotal: | 985,740 | 987,005 | 1,323,376 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| U.S. - Oregon | 1,468,614 | 1822725 | 2654033 |
| U.S. - Washington | 2,010,877 | 1639238 | 1943822 |
| U.S. - Idaho | 539,623 | 519804 | 671617 |
| U.S. - Illinois | 81,838 | 62421 | 68600 |
| Subtotal: | 4,100,952 | 4,044,188 | 5,338,072 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| U.S. - Oregon | 3,491,358 | 2485844 | 5227573 |
| U.S. - Washington | 1,460 | 1665 | 57564 |
| U.S. - Idaho | 32,926 | 22248 | 37847 |
| U.S. - Arizona | 11 | 550 | 1633 |
| U.S. - Illinois | 463 | 20098 | 1330 |
| U.S. - California | 14,399 | 0 | 192 |
| U.S. - Colorado | 0 | 7512 | 0 |
| France | 0 | 3968 | 3431 |
| Argentina | 0 | 24990 | 0 |
| Uruguay | 94,085 | 90693 | 0 |
| Italy | 21,000 | 7500 | 0 |
| Subtotal: | 3,655,702 | 2,665,068 | 5,329,570 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| U.S. - California | 12,391 | 0 | 0 |
| Subtotal: | 12,391 | 0 | 0 |
| All Exporting Countries | 2006 | 2007 | 2008 |
|---|---|---|---|
| Total: | 26,083,992 | 27,346,515 | 34,828,973 |
Source: Statistics Canada, 2009
Note: Only those countries having an import value greater or equal to $10,000 in any year between 2006 and 2008 are included in this table.
Appendix 14E: Import Data for Hay and straw from countries where Solanum elaeagnifolium is present (2006-2008)
Value in Canadian Dollars ($)
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| Washington | 5,056,544 | 5,685,096 | 6,397,081 |
| India | 284,039 | 258,851 | 302,025 |
| North Carolina | 272,458 | 316,202 | 212,617 |
| Arkansas | 54,818 | 50,870 | 173,146 |
| Illinois | 40,714 | 114,980 | 111,185 |
| New Mexico | 9,709 | 28,495 | 23,099 |
| Oregon | 4,875 | 1,750 | 18,492 |
| Florida | 1,348 | 5,068 | 9,550 |
| Maryland | 3,151 | 7,596 | 8,908 |
| Oklahoma | 0 | 3,347 | 2,079 |
| California | 2,472 | 501 | 597 |
| Georgia | 0 | 60,000 | 0 |
| Arizona | 1,365 | 885 | 0 |
| Indiana | 5,682 | 0 | 0 |
| Subtotal: | 5,737,175 | 6,533,641 | 7,258,779 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| Washington | 3,849,245 | 4,045,927 | 4,115,211 |
| Oregon | 1,086,048 | 630,851 | 330,770 |
| Nebraska | 29,892 | 24,792 | 48,591 |
| Kansas | 0 | 13,410 | 46,197 |
| Chile | 0 | 41,354 | 32,946 |
| Colorado | 32,133 | 26,652 | 28,890 |
| Kentucky | 619 | 30 | 15,743 |
| California | 12,740 | 23,511 | 9,755 |
| Nevada | 5,128 | 0 | 4,135 |
| Arizona | 134,345 | 131,764 | 3,429 |
| Missouri | 67 | 3,685 | 2,935 |
| Illinois | 774 | 10 | 1,728 |
| India | 2,933 | 28,297 | 76 |
| Idaho | 6,317 | 55,099 | 53 |
| Indiana | 1,546 | 8 | 1 |
| Oceania | 0 | 19,132 | 0 |
| France | 19 | 2,014 | 0 |
| Subtotal: | 5,161,806 | 5,046,536 | 4,640,460 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| Total: | 10,898,981 | 11,580,177 | 11,899,239 |
Source: Statistics Canada, 2009
Appendix 15A: Pest Risk Assessment Summary for Zygophyllum fabago (Syrian bean-caper)
- Identity of Organism
- Organism Status
- Current Regulatory Status
- Probability of Entry
- Probability of Establishment
- Probability of Spread
- Potential Economic Consequences
- Potential Environmental and Social Consequences
- Uncertainty
- Conclusion
- Technical Issues for Consideration
Identity of Organism
Name: Zygophyllum fabago
Synonyms: None found
English common names: Syrian bean-caper, Syrian beancaper
French common names: Fabagelle
Description: Zygophyllum fabago is a much-branched herbaceous plant with a deep, well-developed tap root. The plants grow almost 1 m tall and wide and are bushy. The succulent leaves are opposite and compound with a single pair of leaflets each 1 to 3 cm in length. The whitish to yellow flowers are borne in the leaf axils. The fruit is a five-valved capsule with a single seed in each valve. The capsules are oblong, and cylindrical and five sided (Davison and Wargo 2001; Robbins et al. 1951).
Zygophyllum fabago has the ability to dominate a site and eliminate the native vegetation. It grows on disturbed sites such as roadsides, corrals, and gravel pits. It can form large dense colonies that exclude native plants and animals. The thick, waxy leaves allow the plants to survive long periods of drought and the extensive root system provides a competitive advantage over native species. It is considered unpalatable to livestock (Davison and Wargo 2001).
The species reproduces by seed and spreading roots. Root fragments can produce new plants. The stems die back to the ground each winter. In very cold areas it can function as an annual, with new plants growing from seed each year (Davison and Wargo 2001).
Organism Status
Zygophyllum fabago is not reported to occur in Canada, and no evidence was found that it is cultivated in Canada (CFIA 2008; CNLA 2009). Based on this information, it is considered absent from the PRA area.
Current Regulatory Status
Zygophyllum fabago is not currently regulated in Canada. It is not regulated as a federal noxious weed in the U.S., but is regulated in the following states: California, Idaho, Nevada, Oregon and Washington (USDA-ARS 2009; USDA-NRCS 2009).
Probability of Entry
Zygophyllum fabago is thought to have been imported to the United States in contaminated alfalfa seed. There is a possibility that it may have escaped from gardens as the flower buds are used as a caper substitute in its native range (Davison and Wargo 2001).
| Type of pathway | Specific pathways |
|---|---|
| Natural dispersal | Zygophyllum fabago spreads primarily by seeds (Davison and Wargo, 2001). No information could be found to document how seeds spread in natural situations. This pathway is unlikely given that there are no known documented occurrences in counties adjacent to the Canadian border. The species is reported as possible in Okanogan County, Washington, however there are no supporting collections (Wooten 2010). |
| Intentional introduction | Some websites list seeds of this species for sale as a medicinal herb. It is thought that it might have escaped from gardens as the flower buds are used as a caper substitute in the native range (Davison and Wargo 2001). Intentional introduction for planting is the most likely pathway for entry into Canada. It is available for sale on-line, but it is difficult to know how much interest there is. |
| Unintentional introduction | The species is "thought to have been imported to the United States at the turn of the century in contaminated alfalfa seed" (Davison and Wargo 2001). This pathway is unlikely, unless seed is imported from the native range. The species is not a common weed of agricultural fields. Root fragments can sprout to form new plants (Davison and Wargo 2001). It is possible that they could be transported on equipment. This is certainly a possibility, but it could be reduced as a pathway by keeping soil from entering the country. |
Probability of Establishment
Zygophyllum fabago is native to Afghanistan, Iran, Iraq, Israel, Jordan, Lebanon, Syria, Türkiye, Armenia, Azerbaijan, Georgia, Ciscaucasia, Turkmenistan, Pakistan, Russian Federation, Ukraine and Romania (USDA-ARS 2009). It is introduced in southern Europe (France, Spain, Sardinia) (Pankhurst 1998), North America (western U.S.) (USDA-NRCS 2009), Puerto Rico and Australia (Western Australia and New South Wales) (Randall 2007).
Within the western U.S., there are populations in 6 counties in Washington and 5 counties in Idaho. It has not yet been reported from Oregon and all infestations in California are thought to have been eradicated (Karl et al. 1996). There are reports from a number of other states (see figure 1). However, the record for Pennsylvania is based on an old collection at ballast dumps at Philadelphia harbour which has never been repeated (Ruiz and Carlton 2003). As the species is not included in any of the major northeastern flora, it is very likely that the New York record is also based on ephemeral populations at a port location (Fernald 1950; Gleason 1968; Gleason and Cronquist 1963).
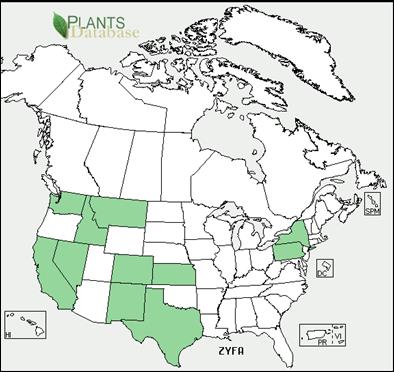
Description for Figure 1:
This map shows the range of Zygophyllum fabago (Syrian bean-caper) in North America indicating its presence through the use of the colour green. The states covered in green are: New York, Pennsylvania, Texas, Kansas, Colorado, New Mexico, Montana, Idaho, Washington, Nevada and California
Source: USDA-NRCS 2009
The current range in the U.S. suggests that the species would be hardy to NAPPFAST zone 5. However, persistent Zygophyllum fabago populations are restricted to dry regions, including deserts (Davison and Wargo 2001), so the potential range as an invasive weed species is limited to southern B.C. in Canada (see figure 2). It should be noted that the coastal parts of the range on the map are probably too wet to be suitable for this species.
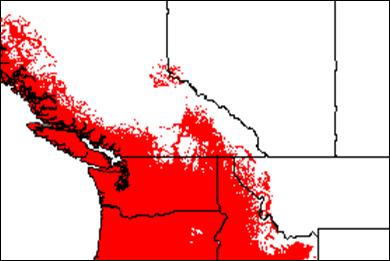
Description for Figure 2:
This image shows the potential range of Zygophyllum fabago (Syrian bean-caper) in Canada, more specifically in British Columbia. Red is used to indicate the regions in which Zygophyllum fabago at risk according to the Canadian Plant Hardiness Zones map, in this case NAPPFAST Hardiness Zones 5-9. In Canada, the potential range of Zygophyllum fabago is limited to southern British Columbia where it has a scattered presence. This image is narrowed to include parts of the U.S. more specifically Washington, Oregon, and Idaho where the red has more of a prevalent presence and is heavily condensed downwards beyond the image.
NAPPFAST zones 5-9.
Probability of Spread
On a local scale, plants can spread by root fragments, but the usual means of dispersal is by seeds (Davison and Wargo 2001). The seeds of Zygophyllum speciesare mucilaginous (Beier et al. 2003), which might allow them to attach to animals or people for distribution, but this does not seem to be documented.
Potential Economic Consequences
The biggest economic threat from Zygophyllum fabago is to ranchers. The plants can form dense masses that displace beneficial species on rangelands. Control with herbicides is difficult because of the waxy leaf surfaces and because of the extensive root system. The plants are not palatable to livestock (Anonymous 2007), so infestations reduce the amount of useful forage available to browsers. Herbicides have shown some promise in controlling this weed, but repeated applications are necessary because of the thick, waxy leaves (Davison and Wargo 2001). In the dry rangelands invaded by this species, it is unlikely that control with herbicides would be economically feasible.
Potential Environmental and Social Consequences
Zygophyllum fabago has the ability to dominate native vegetation in suitable dry habitats, being described as "almost as invasive as" Peganum harmala (African-rue) and Tribulus terrestris (puncture vine), which are related weed species (Davison and Wargo 2001).
Uncertainty
There is not a great deal of information available on the biology and impacts of this species. So far, its distribution and impacts have been limited. In particular, there is almost no information about seed dispersal.
Conclusion
Based on the outcome of this pest risk assessment, Zygophyllum fabago is likely to establish and become invasive in parts of Canada, including southern B.C., if it is introduced to these areas. This plant should be considered for regulation under the Plant Protection Act and Seeds Act.
Technical Issues for Consideration
There should be no difficulties in detection and identification of seeds or plants of this species. Root pieces might be more difficult to identify.
Appendix 15B: Risk Management Considerations for Zygophyllum fabago (Syrian bean-caper)
- Potential Mitigation Measures for Natural Means of Dispersal
- Potential Mitigation Measures for Intentional Introduction Pathways
- Potential Mitigation Measures for Non-Intentional Introduction Pathways
Potential Mitigation Measures for Natural Means of Dispersal
As described in Section V, natural dispersal is a possible pathway; however there is a high level of uncertainty regarding this. The species is regulated in Washington and Idaho and populations have been controlled. Therefore, risk mitigation measures will not be suggested.
Potential Mitigation Measures for Intentional Introduction Pathways
Plants for Planting Excluding Seed
Intentional introduction for planting is the most likely pathway for entry of Zygophyllum fabago into Canada, as it can be used as a medicinal herb or as a flavoring.
Previous imports
The CFIA requires a Permit to Import, with the scientific and common names, for all plants and propagative material from origins other than the continental U.S. (CFIA 2008, 2009). Some plants from the continental U.S. also require a Permit to (CFIA 2009).
Based on the information available in the CFIA's Import Permit System, Import Retrieval System, and information compiled at the CFIA's Import Service Centres, Zygophyllum fabago has not been recently imported into Canada.
Potential risk mitigation measures
Non-regulatory measures:
- a) Use sterile cultivars if they exist. The efficacy of this measure is unknown. The CFIA has never received an application to import sterile cultivars of Zygophyllum fabago.
- b) Encourage voluntary cessation of the sale of Zygophyllum fabago. Voluntary cessation is not effective by itself, but could support other measures.
- c) Increase public awareness of the risk posed by this plant, publish a factsheet online, and distribute awareness material to garden centers, botanical gardens, gardeners associations, horticulture industry groups, etc. This measure is not considered effective by itself, but could support other measures.
Regulatory measures:
- d) Allow sale of Zygophyllum fabago with special conditions such as not to be grown close to natural areas and mandatory control of adventives. Allowing sale with special conditions is not considered effective because once the plant is grown in private gardens, CFIA does not have adequate resources to monitor.
- e) Regulate Zygophyllum fabago under the Plant Protection Act as a quarantine pest in order to:
- Prohibit the importation of plants of Zygophyllum fabagoFootnote 1.
- Require importers of material to specify the scientific name when applying for a Permit to Import plant and propagative material.
- Require the scientific name to be provided on the Phytosanitary Certificate for all plants exported to Canada.
For species intended for large scale cultivation in Canada:
- f) Zygophyllum fabago can proliferate in coarse mineral substrates contaminated with heavy metals and studies are being conducted to assess its potential use in phytoremediation (Lefèvre et al. 2006). Nothing indicates that Zygophyllum fabago is currently cultivated in Canada, but there may be an interest in the future.
- g) The commercial cultivation of Zygophyllum fabago as a crop requires conditions in order to mitigate risks to food, feed, or other environmental risks. Example conditions include geographic limitations or reproductive isolation. Regulation of Zygophyllum fabago as a New Crop under Part V of the Seeds Regulations could be explored.
Trade Implications
- If Zygophyllum fabago is regulated under the Plant Protection Act, no market losses are expected since there is no known current trade of this species in Canada.
- Regulation of Zygophyllum fabago under the Plant Protection Act could facilitate the export of Canadian greenhouse and nursery plants, seed and grain to countries that currently or will in the future prohibit Zygophyllum fabago by maintaining country/area freedom.
Cost-effectiveness and Feasibility
- If Zygophyllum fabago is regulated, additional staff would not be required at the CFIA Plant Health Permit Office to regulate the species. Applications for Permits to Import are reviewed by officers on a regular basis. Enforcement of the prohibition to import would require importers to specify full scientific names on their applications for permits to import, instead of only the genus name, as is often the case currently. This requirement is already in place (see CFIA Directive 97-04; CFIA 2008), but Permit Office staff may need to spend some time communicating this requirement to importers.
- CFIA Plant Health inspection staff will need to be trained to identify Zygophyllum fabago.
Seed
Previous imports
Websites list seeds of this species for sale as a medicinal herb (CFIA, 2009), but it is difficult to know the level of interest in the plant in Canada.
Potential risk mitigation measures
Regulate Zygophyllum fabago as a prohibited noxious weed (Class 1) under the Weed Seeds Order of the Seeds ActFootnote 2.
- This species meets the definitions for Class 1Footnote 3 species under the Weed Seeds Order.
- All imported and domestic seed lots must be free of prohibited noxious weed seeds. Imported seed lots require a certificate of analysis stating Zygophyllum fabago is absent from the seed lot before it can be imported.
Regulate as a quarantine pest under the Plant Protection Act and add this species to the List of Pests Regulated by Canada (CFIA 2009) in order to:
- prevent the importation, movement, and cultivation of this species in Canada. Currently, seed of many new crop species, such as field crops, can be imported without a Permit to Import or a Phytosanitary CertificateFootnote 4.
- enable inspectors to take appropriate action for the purposes of eradicating the pest or preventing its spreadFootnote 5.
Regulatory actions under the Plant Protection Act could include the following:
- Prohibit importation of Zygophyllum fabago. The only exceptions would be for the importation of devitalized seed and the importation of seed or preserved specimens for scientific research purposes by recognized herbaria and research facilities.
- Require importers of plant material from the non-continental U.S. to apply for a Permit to Import with scientific name specified.
- Seed of horticultural plants is not within the scope of CFIA's draft directive D-08-04 on plants for planting (2008). Therefore, phytosanitary requirements will be specified under a new regulatory directive or D-08-04 will be revised.
Sufficient information about the use of Zygophyllum fabago as a potential crop does not exist. If the proponent, located in Canada, needs to collect more information about the plant (e.g. to generate data for a determination of environmental safety), then confined research field trials under Part V of the Seeds Regulations could be authorized by the Plant Biosafety Office.
Trade Implications
- If Zygophyllum fabago seeds are regulated under the Weed Seeds Order of the Seeds Act, no market losses are expected since there is currently no known trade of Zygophyllum fabago seeds in Canada.
- There may be an impact on the importation of other Zygophyllum spp. as foreign NPPOs will need to identify Zygophyllum fabago seeds as well as any other Zygophyllum species listed on Phytosanitary Certificates.
Cost-effectiveness and Feasibility
- The CFIA Seed Program is already in place to prevent the entry of prohibited noxious weeds.
- There should be no difficulties in detection and identification of seeds (Allison 2009).
Potential Mitigation Measures for Non-intentional Introduction Pathways
Seed
Zygophyllum fabago is "thought to have been imported to the United States at the turn of the century in contaminated alfalfa seed" (Davison and Wargo 2001).
Previous imports
- The Seed Science and Technology Section of the CFIA Saskatoon laboratory maintains a record of contaminants found through marketplace monitoring of domestic and imported seed. Zygophyllum fabago has not been recorded in imported or Canadian seed samples.
- From 2006-2008, an average of approximately $3.9 million and 779,000 kilograms per year of alfalfa seed was imported from countries where Zygophyllum fabago is present (Table 33; Statistics Canada 2009). This represents approximately 78% of total alfalfa seed imported between 2006 and 2008 (Industry Canada, 2009).
Import data for Alfalfa seed originating from countries where Zygophyllum fabago (Syrian beancaper) is present.
Although a large percentage of imported alfalfa seed originates from areas where Zygophyllum fabago is present, the level of risk associated with this pathway is relatively low since the species is not a common weed of agricultural fields (CFIA 2009). No imports are coming from countries in the weed’s native range, which would be the most likely source of contaminated alfalfa seed.
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| Australia | 208,607 | 234,443 | 198,929 |
| France | 27 | 2,529 | 52,220 |
| Spain | 0 | 3,662 | 0 |
| U.S. - Idaho | 4,772,722 | 1,580,344 | 2,799,074 |
| U.S. - California | 421,346 | 649,464 | 197,474 |
| U.S. - Washington | 26,447 | 404,826 | 199,382 |
| U.S. - Montana | 7,709 | 10,583 | 18,097 |
| Total: | 5,436,858 | 2,885,851 | 3,465,176 |
| Exporting Country | 2006 | 2007 | 2008 |
|---|---|---|---|
| Australia | 69,571 | 41,718 | 31,372 |
| France | 19 | 180 | 6,335 |
| Spain | 0 | 600 | 0 |
| U.S. - Idaho | 955,171 | 327,406 | 440,995 |
| U.S. - California | 114,149 | 166,799 | 43,479 |
| U.S. - Washington | 5,457 | 80,165 | 45,398 |
| U.S. - Montana | 971 | 2,158 | 5,275 |
| Total: | 1,145,338 | 619,026 | 572,854 |
Source: Statistics Canada 2009
Potential risk mitigation measures
Regulate Zygophyllum fabago as a prohibited noxious weed (Class 1) under the Weed Seeds Order of the Seeds Act Footnote 6.
- This species meets the definitions for Class 1 Footnote 7 species under the Weed Seeds Order.
- All seed lots sold or imported into Canada must be free of prohibited noxious weed seeds. Imported seed lots would require a certificate of analysis stating Zygophyllum fabago is absent from the seed lot before it can be imported.
Regulate Zygophyllum fabago as a quarantine pest under the Plant Protection Act. and add this species to the List of Pests Regulated by Canada (CFIA 2009) in order to:
- prevent the importation, movement, and cultivation of this species in Canada.
- enable inspectors to take appropriate action for the purposes of eradicating the pest or preventing its spreadFootnote 8.
Regulatory actions under the Plant Protection Act could include one or more of the following:
- Exporters may be required to provide a Phytosanitary Certificate with or without an Additional Declaration stating freedom from Zygophyllum fabago.
- Requirement for seed from infested areas to be allowed entry into Canada only after the importer obtains a CFIA issued import permit indicating specific import requirements and conditions for pest status, handling and use of the commodity. Prior to permit issuance, a facility inspection by CFIA and verification of the importer's ability to meet permit requirements may be required.
- Recognition of foreign government phytosanitary noxious weed certification in countries or states of origin may allow for additional phytosanitary requirements to be waived.
Trade Implications
- Regulation of Zygophyllum fabago under the Seeds Act and Plant Protection Act will safeguard seed trade with the five U.S. states (Idaho, California, Nevada, Oregon and Washington) where Zygophyllum fabago is regulated as a noxious weed. Canada exported approximately $23.7 million worth of alfalfa seed in 2008 to these states (Industry Canada, 2009).
- Exporting countries currently devote resources towards inspection of seed lots and issuance of Phytosanitary Certificates. Laboratories in foreign countries will need to be able to identify seed of Zygophyllum fabago within a seed sample. Exporters will need to ensure freedom of Zygophyllum fabago in seed lots, otherwise CFIA can refuse import.
- There is potential for reduction and or loss of some import markets for Canada if exporting countries are unable to meet proposed phytosanitary requirements.
Cost-effectiveness and feasibility
- The CFIA Seed Program is already in place to prevent the entry of prohibited noxious weeds. The CFIA monitors compliance with the Canadian standards through the Marketplace Monitoring Program Footnote 9.
- The seeds of Zygophyllum fabago are easily identified by trained seed analysts (Allison 2009).
Vehicles and Used Farm Machinery
Root fragments can sprout to form new plants (Davison and Wargo, 2001). It is possible that root fragments could be transported on equipment. Since Zygophyllum fabago is not a common weed of agricultural fields, this is considered a low-risk pathway.
Previous imports
- A considerable volume of vehicles cross the U.S.-Canada border every year.
- Information is not available on the volume of imports of used farm machinery.
Potential Risk mitigation measures
Enforcement of the Directive 95-26: "Phytosanitary requirements for soil and related matter, alone or in association with plants" (CFIA, 2008).
In 2003, the Canada Border Services Agency (CBSA) assumed responsibility for the initial import inspection services in respect to the Acts and Regulations administered by the CFIA to the extent that they are applicable at Canadian border points. The inspection of goods that may be contaminated with soil are among the responsibilities that were transferred to the CBSA in 2003. The Food, Plant and Animals Programs Section of the CBSA has developed Standard Operating Procedures (SOP) concerning the "Inspection of Imported Goods Potentially Contaminated with Soil." This SOP provides the CBSA's Border Services Officers with formal procedures for the inspection and disposition of goods that may be contaminated with soil, including used agricultural machinery and vehicles.
Appendix 16: Consultation
The following stakeholders were consulted:
- Federal agencies or official organizations
- Provincial departments
- Industry
- Other organizations including but not limited to:
- Invasive Plants Councils
- Academia
- None governmental associations
Significant support for this regulatory decision was received through responses from provincial and federal departments and other stakeholders. Suggested comments and revisions were included in this document where appropriate.





















