List of ingredients and allergens on food labels
On this page
- Requirements
- Manner of declaring
- Legibility and location
- Artificial flavour
- Food additives in the list of ingredients
- Information letters
- Annex 1: examples of sugars-based ingredients that require grouping
- Annex 2: examples of ingredients for which grouping with sugars is not required
- Definitions
Requirements
General
List of ingredients
All prepackaged products with more than 1 ingredient must declare their ingredients and components in a list of ingredients [B.01.008(1), FDR], unless they are exempt. For more information, refer to Exemptions.
Ingredients must be declared in descending order of proportion by weight, as determined before they are combined to make the food [B.01.008.2(3)(a), FDR].
Food allergens, gluten and added sulphites must be declared, provided there are no exemptions or exceptions. Added sulphites at levels of 10 ppm or more, and that are not already required to be shown in the list of ingredients, must be declared by their prescribed source names. A cross-contamination statement may be declared on the label of a prepackaged food when, despite all reasonable measures, there is the unintended presence of food allergens in the food.
The Food and Drug Regulations (FDR) specify the manner in which ingredients and components must be declared, including grouping sugars-based ingredients, common names, and declaring food allergens, gluten and added sulphites. For further details, refer to Manner of declaring.
Exemptions
Prepackaged products that do not require a list of ingredients
The following products are exempt from declaring a list of their ingredients:
- Prepackaged products that are packed on retail premises from bulk, including candies, flour, salt, spices, single ingredient nuts and in store baked breads.
This exemption does not apply to mixed nuts and to uncured meat and poultry products that contain phosphate salts and/or water and are packaged at retail [B.01.008(2)(a), B.01.091, FDR]- "prepackaged from bulk" includes any product made on the retailer's premises (for example, assembled, baked, transformed) and any product packaged by the retailer from the supplier's containers
- "packed on the retail premises" includes
- foods packaged in the retail establishment where the prepackaged food is sold
- foods packaged on premises (on or off-site) owned by the same person as the retail establishment where the food is sold, as long as these premises supply only that retail establishment
- products sold by a single retailer at more than 1 building on the same premises would be considered to be sold in a single retail establishment (for example, products packed in a retail store and sold at both that store and in a gas bar at the same address that is owned by the same person would be exempt)
Prepackaged food does not qualify for the exemption when it is packed at off-site premises that are owned by the same person as the retail establishment where the food is sold, but these premises supply more than 1 establishment or company, or sell the product directly to consumers (for example, product packed at a warehouse or packing house that supplies multiple stores would not be exempt).
- Prepackaged individual portions of food intended solely to be served with meals or snacks by restaurants, airlines [B.01.008(2)(b), FDR]
- for example, coffee creamers, jam, tartar sauce, pancake syrup, salad dressing, crackers and ketchup
- Prepackaged individual servings of food prepared by commissaries and sold in mobile canteens or vending machines [B.01.008(2)(c), FDR]
- for further details on commissaries, refer to Date markings
- Prepackaged meat and meat by-products that are barbecued, roasted or broiled on the retail premises [B.01.008(2)(d), FDR]
- for example, spare ribs that have been barbequed on the retail premises
- Prepackaged poultry and poultry meat or poultry by-products that are barbequed, roasted or broiled on the retail premises [B.01.008(2)(e), FDR]
- for example, chicken roasted in a grocery store
- Bourbon whiskey and standardized alcoholic beverages [B.01.008(2)(f), FDR]
- for example, whisky, rum, vodka, gin, wine, liquors and beer
- unstandardized alcoholic beverages like cream liquors, shandies, coolers, and cocktails require a complete list of ingredients
- Standardized vinegars [B.01.008(2)(g), FDR]
- for example, wine vinegar, white vinegar, malt vinegar, cider vinegar and blended vinegar
- unstandardized vinegars require a complete list of ingredients, for example, raspberry vinegar
Prepackaged products that are not required to declare allergens, gluten and/or added sulphites
Food allergens, gluten and added sulphites do not have to be declared when present in prepackaged products that are exempt from bearing a list of ingredients under B.01.008(2)(a) to (e) of the FDR (which are also specified in items 1 to 5 above), unless a list of ingredients is voluntarily shown on the product's label [B.01.010.1(4), B.01.010.2(4), FDR]. This exemption, however, does not apply to distilled vinegars or standardized alcoholic beverages regardless of the fact that they are exempt from bearing a list of ingredients under B.01.008(2)(f) and (g) of the FDR.
Triggers to provide allergen labelling on otherwise exempted products
If a manufacturer chooses to voluntarily declare a list of ingredients on the product label of any product exempt from bearing a list of ingredients, then the prescribed source names for allergens, gluten and added sulphites that are present must be declared in the list of ingredients [B.01.010.1(2), B.01.010.2(3), FDR].
In addition, if a prepackaged product that is exempt from the requirement to carry a label voluntarily carries a label with a list of ingredients, the allergen labelling requirements apply.
Exceptions
Exceptions from allergen declarations
Vintage wines
Although standardized alcoholic beverages are not exempt from declaring allergens, Health Canada has developed a position on the labelling requirements for vintage wines. This position indicates that the allergen labelling regulations apply to all non-vintage wines and vintage wines with a year date of 2012 and later, and that vintage wines with a year date of 2011 or earlier can continue to be sold with their original labels. For more information on this subject, refer to Health Canada's position on Vintage wine and application of enhanced allergen regulations.
Foods with temporary marketing authorization letters
Some products that were previously classified as natural health products are now classified as foods by Health Canada. Health Canada has issued temporary marketing authorization letters to grant these products a transition period to bring labels into compliance with food labelling requirements.
For more information on this subject, refer to Health Canada's Lists of foods that have received temporary marketing authorization letters.
Highly refined oils
Based on a detailed assessment by Health Canada, highly refined (degummed, neutralized, bleached and deodorized) oils derived from food allergen sources are not subject to the enhanced labelling requirements as the refining process has been determined to remove the allergenic protein from the oil. Based on the available science, the very low levels of protein present within highly refined oils are not considered to pose a risk to the health of individuals with food allergies. For more information on this subject, refer to Health Canada's position on highly refined oils derived from food allergen sources.
Manner of declaring
Ingredients
Order of ingredients
Ingredients must be declared by their common name in descending order of their proportion by weight of the prepackaged product. The order must be the order of the ingredients before they are combined to form the prepackaged product. In other words, based on what was added to the mixing bowl [B.01.008.2(3)(a), FDR].
The following ingredients, however, can be listed at the end of the ingredients list in any order [B.01.008.2(4), FDR]:
- spices and herbs
- seasoning ingredients, other than spices, herbs and salt added separately, if the total collective weight of these seasonings is no more than 2% of the total weight of all ingredients used in the manufacture of the product, excluding added water used as an ingredient that has been removed during the manufacture of the product
Note: to calculate this percentage,
- the total collective weight of these seasonings does not include the weight of spices, herbs and salt that are added as separate ingredients to the prepackaged product
- the total weight of all ingredients excludes the weight of any added water used as an ingredient that has been removed or lost during the manufacture of the product
- natural and artificial flavours
- flavour enhancers
- food additives, except ingredients of food additive preparations or mixtures of substances for use as a food additive
- vitamins and their salts or derivatives of vitamins
- mineral nutrients and their salts
Sugars
Sugars-based ingredients are required to be grouped within the list of ingredients following the term "Sugars" [B.01.008.3(1), FDR]. For more information, refer to Grouping sugars-based ingredients.
Ingredients that generally do not have to be declared
When present in a prepackaged product, the following ingredients and their components are not required to be declared in the list of ingredients, unless they contain known allergens, gluten, or added sulphites at quantities greater than or equal to 10 parts per million. Refer to Food allergen, gluten and added sulphite declaration for exceptions.
- Wax coating compounds, their components and other protective edible coatings are not required to be shown on the labels of prepackaged fresh fruits or fresh vegetables as an ingredient or a component [B.01.008(3)(a), FDR]
Apples, turnips and cucumbers are examples of fruits and vegetables that have wax coatings - Sausage casings are not required to be shown on the labels of prepackaged sausages as an ingredient or as a component [B.01.008(3)(b), FDR]
However, the declaration of a natural casing is required if it is of a different species than the meat ingredients used in the sausage. For more information, refer to Sausages wrapped in natural casings - Hydrogen, when used for hydrogenation purposes, is not required to be shown on the labels of any prepackaged products as an ingredient or component [B.01.008(3)(c), FDR]
- Added water used as an ingredient that has been removed during the manufacture of the prepackaged product [B.01.008(3)(e), FDR]
Components
Order of components
Components (ingredients of ingredients) must be declared by their common name as part of the list of ingredients. They can be declared in 1 of 2 ways:
- 1) in parentheses following the ingredient common name in descending order of proportion by weight in the ingredient [B.01.008.2(5)(b), FDR]
For example:
Ingredients: Tomato paste (tomatoes, salt, benzoic acid) • Sugar • Modified corn starch • Lemon juice from concentrate (water, concentrated lemon juice, sugar, benzoic acid) • Water • Spices • Salt • Allura red
or
- 2) in descending order of proportion by weight in the finished food as if they were ingredients [B.01.008.2(6), FDR]
This option saves space in the list of ingredients since all ingredients and components are only listed once in descending order based on their total weight in the final food.
For example:
the following ingredient list illustrates how components of tomato paste and lemon juice from concentrate in the above example could be listed along with the other ingredients in the list of ingredients. In order for this option to be plausible, a company must know the exact proportions of all components used by their ingredient suppliers.
Ingredients: Tomatoes • Sugar • Modified corn starch • Water • Concentrated lemon juice • Salt • Spices • Benzoic acid • Allura red.
Generations
Except in the case of allergens (noted below in the second example), ingredients of the third generation and so on are generally not required to be included in the ingredients list.
For example:
consider the ingredients and components of an ice cream containing vanilla cookie pieces. In the ice cream, the vanilla cookies are an ingredient (first generation) that is flavoured with vanilla extract. The vanilla extract is a component of the cookies (second generation) and contains alcohol. The alcohol is a component of the vanilla extract, which can also be referred to as an ingredient of a component within the ice cream (third generation), therefore does not need to be declared in the list of ingredients.
However, some exceptions exist when allergens are present in the third generation or further.
For example:
exceptions will exist if we consider the ingredients of an ice cream containing coconut cookie pieces. In the ice cream, the coconut cookies are an ingredient (first generation) that are made from ground dried coconut that contains sulphites. The dried coconut is a component of the cookies (second generation). Sulphites are a component of the dried coconut, which can also be referred to as a component within the ice cream (third generation). In this case, the ice cream with coconut pieces contains 15 ppm of sulphites. Although the sulphites are a third generation ingredient in the ice cream, as the final product contains more than 10 ppm of sulphites, they must still be declared.
For details, refer to the Food allergens, gluten and added sulphites.
Ingredients that generally do not have to declare their components
The following foods, when used as ingredients in other foods, are exempt from declaring a component (ingredients of ingredients) [B.01.009(1), FDR]. Refer to Food allergens, gluten and added sulphites declaration for exceptions.
List A: ingredients exempt from component declaration
- butter
- margarine
- shortening
- lard
- leaf lard
- monoglycerides
- diglycerides
- rice
- starches or modified starches
- breads subject to compositional standards in sections B.13.021 to B.13.029
- flour
- soy flour
- graham flour
- whole wheat flour
- baking powder
- milks subject to compositional standards in sections B.08.003 to B.08.027
- chewing gum base
- sweetening agents subject to compositional standards in sections B.18.001 to B.18.018
- cocoa, low-fat cocoa
- salt
- vinegars subject to compositional standards in sections B.19.003 to B.19.007
- alcoholic beverages subject to compositional standards in sections B.02.001 to B.02.134
- cheese for which a standard is prescribed in Division 8 of the FDR, if the total amount of cheese in a prepackaged product is less 10% of that packaged product
- jams, marmalades and jellies subject to compositional standards in sections B.11.201 to B.11.241 of the FDR when the total amount of those ingredients is less than 5% of a prepackaged product
- olives, pickles, relish and horseradish when the total amount of those ingredients is less than 10% of the prepackaged product
- 1 or more vegetable or animal fats or oils for which a standard is prescribed in Division 9 of the FDR, and fully hydrogenated, modified or interesterified vegetable or animal fats or oils, if the total of those fats and oils contained in a prepackaged product is less than 15% of that prepackaged product
- prepared or preserved meat, fish, poultry meat, meat by-product or poultry by-product when the total amount of those ingredients is less than 10% of the prepackaged product that consists of an unstandardized food
- alimentary paste that does not contain egg in any form or any flour other than wheat flour
- bacterial culture
- hydrolyzed plant protein
- carbonated water
- whey, whey powder, concentrated whey, whey butter and whey butter oil
- mould culture
- chlorinated water and fluorinated water
- gelatin
- toasted wheat crumbs used in or as a binder, filler or breading in or on a food product
Additional notes about ingredients that generally do not have to declare their components:
- partial declaration of some components for an ingredient exempt from component declaration is considered misleading as per subsection 5(1) of the Food and Drugs Act (FDA), as this gives the erroneous impression that those components are the only ones present in the final product
- if the component of an ingredient included in list A [B.01.009(1), FDR] (that is to say, ingredients exempt from declaring a component) is a vitamin or mineral, when a vitamin and mineral nutrient claim or statement is made on the food label, all components must be shown in brackets following the ingredient [D.01.007, D.02.005, FDR]
- the ingredients of a sandwich made with bread are exempt from declaring components [B.01.008(3)(d), FDR].
Although a list of ingredients is required on the label of a sandwich, the components of those ingredients are not required to be declared. Some common ingredients (for example, salami or mayonnaise) are not exempt from component declarations as per B.01.009(1) of the FDR, but when those ingredients are used to make a sandwich they are granted an exemption from declaring their components. Sandwiches are generally understood to be a filling between 2 pieces of bread, but as there is no legal definition, it was determined that the exemption should include foods commonly considered to be sandwiches, even if they are made with bread-like products. This exemption would apply to a sandwich made with pita or other bread-like products such as tortillas, chapatis and croissants, provided the pita or other bread-like product surrounds the filling or the filling is contained between portions of the bread-like product. This exemption does not apply to open-faced "sandwiches" (for example, pizza and tuna melt) or foods not commonly thought of as sandwiches, such as dessert and confectionery-type products (for example, ice cream or cookie "sandwiches" and donuts with fillings). There are no conditions to this exemption except for the fact that priority food allergens, such as milk, must be declared when present
For example:
the label of a sandwich may have the following list of ingredients:
Ingredients: Whole wheat bread • Salami • Cheddar cheese (milk) • Mayonnaise (egg) • Lettuce • Salt • Pepper
Food preparations that generally do not have to declare their components
The following food preparations and mixtures, when used as ingredients in other foods, are exempt from declaring their components (except for the components included in list C and list D) [B.01.009(2), FDR]. Refer to Food allergen, gluten and added sulphites declarations for additional exceptions.
List B: preparations and mixtures generally exempt from component declaration
- food colour preparations
- flavouring preparations
- artificial flavouring preparations
- spice or herb mixtures
- seasoning mixtures, other than those set out in item 4 and salt added separately, if the total weight of those seasoning ingredients is no more than 2% of the total weight of ingredients used in the manufacture of the prepackaged product excluding added water used as an ingredient that has been removed during its manufacture
- vitamin preparations
- mineral preparations
- food additive preparations
- rennet preparations
- food flavour-enhancer preparations
- compressed, dry, active or instant yeast preparations
Regardless of the quantities in which spice or herb mixtures are added to a prepackaged product, they may be shown at the end of the list of ingredients without declaring their ingredients and components.
The ingredients and components of seasoning mixtures or preparations are exempt from declaration only when the total collective weight of the seasoning mixtures and preparations added to a prepackaged product is no more than 2% of the total weight of all added ingredients. If more than 2%, then the ingredients and components of each seasoning mixture or preparation must be declared in the list of ingredients. Refer to Order of ingredients, as well as the example below, for important details on how to calculate this percentage.
The above exemptions do not apply to ingredients or components that are listed in B.01.009(3) and (4) of the FDR (that is, components of preparations or mixtures which must always be declared) or to food allergens (for example, mustard).
For example:
consider a pasta made with the following ingredients:
- 60.25 kg semolina
- 30.00 kg water (note: 17.50 kg of added water is lost during manufacture)
- 2.50 kg salt
- 4.00 kg herb mixture (containing rosemary, tarragon, silicon dioxide)
- 0.75 kg cheese seasoning (containing parmesan cheese, cayenne pepper, salt, silicon dioxide)
- 0.50 kg basil and lime dressing (containing basil, lime juice, corn syrup solids)
Regardless of the quantity of the herb mixture added, "herbs" may be shown at the end of the list of ingredients without declaring the ingredients and components of the mixture.
With respect to the cheese seasoning and basil and lime dressing, their total collective weight must be calculated as a percentage of the total weight of all ingredients (excluding the weight of added water lost during manufacture):
Total collective weight of the cheese seasoning and the basil and lime dressing = 0.75 kg + 0.50 kg = 1.25 kg
Total weight of all ingredients (excluding the weight of added water lost during manufacture) = 98.00 kg – 17.50 kg = 80.50 kg
% cheese seasoning and basil and lime dressing = (1.25 kg / 80.50 kg) x 100 = 1.55%
Since this amount is no more than 2%, the cheese seasoning and the basil and lime dressing may be simply referred to as "seasonings" at the end of the list of ingredients and none of their ingredients and components must be declared in the list of ingredients, other than salt which must be declared separately. Refer to Food allergen, gluten and added sulphites declarations for manner of declaring the milk allergen in the cheese seasoning and any other allergens present.
Refer to table 2: common names for ingredients or components when those of the class are not shown separately for the optional common names that may be used for spices, herb and seasonings in the list of ingredients.
Components of preparations and mixtures which must always be declared
The following substances, when present in the preparations and mixtures included in list B, must always be shown by their common names in the list of ingredients of the food to which the preparation or mixture is added, as if they were ingredients of that food [B.01.009(3), FDR].
List C: preparation and mixture components which are never exempt from declaration
- salt
- glutamic acid or its salts, includes monosodium glutamate (MSG)
- hydrolyzed plant protein
- aspartame
- potassium chloride
- any ingredient or component that performs a function in, or has any effect on, that food
The components included in list C must be declared as if they were ingredients, as they perform a function in, or have an effect on, the final food (for example, flavour enhancers). As the function of flavour enhancers is to make other flavours more effective, such ingredients are considered to have an effect on the final food and, therefore, need to be declared by their common names in the list of ingredients of the final food (for example, ethyl maltol, disodium guanylate, calcium inosinate and sodium ribonucleotides).
Note: maltol and ethyl maltol can be added to any food product where a flavour preparation or seasoning is permitted.
The components of the flavour-enhancer preparation that simply perform a function on the flavour enhancer preparation (that is to say, they make the preparation easier to handle, measure) and are not considered to have an effect on the final food do not have to be declared.
For example:
- if a lemon flavour preparation has a yellow colour added to it which will also turn the cake yellow when the flavour is added to the cake, then the colour will be considered to have an effect on the food and must be declared in the list of ingredients
or
- if a preservative is added to a flavour enhancer preparation to preserve the stability of the preparation, but will not have the same preservative functions on the final product, then it will not have to be declared in the list of ingredients
Components of foods which must always be declared
The following foods must always be listed by name in the list of ingredients when they are present in the foods included in list A: ingredients exempt from component declaration and the preparations and mixtures included in list B: preparations and mixtures generally exempt from component declaration [B.01.009(4), FDR].
List D: components of foods which are never exempt from declaration
- peanut oil
- fully hydrogenated peanut oil, as per table 1 of the Common Names for Ingredients and Components document
- modified peanut oil
Grouping sugars-based ingredients
Purpose
The requirement to group sugars-based ingredients is intended to help consumers understand their relative proportion in the food compared to other ingredients as well as identify unfamiliar sources of sugars in their foods.
Ingredients with common names such as agave syrup, isomaltose and pear juice concentrate may not be recognized by most Canadians as sugars-based ingredients.
In cases where a product contains a large proportion of these ingredients, grouping would move the sugars-based ingredients closer to the beginning of the ingredient list. This way, the relative proportion of sugars-based ingredients in the product is indicated more clearly.
Grouping required
In a prepackaged product, each sugars-based ingredient must be grouped after the term "Sugars" [B.01.008.3(1), FDR]. It is the manufacturer's responsibility to determine if the ingredients they use fall within the definition of a sugars-based ingredient. Manufacturers must be able to provide evidence of their approach taken to identify sugars-based ingredients for CFIA to verify.
Sugars-based ingredients can be 1 of the following:
Ingredient that is a monosaccharide or disaccharide or a combination of these
Monosaccharides are basic units of sugar and there are only 3 monosaccharides: glucose, fructose and galactose. Disaccharides are sugars made up of 2 monosaccharide units. Common examples of disaccharides include sucrose, lactose and maltose. Therefore, a sugars-based ingredient that is a monosaccharide, disaccharide or a combination of these refers to sugars that have their common name ending in "-ose" or contains the word "sugar". This includes glucose-fructose, cane sugar, sucrose, beet sugar, and lactose. Refer to Annex 1A: examples of sugars-based ingredients that are monosaccharide, disaccharide or a combination of these for a list of additional examples.
Ingredient that is a sweetening agent
This refers to sweetening agents as defined in, but not limited to, Division 18 of the FDR. Some sweetening agents are not in Division 18 but may have prescribed standards by other regulations or other applicable legislation, for example, maple syrup.
Examples of sweetening agents include fancy molasses, maple syrup, brown sugar, agave syrup, refined sugar syrup, honey, and other syrup. Refer to Annex 1B: examples of sugars-based ingredients that are sweetening agents for a list of additional examples.
Ingredient that is a functional substitute for a sweetening agent
With reference to any prepackaged products, a functional substitute for a sweetening agent means a food,
- that is not a sweetener or sweetening agent including any sugars, but
- replaces a sweetening agent and has 1 or more functions of the sweetening agent including, sweetening, thickening, texturing or caramelizing [B.01.001(1), FDR]
Sweetening agents may also have other functions including, flavouring, preservation, browning/caramelization, and colouring.
A "functional substitute for a sweetening agent" is generally not an obvious source of sugars in the food. For example, fruit juice concentrate may not be familiar to some as a source of sugars. Grouping of sugars-based ingredients may help consumers identify these hidden sources of sugars in their foods.
For example:
below are the ingredients in a fruit-berry salad dressing in descending order.
Ingredients: Water, Concentrated white grape juice, White wine vinegar, Canola oil, Strawberry puree, Dijon mustard (water, mustard seeds, vinegar, salt, turmeric), Concentrated raspberry juice, Concentrated blackberry juice, Salt, Concentrated lemon juice, Poppy seeds, Spices and Xanthan gum
The ingredients "concentrated white grape juice, strawberry puree, concentrated raspberry juice, and concentrated blackberry juice" are replacing a sweetening agent (for example, sugar) and have 1 or more functions of the sweetening agent such as texturing (strawberry puree) and colour (concentrated juices). Therefore these ingredients are functional substitutes for a sweetening agent and must be grouped together in brackets following the term "Sugars", as follows:
Ingredients: Water • Sugars (concentrated white grape juice, strawberry puree, concentrated raspberry juice, concentrated blackberry juice) • White wine vinegar • Canola oil • Dijon mustard (water, mustard seeds, vinegar, salt, turmeric) • Salt • Concentrated lemon juice • Poppy seeds • Spices • Xanthan gum
Contains: Mustard
As such, the sugars-based ingredients listed below in Annex 1C: list of sugars-based ingredients that are functional substitutes for sweetening agents must always be grouped after the term "Sugars" in the list of ingredients. Apart from this list, there are other ingredients containing sugars, which may have a function in the food in addition to sweetening. It is the responsibility of the manufacturer to be able to demonstrate that such an ingredient performs a function other than sweetening the food, otherwise it should be grouped. It is also permitted to include in the sugars grouping, any other ingredient containing sugars regardless of its purpose in the food.
Refer to Annex 1C: list of sugars-based ingredients that are functional substitutes for sweetening agents for a list of substitutes that must be grouped.
No grouping required
Sugar alcohols (for example, maltitol) and sweeteners such as steviol glycosides or aspartame cannot be grouped as "Sugars" in the list of ingredients.
Since the aim is to group hidden sources of sugars, there is no need to group ingredients such as chocolate that are well-known as sources of sugar. In addition, no grouping is required for ingredients that are visible in intact pieces or layers such as yogurt coating or pieces of frozen fruits.
For example:
consider a cookie manufacturer who produces raisin and cranberry cookies. The manufacturer adds dried raisins and cranberries to the cookie dough at the final stage of production just before baking the cookies. The dried fruits appear as distinct and intact pieces within the cookie dough and therefore are considered as obvious sources of sugars.
Some ingredients do not meet the definition of sugars-based ingredients. However, the total sugars content of the prepackaged product they are in will still be captured by the "Sugars" declaration in the Nutritional Facts table (NFt).
Refer to Annex 2: examples of ingredients for which grouping with sugars is not required for additional examples that do not meet the definition of sugars-based ingredients.
Some ingredients fall within the definition of sugars-based ingredients but are not required to be grouped when they are in the following prepackaged products [B.01.008.3(4), FDR]:
1. Sweetening agents packaged and sold as such [B.01.008.3(4)(a), FDR].
Sweetening agents include icing sugar, maple syrup, lactose, agave syrup, sugar, golden corn syrup, molasses, glucose syrup, and dextrose.
For example:
consider the ingredient list on prepackaged golden corn syrup. The list of ingredients will show:
Ingredients: Glucose • Water • Refiner's syrup • Salt • Vanillin
2. Fruit or vegetable juice or vegetable drink that does not contain any sweetening agent, including any blend of these juices and drinks [B.01.008.3(4)(b), FDR]
These prepackaged products are fruit or vegetable juices or blends that do not contain any added sweetening agents.
For example:
the prepackaged product "100% orange and tangerine juice" will have the following list of ingredients.
Ingredients: Orange juice • Tangerine juice
3. (a) Fruit or vegetable juice or vegetable drink that does not contain any sweetening agent, including any blend of those juices and drinks to which fruit or vegetable purée or any blend of these purées has been added [B.01.008.3(4)(b)(i), FDR]
These prepackaged products are fruit or vegetable juices or vegetable drinks and purées or a blend of these, with no added sweetening agents.
For example:
the prepackaged product "ABC fruit and vegetable juice" is made from a blend of fruit and vegetable juices to which fruit and vegetable purées have been added. The ingredient list is as follows:
Ingredients: Apple juice • Carrot juice • Mango purée • Sweet potato purée • Natural flavour • Vitamin C
(b) Fruit or vegetable juice or vegetable drink that does not contain any sweetening agent, including any blend of those juices and drinks that are reconstituted [B.01.008.3(4)(b)(ii), FDR]
These prepackaged products are fruit or vegetable juices or vegetable drinks or a blend of these, with no added sweetening agents, provided they have been reconstituted to regular strength.
For example:
"reconstituted orange and apple juice" will have the following list of ingredients.
Ingredients: Water • Concentrated orange juice • Concentrated apple juice
This exemption also applies to a prepackaged product that is a blend of reconstituted fruit or vegetable juices. The ingredient list is shown as follows:
Ingredients: Reconstituted apple juice (water, concentrated apple juice) • Reconstituted grape juice (water, concentrated grape juice) • Natural flavour • Vitamin C
(c) Fruit or vegetable juice or vegetable drink that does not contain any sweetening agent, including any blend of those juices and drinks that are a concentrate intended for dilution and consumption as juice or drink [B.01.008.3(4)(b)(iii), FDR]
An example of this type of prepackaged product is a frozen concentrated fruit juice that only requires dilution with water before consumption.
For example:
the list of ingredients of a frozen concentrated orange juice will be shown as follows:
Ingredients: Concentrated orange juice
4. Fruit or vegetable purée, including any blend of these purées, that does not contain any sweetening agent [B.01.008.3(4)(c), FDR]
These prepackaged products are fruit or vegetable purées or blends made without added sweetening agents.
For example:
a jar of unsweetened apple sauce is made of reconstituted apple purée without any added sweetening agent. The ingredient list on the jar would be declared as follows:
Ingredients: Apples • Water • Ascorbic acid
Consider also a jar of unsweetened strawberry-kiwi apple sauce is made of concentrated apple puree and a blend of fruit purees, without any sweetening agent. Water is added to reconstitute it to regular strength.
Ingredients: Apples • Water • Concentrated apple puree • Concentrated black carrot juice (for colour) • Concentrated blackcurrant juice (for colour) • Strawberry puree • Kiwi puree • Ascorbic acid • Natural flavour
5. Prepackaged products that contain only 1 sugars-based ingredient and the sugars-based ingredient contains the word "sugar" in its common name [B.01.008.3(4)(d), FDR]
For example:
the prepackaged product "lemon cookies" is made with cane sugar, the only sugars-based ingredient in the product, and "cane sugar" contains the word "sugar" in its common name. Therefore, this product is exempt from grouping cane sugar in brackets following the term "Sugars", as shown in the list of ingredients below:
Ingredients: Enriched flour • Buttermilk (cultured milk, salt, sodium citrate) • Butter • Cane sugar • Lemon zest • Sea salt • Sodium bicarbonate.
Contains: Wheat • Milk.
6. Formulated liquid diets, human milk fortifiers and infant formula [B.01.008.3(4)(e), FDR]
Prepackaged products such as infant formula, human milk fortifiers and formulated liquid diets are exempted from grouping their sugars-based ingredients because these products are already strictly regulated and should not be discouraged for consumption because of added sugars-based ingredients.
7. Prepackaged products that contain less than 0.5 g of sugars per serving of stated size [B.01.008.3(4)(f), FDR]
All sugars-based ingredients are exempt from the requirement to group them as "Sugars" in the list of ingredients when total sugars are declared as 0 g in the NFt. This also applies when the NFt is not required.
Sugars-based ingredients may still be grouped voluntarily when the total sugars are declared as 0 g in the NFt.
For example:
a seasoning mix contains maltodextrin, a sugars-based ingredient, and the sugar declared in the NFt is 0 g. The maltodextrin may still be grouped voluntarily, but it is not required, as shown in the lists of ingredients below:
Ingredients: Sugars (maltodextrin), Spices, Salt, Corn starch, Natural flavour
or
Ingredients: Maltodextrin, Spices, Salt, Corn starch, Natural flavour
Order of sugars-based ingredients
When a prepackaged product contains 1 or more sugars-based ingredients, these ingredients must be declared in parentheses immediately following the term "Sugars" in the list of ingredients [B.01.008.3(1), FDR].
The order must be in descending order of proportion by weight of all the sugars-based ingredients added to the mixing bowl [B.01.008.3(2), FDR].
For example:
a prepackaged product of molasses cookies has the following ingredients:
- flour
- fancy molasses
- butter
- brown sugar
- liquid whole egg
- sugar
- salt
- sodium bicarbonate
- spices
- allura red
In this example, the sugars-based ingredients "fancy molasses", "brown sugar" and "sugar" must be grouped together in brackets after the term "sugars", in descending order of proportion by weight, as follows:
Ingredients: Sugars (fancy molasses, brown sugar, sugar) • Flour • Butter • Liquid whole egg • Salt • Sodium bicarbonate • Spices • Allura Red
Contains: Wheat • Egg
Sugars-based ingredients in components
It is not required to group sugars-based ingredients in components. This means when components such as sugar or dextrose appear in parentheses after an ingredient, they will stay ungrouped in those parentheses along with other components. None of the components need to be part of the overall "Sugars" grouping in the list of ingredients.
However, when suppliers sell certain foods in Canada, the sugars-based ingredients would already be grouped. In such cases, manufacturers are allowed to keep the sugars-based ingredients grouped when declaring components.
For example:
consider the ingredient list of prepackaged chocolate chip cookie, made with chocolate chips that contain 2 sugars-based ingredients (sugar and dextrose). The ingredient list declared on the chocolate chips by the supplier is as follows:
Ingredients: Unsweetened chocolate • Sugars (sugar, dextrose) • Soy lecithin • Artificial flavour
In this example, chocolate chips are not required to be grouped after the term "sugars". The manufacturer of the chocolate chip cookie could transfer the components just as they are to the cookie's list of ingredients:
Ingredients: Sugars (brown sugar, sugar) • Flour • Chocolate chips [unsweetened chocolate, Sugars (sugar, dextrose) • soy lecithin • artificial flavour] • Butter • Dried whole egg • Sodium bicarbonate • Vanilla extract • Salt
In the above example, there is no need to "pull out" its components that are already grouped (meaning sugar and dextrose), unless a simplified version of the ingredient list is used.
In a simplified version, sugar and dextrose would need to be grouped with the other sugars-based ingredients in the list of ingredients. For an example of a simplified version with all components listed as if they were ingredients, refer to order of components.
Component declaration for sugars-based ingredients within sugar grouping
If a sugars-based ingredient has components, the requirements related to the order of components must be respected when declaring these components.
For example:
in a salad dressing's list of ingredients, there might be sugars-based ingredients, such as "concentrated white grape juice", with components of their own. While the sugars-based ingredient must be included within the sugar grouping, its components may be declared in 2 ways.
option 1:
after the sugars-based ingredient's common name in the sugar grouping.
Ingredients: Water • Sugars [concentrated white grape juice (white grape juice concentrate, water, preservative, colour)] • Dijon mustard (water, mustard, vinegar, salt)
or
option 2:
in the list of ingredients (if the amounts of each component are known) in descending order of proportion.
Ingredients: Water • Sugars (white grape juice concentrate) • Mustard • Vinegar • Salt • Preservative • Colour
You must use 1 or the other option above.
Common names
Ingredients and their components must be declared in the list of ingredients by their common names [B.01.010, FDR].
Mandatory common names for ingredients and components
To assist consumers in making their food choices, specific mandatory common names are required to be used to identify food ingredients or components. For example, the plant source of certain ingredients, such as hydrolyzed plant proteins, starches, modified starches and lecithin, must be named (for example, hydrolyzed soy protein, wheat starch, modified wheat starch, soy lecithin).
For example:
shortening that contains vegetable oil must be listed in the list of ingredients of a food as "vegetable oil shortening" (unless it contains one of those fats and oils that must be mentioned by name, such as coconut oil, palm oil, palm kernel oil, cocoa butter, peanut oil). Shortening containing lard should be called "lard shortening". Shortening does not have to be qualified in a list of ingredients as "hydrogenated".
The mandatory common names for foods used as ingredients or components (ingredients of ingredients) in other foods are set out in table 1 of the Common Names for Ingredients and Components document, which is incorporated by reference (IbR) into the FDR [B.01.010(3)(a), FDR].
Class/Collective names for ingredients and components
Certain foods and classes of foods may be listed by collective or class names. These collective names may only be used if the individual ingredients/components of that class are not shown separately in the list of ingredients by their individual common name.
The optional common names for foods or classes of foods used as ingredients or components in other foods are set out in table 2 of the Common Names for Ingredients and Components document, which is incorporated by reference (IbR) into the FDR [B.01.010(3)(b), FDR].
Regardless of the quantities in which spices or herbs are added as ingredients or components to a prepackaged product, they may be declared in the list of ingredients using any appropriate combination of names listed in item 6 of table 2 of the Common Names for Ingredients and Components document. For example, if black pepper, basil and thyme are added as ingredients to a prepackaged product, they could be declared as "spices and herbs" or "mixed spices" or "seasonings" at the end of the list of ingredients.
Seasoning ingredients, other than spices, herbs and salt added separately, may only be declared in the list of ingredients as "seasonings" when their total collective weight is no more than 2% of the total weight of all ingredients added to the prepackaged product. If it is more than 2%, then the "seasonings" class name may not be used. Refer to Order of ingredients for important details on how to calculate this percentage.
Item 6.2 of table 2 of the Common Names for Ingredients and Components document clarifies that "seasonings" must only be declared once in the list of ingredients if spices, herbs and seasoning ingredients complying with the maximum 2% total collective weight are added to a prepackaged product.
Common name and ingredient function
When vitamins, mineral nutrients and amino acids are added to food, the purpose of the addition must be determined (for example, as food additives or fortification) in order to determine the appropriate common name.
For example:
when ascorbic acid is added to a food as a vitamin for enrichment purposes, it may be declared as either "vitamin C" or "ascorbic acid" in the list of ingredients. However, when ascorbic acid is used as a food additive for purposes such as bleaching, maturing, dough conditioning, preservation, the name "ascorbic acid" must be used in the list of ingredients, and not "vitamin C". If other forms of ascorbic acid are used, for example sodium ascorbate or ascorbyl palmitate, the correct name must be used in the list of ingredients.
Additionally, when preparations of vitamins, mineral nutrients, food additives and flavour enhancers are added to food, they must be shown in the list of ingredients by the common name of their active ingredient(s) present, for example, vitamin A palmitate. Yeast preparations may be declared as "yeast".
Additional information in the list of ingredients
In general, the common name of an ingredient is the only information that should appear in the list of ingredients, unless a particular adjective or descriptive phrase is prescribed by regulation. Although there is no specific prohibition against additional descriptive information from appearing in the list of ingredients, the general prohibition from declaring any information that is misleading or likely to create an erroneous impression applies [6(1), SFCA; 5(1), FDA]. Declarations about nutrient or other characteristics of ingredients that imply characteristics about the final food therefore may be considered misleading. Refer to General principles for labelling and advertising for more information about misleading.
For example:
the term "iron" on its own is sufficient to represent iron in the list of ingredients as a food regardless of whether or not the iron is in reduced carbonyl or electrolytic form. The terms "reduced", "carbonyl" and "electric" represent processes only and are not considered part of the common name of the mineral nutrient. It is unadvisable to use the term "reduced iron" in the list of ingredients as some consumers can be confused by this terminology and it may lead consumers to believe that the food is reduced in iron which can have serious consequences for those who suffer from hemochromatosis and must avoid excess iron.
As ingredients must be declared by their common names, it is generally not appropriate for brand names to appear in the list of ingredients. There will however be no objection to factual descriptions appearing separately and distinctly from the list of ingredients on any other part of the label.
Acceptable additional information appearing in the list of ingredients can include examples such as:
- a statement of an additive's function, in brackets, for example, "soy lecithin (an emulsifier)"
- a description naming the type of vitamin, in brackets, following the form of the vitamin added, for example, "thiamine mononitrate (vitamin B1)"
- adjectives and descriptions that help to differentiate different versions of the same ingredient, for example, "organic", "fresh tomatoes", "pure milk chocolate", "natural spring water"
Other acceptable descriptions for use in the list of ingredients will be considered on a case-by-case basis. For further details, refer to the Common name page.
Multiple ingredient lists
There should be a list of ingredients for each different kind of food in a multi-pack product in both official languages. These declarations must all use the appropriate Canadian ingredient and component common names in accordance with the FDR or the Safe Food for Canadians Regulations. For further details, refer to the Common name page.
Phenylalanine statement
Foods sweetened with aspartame must include a statement warning individuals with phenylketonuria that the food contains phenylalanine [B.01.014(1), FDR].
Examples of the statement include:
- "Persons with phenylketonuria: contains phenylalanine"
- "Aspartame contains phenylalanine"
Refer to Legibility and location for further details on how statements should appear in the list of ingredients.
Food allergens, gluten and added sulphites
Food allergens and gluten must be declared in the list of ingredients in which they are present, or in a "food allergen source, gluten source and added sulphites statement", by their prescribed source names [B.01.010.1(2), FDR].
Note: the words "contains statement" and "food allergen source, gluten source and added sulphites statement" have the same meaning and are used interchangeably in this section for ease of reference.
Added sulphites at levels of 10 ppm or more, and that are not already required to be shown in the list of ingredients, must be declared in the list of ingredients by their prescribed source names. If a contains statement is used, the declaration must use 1 of these common names: "sulfites", "sulfiting agents", "sulphites" or "sulphiting agents" [B.01.010.2(3), B.01.010.2(6), FDR].
If a contains statement is used on the label, all allergen, gluten and added sulphite information must appear in the statement at least once, even if that information is already shown in the list of ingredients for the product [B.01.010.3(1)(b), B.01.010.3(2), FDR].
For example:
Ingredients: Wheat flour • Water • Vegetable oil margarine • Sugar • Yeast • Canola oil shortening • Potato starch • Garlic • Salt • Parsley • Seasoning • Diacetyl acid esters of mono and diglycerides • Whey powder • Calcium propionate • Potassium bisulphite
Contains: Wheat • Milk • Sesame • Sulphites
In this example, all food allergens, gluten sources and added sulphites are declared at least once in the contains statement, even though wheat and potassium bisulphite (sulphites) already appear in the ingredient list. Since a contains statement is being used to declare other known allergen sources (milk and sesame), wheat and sulphites must appear in both places.
Order of food allergen and gluten source declaration
The requirement for food allergens and gluten declarations apply to all generations of ingredients. Therefore, food allergens and gluten must be declared regardless of which generation they are present in. For example, if they are present in the third or fourth generation of ingredients, they will still have to be declared in the list of ingredients on the product label or in a contains statement. Refer to Generations for more information on the generations of ingredients.
The source of food allergens and gluten must be declared in 1 of 2 ways:
- in the list of ingredients [B.01.010.1(2)(a), FDR], or
- in a "food allergen source, gluten source and added sulphites statement" [B.01.010.1(2)(b), FDR]
option 1: the prescribed source name of the food allergen or gluten may be shown in parentheses in the list of the ingredients, as follows:
- (a) immediately after the ingredient that is shown in the list, if the food allergen or gluten [B.01.010.1(8)(a)(i) to (iii), FDR]:
- (i) is that ingredient
- (ii) is present in that ingredient but is neither a component of that ingredient nor present in a component of that ingredient, or
- (iii) is a component of that ingredient or is present in a component of that ingredient, and the component is not shown in the list of ingredients. Note that if the source of a food allergen or gluten is shown immediately following the ingredient, the components of the ingredient must be shown immediately after the allergen source [B.01.008.2(5)(a), FDR]
For example:
Ingredients: Flour (wheat) • Liquid albumin (egg) • Vegetable oil • Sugar • Chocolate chips (milk) (sugar, chocolate liquor, cocoa butter, milk ingredients, soy lecithin, salt, natural flavour).
In this example:
- wheat protein is an inherent part of flour but is not a component
Since wheat is both a food allergen and gluten source, it must be declared - liquid albumin is an egg protein
As eggs are a food allergen, egg must be declared - the components of the ingredient "chocolate" are shown in parentheses immediately after the allergen source milk
or
- (b) immediately after the component that is shown in the list of ingredients, if the food allergen or gluten is the component or is present in the component [B.01.010.1(8)(b), FDR]
For example:
Ingredients: Pastry pieces [flour (wheat), butter (milk), liquid albumin (egg), canola oil] • Sugar • Natural flavour.
In this example, as "pastry pieces" are not exempt from component declaration, its components must be declared. The required food allergen and gluten prescribed source names are declared in parentheses after the component in which they are present:
- wheat is a part of flour
- butter is made from milk
- liquid albumin is an egg protein
option 2: the prescribed source name of a food allergen or gluten may be shown on the label of a product in a contains statement that complies with the naming and location restrictions outlined in B.01.010.3(1) of the FDR if the food allergen or gluten:
- (a) is an ingredient, or present in an ingredient that is not shown in the list of ingredients, but is not a component of that ingredient or present in a component of that ingredient [B.01.010.1(9)(a), FDR]
or
- (b) is a component or present in any generation of a component and neither the component nor the ingredient in which it is present is shown in the list of ingredients [B.01.010.1(9)(b), FDR]
For example:
protective edible coatings, including waxes, are part of the post-harvest and handling technologies routinely used by the fresh produce industry to minimize moisture loss, prolong the shelf life and to improve the appearance of fresh produce. Edible wax coatings on fresh fruits and vegetables can potentially contain soy, chitosan (derived from crustaceans) and caseinate components (derived from milk).
Wax coating compounds, their components and other protective edible coatings are not required to be shown on the label of any prepackaged fresh fruits or vegetables. Therefore, if any food allergens, gluten or added sulphites are present in these coatings, they must be declared on the labels of prepackaged fruits and vegetables. As wax coatings and other protective edible coatings are exempt from being declared as ingredients and components as per B.01.008(3)(a) of the FDR, the other components of these coatings are still exempt from declaration and do not need to appear in a list of ingredients.
For instance, if a bag of prepackaged apples has a wax coating containing caseinate compounds, as casein contains milk, the food allergen source "milk" must be declared on the label of the apples. The food allergen source "milk" can be declared in a contains statement on the label: "Contains: milk".
Order of added sulphites declaration
Sulphites, like any other food additive, are required to be declared in the list of ingredients of food labels when they are added directly to a prepackaged product as an ingredient or a component of an ingredient that is not exempt from component declaration. If added sulphites are present in a prepackaged product in the first generation (that is to say, an ingredient) or second generation (a component), and are not exempt from declaration, they must be declared in the list of ingredients regardless of their quantity. Refer to Generations for more information on the generations of ingredients.
In the following examples, sulphites are a component of the dried apricots. Dried apricots are not exempt from component declaration, therefore sulphites must be declared along with all the other components of the dried apricots, regardless of their quantities.
- Sulphites added as a food additive ingredient
For example: consider an example of dried apricots where sulphites added at a level of 5 ppm
Ingredients: Apricots • Sugar • Sulphites
- Sulphites added as a component of an ingredient that is not exempt from component declaration
For example:
Ingredients: Rolled oats • Flour (wheat) • Dried apricots (apricots, sugar, sulphites) • Liquid whole egg (liquid whole egg, beta-carotene) • Salt • Sodium bicarbonate • Soy lecithin
In this example, allergen sources such as wheat in flour may instead be shown in the contains statement.
Added sulphites declaration
If added sulphites are present in a prepackaged product in a total amount of 10 parts per million or more, and are not already required to be shown in the list of ingredients under section B.01.008 or B.01.009 of the FDR, the sulphites must be shown on the label of the product in:
- the list of ingredients, or
- in a "food allergen source, gluten source and added sulphites statement" that complies with the requirements under section B.01.010.3(1) of the FDR [B.01.010.2(3), FDR]
Sulphites that are declared in the list of ingredients must be shown as follows:
(a) sulphites that are a component of an ingredient that is shown in the list of ingredients must be shown either in parentheses immediately after the ingredient or at the end of that list, where they may be shown in any order with the other ingredients that are shown at the end of that list under B.01.008.2(4) of the FDR [B.01.010.2(7)(a), FDR], and
(b) in all other cases, the sulphites must be shown at the end of the list of ingredients where they may be shown in any order with the other ingredients that are shown at the end of that list under subsection B.01.008.2(4) of the FDR [B.01.010.2(7)(b), FDR]
For example:
in a box of cookies that are made with apricot jam, sulphites are a component of the apricot jam with pectin. Assuming that the jam is only 5% or less of the cookie, it is exempt from component declaration [B.01.009(1), item 24, FDR], and that the sulphites are at a level of 13 ppm (therefore greater than 10 ppm), the sulphites must be declared as per subsection B.01.010.2(3) of the FDR. Both of the examples below are acceptable ways to declare sulphites (at levels of 10 ppm or more) in the list of ingredients.
In the list of ingredients, sulphites can be declared in parentheses after apricot jam with pectin:
Ingredients: Rolled oats • Flour (wheat) • Liquid whole egg • Apricot jam with pectin (sulphites) • Salt • Sodium bicarbonate • Soy lecithin
or at the end of the list of ingredients in any order:
Ingredients: Rolled oats • Flour (wheat) • Liquid whole egg • Apricot jam with pectin • Salt • Sodium bicarbonate • Soy lecithin • Sulphites
Note: in situations where added sulphites are a component that must be declared in the list of ingredients as per B.01.008(1)(b) of the FDR, but the total amount is less than 10 parts per million, then the sulphites will not have to be declared in the contains statement. In both examples, allergen sources such as wheat in flour may instead be shown in the contains statement.
Cross-contamination statements
A cross-contamination (or precautionary) statement is a declaration on the label of a prepackaged product that alerts consumers of the possible presence of an allergen in the food. For more details, refer to Food allergen cross-contamination (or precautionary) statements.
Prescribed source names
Food allergens and gluten prescribed source names
The prescribed source of a food allergen present in a prepackaged product must be declared as listed in the table below [B.01.010.1(6), FDR]. Refer to Health Canada – Common food allergens for other names of priority food allergens which must be declared by their prescribed source names.
| Food allergens | Prescribed source name |
|---|---|
| Almonds | Almond, almonds |
| Brazil nuts | Brazil nut, Brazil nuts |
| Cashews | Cashew, cashews |
| Hazelnuts | Hazelnut, hazelnuts |
| Macadamia nuts | Macadamia nut, Macadamia nuts |
| Pecans | Pecan, pecans |
| Pine nuts | Pine nut, pine nuts |
| Pistachios | Pistachio, pistachios |
| Walnuts | Walnut, walnuts |
| Peanuts | Peanut, peanuts |
| Sesame seeds | Sesame, sesame seed, sesame seeds |
| Wheat or triticale | Wheat or triticale |
| Eggs | Egg, eggs |
| Milk | Milk |
| Soybeans | Soy, soya, soybean or soybeans |
| Powdered mustard | Mustard, mustard flour, ground mustard |
| Fish, crustaceans, molluscs | Common name of the fish, crustacean or mollusc |
| Mustard seeds | Mustard, mustard seed or mustard seeds |
The prescribed source of gluten from the grain of a cereal referred to in the definition of "gluten", derived from that grain, or from the grain of a hybridized strain cereal from 1 or more of the cereals referred to in the "gluten" definition, must be declared as follows [B.01.010.1(7), FDR]:
| Gluten | Prescribed source names |
|---|---|
| Barley | Barley |
| Oats | Oats |
| Rye | Rye |
| Triticale | Triticale |
| Wheat | Wheat |
Food allergens and gluten only have to be declared once in the list of ingredients on the label of all prepackaged products. Further declaration is not required when the prescribed source names for allergens and gluten are already part of the common name of the ingredient or component which they are in, or are already declared in the parenthesis immediately after an ingredient or component [B.01.010.1(10), FDR]. There is however no prohibition from declaring food allergens or gluten more than once unless it would result in a non-compliance with other regulations or legislation.
For example:
Ingredients: Enriched flour (wheat) • Buttermilk (cultured milk, salt, sodium citrate) • Butter • Cane sugar • Lemon zest • Sea salt • Sodium bicarbonate.
In the above example, all food allergen and gluten sources are declared appropriately in the list of ingredients, either in the parentheses or as part of the common name of the ingredient. Although butter is a milk source, as the FDR states, food allergens, gluten and added sulphites only have to be declared once on the label. As milk is identified in the common name of cultured milk, it does not have to be declared as a source for butter.
Sulphites prescribed source names
If sulphites are declared in the list of ingredients, they must be declared as follows [B.01.010.2(6), B.01.010.2(8), FDR]:
| Sulphites | Prescribed source names |
|---|---|
| potassium bisulphite | sulfites, sulfiting agents, sulphites or sulphiting agents or potassium bisulphite |
| potassium metabisulphite | sulfites, sulfiting agents, sulphites or sulphiting agents or potassium metabisulphite |
| sodium bisulphite | sulfites, sulfiting agents, sulphites or sulphiting agents or sodium bisulphite |
| sodium metabisulphite | sulfites, sulfiting agents, sulphites or sulphiting agents or sodium metabisulphite |
| sodium sulphite | sulfites, sulfiting agents, sulphites or sulphiting agents or sodium sulphite |
| sodium dithionite | sulfites, sulfiting agents, sulphites or sulphiting agents |
| sulphur dioxide | sulfites, sulfiting agents, sulphites or sulphiting agents |
| sulphurous acid | sulfites, sulfiting agents, sulphites or sulphiting agents |
As long as 1 of the prescribed source names for sulphites listed above are present, or are already declared in the parenthesis immediately after another ingredient or component, further declaration is not required in the list of ingredients [B.01.010.2(10), FDR]. However, note that there is no prohibition from declaring sulphites more than once unless it would result in a non-compliance with other regulations or legislation.
Bilingualism
The list of ingredients must be shown in both English and French unless the product is exempt from bilingual labelling [B.01.012, FDR]. For further details, refer to Bilingual labelling.
The English and French versions of the list of ingredients must begin on a separate line, except, for small packages with an available display surface (ADS) of less than 100 cm2, where the second language version of the list of ingredients may start after the end of the first language list [B.01.008.2(9), FDR].
Flexibility in the declaration of a list of ingredients
Sometimes there is variation in the supply of certain ingredients for natural or economic reasons, which results in manufacturers substituting, varying or omitting certain ingredients that are normally used. Section B.01.011 of the FDR and section 284 of the SFCR provide an orderly procedure to outline how manufacturers can substitute, vary or omit ingredients while maintaining a correct list of ingredients for consumers.
Omissions and substitutions
When ingredients or components are omitted or substituted during a 12-month period, all of the foods that may be used as ingredients or components throughout the 12-month period must be shown in the list of ingredients. It must be clearly stated as part of the list of ingredients that the ingredient or component might be omitted/substituted. The ingredients that may be omitted or substituted are grouped with the same class of foods that are otherwise used as ingredients or components and the foods within each group must be listed in descending order of the proportion by weight in which they will be needed during the 12-month period [B.01.011(1), FDR; 284(1), SFCR].
Variations
When proportions of ingredients or components are varied, the list of ingredients may show the ingredients or components on the label in the same proportions throughout the 12-month period if it is clearly stated as part of the list of ingredients that the proportions indicated are subject to change, and the ingredients or components are listed in descending order of the proportion by weight in which they are likely to be used for the majority of the 12-month period [B.01.011(2), FDR; 284(2), SFCR].
Therefore, there are different ways that a manufacturer can indicate in the list of ingredients if an ingredient or component has been omitted, substituted or varied. In all circumstances, the ingredients must appear at the proper place in the list of ingredients of the finished product. The class group should then appear at the proper place in the list of ingredients of the finished product.
No objection will be taken if an asterisk appears next to an ingredient to indicate that an explanation regarding an omission, substitution or variation will appear at the end of the list of ingredients.
For example:
flexibility in the declaration of ingredients lists can appear in the marketplace as:
- "Ingredients: Pork and/or Beef..."
This statement means that the amounts of pork and/or beef will be varying in the prepackaged product over a 12-month period. A given product may contain only pork, beef, or a mixture of both pork and beef. However, as pork has been listed first, it is expected that over the 12-month calendar year, pork is present in higher quantities - "Ingredients: Milk solids, Bacterial culture, Salt, *may contain Carotene."
This ingredients list represents commercial butter which uses colour at some points during the year. The colour of butter varies seasonally, based on the cattle's diet. To ensure consistency, colour may be added or omitted. "May contain" is considered an appropriate way to indicate that an ingredient is sometimes omitted from the recipe - "Ingredients: Baby romaine lettuce, Baby swiss chard, Baby spinach, Baby kale, Arugula, Radicchio. The proportion of ingredients in each package may vary."
Mixed green salads are usually produced in varying quantities of lettuces depending on the availability of ingredients. All of the ingredients are the same over a 12-month period, but the proportions vary. It must be clearly stated that the proportions indicated are subject to change, and they must be listed in descending order of the proportion in which they will be in for the majority of the 12-month period
Legibility and location
Subsections B.01.008.1(1) to (6) of the FDR govern the font type, height, width, and colour requirements for the following information in the list of ingredients:
- the ingredient list and components [B.01.008.2 and B.01.009, FDR]
- sugars-based ingredients [B.01.008.3, FDR]
- phenylalanine statement [B.01.014, FDR]
- food allergens, gluten and sulphites [B.01.010.1 and B.01.010.2, FDR]
- "food allergen source, gluten source and added sulphites statement" [B.01.010.3, FDR]
- cross-contamination statement [B.01.010.4, FDR]
In addition to this page, detailed examples of the legibility requirements are presented in the Compendium of Templates for Nutrition Facts tables and list of ingredients, which is available for order on the Health Canada website.
Location
The list of ingredients may appear on any panel of the prepackaged product except for the bottom panel [B.01.005, B.01.008(1), FDR]. Only under very specific circumstances is the list of ingredients permitted on the bottom panel, for example, ornamental containers (see Location requirements). All information in the list of ingredients, including the phenylalanine statement, contains statements, and cross-contamination statements, must be shown on the same continuous surface [B.01.010.3(1)(a.2), B.01.010.4(1)(c), B.01.014(2)(c), FDR].
The phenylalanine statement, if applied, must appear immediately after the list of ingredients [B.01.014(2), FDR].
A contains statement, if applied, must appear immediately after the phenylalanine statement or, if there is no such statement, immediately after the ingredient list [B.01.010.3(1)(a.1), FDR].
A cross-contamination statement must appear after the contains statement if there is one, after the phenylalanine statement if there is no contains statement, or after the list of ingredients if there are no such statements [B.01.010.4(1)(a)(i) to (iii), FDR].
These statements, if made, must be grouped together with the ingredient list of the same language version without any intervening printed, written or graphic material [B.01.010.3(1)(a)(iii), B.01.010.3(1)(a.1), B.01.010.4(1)(b) and (d), B.01.014(2)(b), FDR]. The ingredient list or a statement in the other official language is considered intervening material.
In certain cases, such as a single-ingredient product where no ingredient list is shown, the English and French versions of a contains statement, if applicable, must begin on a separate line. Small packages with an available display surface (ADS) of less than 100 cm2 are exempt from this requirement, and the second language version of the statement may start after the end of the first language statement [B.01.010.3(3), FDR].
For example:
consider a package of sesame seeds, a single ingredient product where the common name serves as the list of ingredients. The contains statement must be shown as:
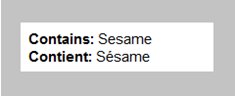
Description for photo – List of ingredients, separate line
The English title "Contains" and the English allergen list appear on the first line. The French title "Contient" and the French allergen list appear below the English on a separate line.
or, if the package meets the small package exemption, the statement may be shown as:

Description for photo – List of ingredients, same line
The English title "Contains" and the English allergen list is immediately followed by the French title "Contient" and the French allergen list on the same line.
Font
Type
Information appearing in the list of ingredients, phenylalanine statement, contains statement and cross-contamination statement must be shown:
- in a single standard "sans serif" font that is non-decorative and the characters never touch each other or any other differentiating feature (for example, the border around the list of ingredients) [B.01.008.1(1)(b), FDR]. Examples of "sans serif" fonts include Arial and Helvetica
- in regular type, except as otherwise provided in sections B.01.008.2, B.01.010.4 and B.01.014 [B.01.008.1(1)(d), FDR]
Ingredients and components must be listed in regular font type as set out in B.01.008.1(1)(d) of the FDR. No bolding, italics, or underline is permitted, except for entries in the contains statement which may appear in regular or bold type [B.01.010.3(1)(c), FDR].
For example:
An acceptable manner of declaring the list of ingredients is as follows:
Ingredients: Flour (wheat) • Water • Vegetable oil margarine (milk) • Sugar • Yeast • Canola oil shortening • Potato starch • Garlic • Salt • Parsley • Seasoning (sesame) • Diacetyl acid esters of mono and diglycerides • Whey powder • Calcium propionate
An unacceptable manner of declaring the list of ingredients is shown below, as the title is underlined and the allergens are shown in bold.
Ingredients: Flour (wheat) • Water • Vegetable oil margarine (milk) • Sugar • Yeast • Canola oil shortening • Potato starch • Garlic • Salt • Parsley • Seasoning (sesame) • Diacetyl acid esters of mono and diglycerides • Whey powder • Calcium propionate
Type height and leading
Except as otherwise stated in sections B.01.008.2 to B.01.010.4, and B.01.014 of the FDR, the type height for the list of ingredients, phenylalanine statement, contains statement and cross-contamination statement must:
- be of the same height (except in the case of titles, which may use a greater type height), and
- be a minimum of 1.1 mm [B.01.008.1(1)(e), FDR]
A type height of 1.1 mm is equivalent to 6 points type in sans serif font and refers to the height of the lower case letter "x" [B.01.008.1(6), FDR]. Each character must also use identical leading of at least 2.5 mm [B.01.008.1(1)(e), FDR].
If a Nutrition Facts table appears on the label and the nutrients are shown in a type size of at least 8 points, the information appearing in the list of ingredients, phenylalanine statement, contains statement and cross-contamination statement must be shown in a type height:
- that is the same height as the nutrients in the table [B.01.008.1(3)(a), FDR], and
- of at least 1.4 mm (8 points equivalency) with identical leading of at least 3.2 mm [B.01.008.1(3)(b), FDR].
If the contains statement and cross-contamination statement are preceded by a phenylalanine statement, and they begin on the same line as the phenylalanine statement, the titles, or the cross-contamination statement itself if no title appears, must be shown in a type that is at least 0.2 mm greater than the type height used in the phenylalanine statement [B.01.010.3(1)(a)(iv), B.01.010.4(1)(g), FDR].
Width
The font type must appear in a normal or condensed width that is not scaled down so that the characters take up less space horizontally [B.01.008.1(1)(c), FDR].
If a Nutrition Facts table also appears on the label, the type width for information appearing in the list of ingredients, in the phenylalanine statement, in the contains statement and in the cross-contamination statement must be the same as that required to show the nutrients that appear in the Nutrition Facts table [B.01.008.1(1)(c), FDR]. For more information, refer to Nutrition labelling - Presentation of the Nutrition Facts table.
Titles
The titles that introduce the list of ingredients and contains statement must be shown in bold type and without any intervening written, printed or graphic material before the information they introduce. The titles must be declared as "Ingredients:", "Ingredients" or "Ingredients :" and "Contains:", "Contains" or "Contains :" respectively [B.01.008.2(1), B.01.010.3(1)(a), FDR]. Italics and underlining are not permitted.
Health Canada and the CFIA encourage food manufacturers and importers to use the title "May contain:" or "May contain" to introduce the cross-contamination statement on food labels. If a title is used, it must appear in bold when the statement appears on the same line as the ingredient list, contains statement or phenylalanine statement [B.01.010.4(1)(f), FDR].
When the cross-contamination statement appears on the same line as the ingredient list, the contains statement or the phenylalanine statement, and is not introduced by a title, the cross-contamination statement must be in bold [B.01.010.4(1)(e), FDR].
Bolding is optional when the cross-contamination statement appears on a separate line (see examples below).
For example:
in the list of ingredients below, each title appears on a separate line and therefore the title "May contain:" is not required to appear in bold.
Ingredients: Enriched flour • Buttermilk (cultured milk, salt, sodium citrate) • Butter • Cane sugar • Lemon zest • Sea salt • Sodium bicarbonate.
Contains: Wheat • Milk.
May contain: Brazil nuts
In the example below, the cross-contamination statement appears on the same line as the "food allergen source, gluten and added sulphites" statement. Therefore, the title "May contain:" must appear in bold.
Ingredients: Enriched flour • Buttermilk (cultured milk, salt, sodium citrate) • Butter • Cane sugar • Lemon zest • Sea salt • Sodium bicarbonate.
Contains: Wheat • Milk. May contain: Brazil nuts
Titles may be shown in a type height that is greater than the type height used to show each entry in the ingredients list, phenylalanine statement, contains statement and cross-contamination statement [B.01.008.1(4), FDR]. When more than 1 title appears on a label, each title must be shown in the same type height [B.01.008.1(5), FDR]. For example, the titles "Ingredients:" and "Contains:" may both appear in a type height of 1.4 mm and each listed entry in a type height of 1.1 mm.
Subheadings
Subheadings are allowed to appear following the "Ingredients" title however, bolding of subheadings is not permitted.
For example:
Ingredients:
Coating: Sugar • Palm oil • Milk ingredients • Soy lecithin • Salt • Flavour
Pretzel: Flour • Sugars (sugar, corn syrup, malted barley extract) • Salt • Sodium bicarbonate
Topping: Sugars (sugar, tapioca syrup) • Chocolate chips (unsweetened chocolate, sugar, dextrose, soy lecithin, artificial flavour) • Allura red • Flavour • Shellac
Contains: Milk • Soy • Wheat • Barley
In the above example "Coating:", "Pretzel:" and "Topping:" appear as subheadings following the "Ingredients" title. Note: subheadings must still appear in descending order of their proportion in the final product.
Cases and separators
In the ingredients list, contains statement and cross-contamination statement, each entry must appear in lower case letters while the first letter of each entry must be shown in upper case letters [B.01.008.2(3)(b)(i), FDR].
However, the following information must always be shown in upper case letters [B.01.008.2(5)(c), FDR]:
- the name of a food additive written partially or entirely in its acronym form (for example, calcium disodium EDTA, BHA and BHT) [B.01.008.2(5)(c)(i), FDR], and
- the alpha-descriptor that is part of the common name of a food additive, vitamin and micro-organism (for example, chymosin A, vitamin B3 and D-calcium pantothenate) [B.01.008.2(5)(c)(ii), FDR]
Proper nouns such as the geographic origin used to describe an ingredient or a component of an ingredient may be shown with the first letter in upper case. For example: Alaskan pollock.
Ingredients must be separated by either a bullet point or comma [B.01.008.2(3)(c), B.01.010.3(1)(c), FDR]. There is no prohibition against or requirement for the use of a period at the end of the ingredients list or statements and may be used at the discretion of the manufacturer.
Sugars-based ingredients must be shown in lower case letters only and be separated by a comma [B.01.008.3(3)(a) and B.01.008.3(3)(b)(ii), FDR].
Components must be separated by a comma and be shown in lowercase letters only, except for in the instances where information is required to be in upper case [B.01.008.2(5), FDR].
For example:
both lists of ingredients below demonstrate proper use of upper and lower case lettering, and acceptable ways of separating ingredients.
Ingredients: Enriched flour, Buttermilk (cultured milk, salt, sodium citrate), Butter, Sugars (dextrose, cane sugar), Lemon zest, Sea salt, Sodium bicarbonate.
Contains: Wheat • Milk.
or
Ingredients: Enriched flour • Buttermilk (cultured milk, salt, sodium citrate) • Butter • Sugars (dextrose, cane sugar) • Lemon zest • Sea salt • Sodium bicarbonate.
Contains: Wheat • Milk.
Colours and borders
Information appearing in the list of ingredients, in the phenylalanine statement, in the contains statement and in the cross-contamination statement must be shown in a single colour of type that is a visual equivalent of 100% solid black type (for example, black or dark type) and be on a white or neutral background that has a maximum 5% tint of colour [B.01.008.1(1)(a), FDR]. Foods for use in manufacturing other foods and Foods for use in commercial or industrial enterprise or institution are exempt from this background colour requirement [B.01.008.1(2)(a) to (b), FDR].
The ingredients list must be clearly differentiated on the label by using either or both of the following options [B.01.008.2(2), FDR]:
option 1: use a solid-line border around the ingredient list or, 1 or more solid lines appearing above, below or at the sides of the list. The solid line must be the same colour as that used for the font type (meaning, black or dark colour) [B.01.008.2(2)(a), FDR].
For example:
a solid-line border around the list of ingredients:

or, an ingredient list with 1 solid line appearing above and 1 solid line appearing below the list:

option 2: use a colour that creates a contrast between the background colour of the ingredient list and the background colour used on the adjacent surface of the label [B.01.008.2(2)(b), FDR].
For example:
placing the ingredient list on a white background when the adjacent area on label is gray would create contrast and clearly differentiate the ingredient list from the rest of the food label:
Description for photo - Use a colour that creates a contrast
The panel of the package on which the ingredient list appears has a gray background colour. The ingredient list appears on a white background in contrast to the otherwise gray panel.
All information in the list of ingredients, including the phenylalanine statement, contains statement and cross-contamination statements, must be within the same border/lines and be against the same background colour [B.01.010.3(1)(a.2), B.01.010.4(1)(c), B.01.014(2)(c), FDR].
An additional solid line may appear above a cross-contamination statement when the statement begins on a different line than the ingredient list or statement that immediately precedes it [B.01.010.4(1)(d), FDR].
Artificial flavour
Flavouring preparations are subject to Division 10 of the FDR. When an artificial flavour (for example, artificial apple flavour) is added to a food, whether alone or with natural flavouring agents, the word "artificial" or "imitation" must be included as an integral part of the flavouring preparation name. It must be in the same type size and style as the flavouring preparation name [B.10.008, FDR].
For information on artificial flavour as it relates to pictorial representations, refer to Pictures, vignettes, logos and trade-marks.
Food additives in the list of ingredients
The FDR requires food additives to be declared in the list of ingredients of all prepackaged products unless the product is exempt from bearing a list of ingredients. For information related to food additives, including general labelling requirements and manner of declaring food additives in the list of ingredients, refer to the Food additives page.
Information letters
Information for manufacturers and importers of flavour packets
Annex 1: examples of sugars-based ingredients that require grouping
A. Examples of sugars-based ingredients that are monosaccharide, disaccharide or a combination of these
- Barbados sugar (Muscovado)
- Barley malt (unfermented List Note 1)
- Beet sugar
- Burnt sugar (caramelized sugar)
- Cane juice
- Cane juice crystals
- Cane sugar
- Caster (castor) sugar
- Coarse sugar
- Coconut palm sugar
- Coconut sugar
- Crystalline fructose
- Date sugar
- Dehydrated cane juice
- Demerara-style sugar
- Evaporated cane juice (evaporated cane sugar, dried cane syrup, cane syrup solids)
- Fondant sugar
- Free-flowing brown sugars
- Fructose
- Fructose solids
- Fructose syrup
- Fruit sugar
- Galactose
- Glucose-fructose
- Granulated sugar
- Grape sugar
- High-fructose corn syrup
- High-maltose corn syrup
- Isomaltose
- Isomaltulose
- Lactose [S]
- Maltose
- Maple Sugar
- Organic sugar
- Palm sugar
- Pearl sugar (sanding sugar)
- Panocha (cane sugar produced by a crude milling process)
- Raw sugar
- Saccharose (sucrose)
- Soft sugar
- Sucrose
- Sugar [S]
- Superfine sugar
- Table sugar
- Turbinado sugar
- White sugar
B. Examples of sugars-based ingredients that are sweetening agents
- (Naming the source of the Glucose) syrup [S]
- Agave syrup
- Apple cider syrup
- Apple syrup
- Brown rice syrup
- Brown sugar, Yellow sugar or Golden sugar [S]
- Carob syrup
- Concentrated maple water
- Corn sweetener
- Corn syrup
- Corn syrup solids
- Dextrose Anhydrous [S]
- Dextrose Monohydrate [S]
- Fancy molasses [S]
- Glucose or Glucose syrup [S]
- Glucose solid or Dried glucose syrup [S]
- Golden syrup
- Honey [S]
- Icing sugar [S] or Confectioner's sugar or Powdered sugar
- Invert sugar [S]
- Liquid invert sugar [S]
- Liquid sugar [S]
- Malt syrup
- Malted barley extract
- Malted barley syrup
- Maple Syrup
- Maple water
- Oat syrup solids
- Other syrups
- Potato syrup solids
- Raisin syrup
- Refined sugar syrup, Refiners' syrup or Golden syrup [S]
- Refiners' molasses, Blackstrap molasses or Cooking molasses [S]
- Rice syrup
- Sorghum syrup
- Sweet sorghum
- Table molasses [S]
- Tapioca syrup
- Tapioca syrup solids
- Wheat syrup
C. List of sugars-based ingredients that are functional substitutes for sweetening agents
- Condensed milk or sweetened condensed milk
- Decharacterized juice
- Fruit juice (except lime/lemon juice)
- Fruit juice concentrate (except lime/lemon juice concentrate)
- Fruit paste
- Fruit purée
- Fruit purée concentrate
- Malted milk or malted milk powder
- Maltodextrin (no fibre)
- Nectar
Annex 2: examples of ingredients for which grouping with sugars is not required
- Candied fruits
- Caramel
- Carob chips
- Chocolate (milk, white, dark)
- Chocolate chips
- Coconut flakes
- Compound coating
- Fibres (refer to the List of dietary fibres reviewed and accepted by Health Canada's Food Directorate)
- Fruit spreads
- Fruits (fresh/canned in water/ frozen/dried/freeze dried/filling)
- Glaze
- Jams, marmalades and jellies
- Maple flakes
- Maple/Yogurt clusters
- Maraschino cherries
- Marshmallow
- Vegetable juice
- Vegetable purée
- Vegetables (fresh/canned/ frozen/dried/freeze dried/filling)
- Yogurt coating, filling
Definitions
- Components
- Component means an individual unit of food that is combined as an individual unit of food with 1 or more other individual units of food to form an ingredient [B.01.001(1), FDR].
- Flavouring preparation
- Flavouring preparation includes any food for which a standard is provided in Division 10 of the FDR [B.01.001(1), FDR].
- Food allergens
- Food allergen means any protein from any of the following foods, or any modified protein that includes any protein fraction derived from any of the following foods [B.01.010.1(1), FDR]:
- almonds, Brazil nuts, cashews, hazelnuts, macadamia nuts, pecans, pine nuts, pistachios or walnuts
- peanuts
- sesame seeds
- wheat or triticale
- eggs
- milk
- soybeans
- crustaceans
- molluscs
- fish, or
- mustard seeds
- Food allergen source, gluten source and added sulphites statement
- Food allergen source, gluten source and added sulphites statement, means a statement appearing on the label of a prepackaged product that indicates the source of a food allergen or gluten that is present in the product or the presence in the product of added sulphites in a total amount of 10 ppm or more [B.01.010.1(1), FDR].
- Functional substitute for a sweetening agent
- Functional substitute for a sweetening agent means, in respect of a prepackaged product, a food – other than any sweetener or sweetening agent, including any sugars - that replaces a sweetening agent and that has 1 or more of the functions of the sweetening agent including, sweetening, thickening, texturing or caramelizing [B.01.001(1), FDR].
- Generations
-
Generation, although not directly defined by the FDR, means all food ingredients added to form a product at the same time that are often developed from an earlier type of generation.
Components are often referred to in terms of generations, and are explained as follows:
- an ingredient = the first generation
- an ingredient of an ingredient (a component) = the second generation
- an ingredient of a component = the third generation
- an ingredient of an ingredient of a component = the fourth generation
- …and so on…
- Gluten
-
Gluten means [B.01.010.1(1), FDR]:
- any gluten protein from the grain of any of the following cereals or the grain of a hybridized strain created from at least 1 of the following cereals:
- barley
- oats
- rye
- triticale
- wheat (all species, including spelt and kamut)
or
- any modified gluten protein, including any gluten protein fraction, that is derived from the grain of any of the cereals referred to above or the grain of a hybridized strain referred to above
The definition of gluten also includes prolamins, which are a group of plant storage proteins having a high content of the amino acid proline. Prolamins are also found in the seeds of the above cereal grains as follows: gliadin in wheat, hordein in barley, secalin in rye, and avenin in oats.
- any gluten protein from the grain of any of the following cereals or the grain of a hybridized strain created from at least 1 of the following cereals:
- Ingredients
- Ingredient means an individual unit of food that is combined as an individual unit of food with 1 or more individual units of food to form an integral unit of food that is sold as a prepackaged product [B.01.001(1), FDR].
- Leading
- Leading is the space between lines of type, and is measured from the baseline of the letters in 1 line of type to the baseline of the letters in the line of type above it.
- Spices, herbs and other seasonings
-
Spices, herbs and other seasonings are a group of collective terms; however, they are not all interchangeable. Although not defined in the FDR,
- spices are used to describe 1 or more blend of aromatic ingredients that consist of flowers, seeds, fruits, roots, bark or the nuts of a plant, such as cinnamon, cumin, cardamom, turmeric, cloves and nutmeg
- herbs are the edible leaves of herbaceous plants, such as thyme, basil, rosemary, oregano, mint, and chives
- seasonings include savory ingredients not recognized as spices or herbs, such as condiments, sauces, or mixtures of several ingredients that provide a characteristic taste to a product
- Sugars-based ingredient
-
Sugars-based ingredient means, in respect of a prepackaged product,
- (a) an ingredient that is a monosaccharide or disaccharide or a combination of these
- (b) an ingredient that is a sweetening agent other than 1 referred to in paragraph (a); and
- (c) any other ingredient that contains 1 or more sugars and that is added to the product as a functional substitute for a sweetening agent [B.01.001(1), FDR]
- Sulphites
- Sulphites mean 1 or more of the following food additives that are listed in column I of item 21 of table 2 of the Common Names for Ingredients and Components document, namely, potassium bisulphite, potassium metabisulphite, sodium bisulphite, sodium dithionite, sodium metabisulphite, sodium sulphite, sulphur dioxide and sulphurous acid, and are present in a prepackaged product. This definition only applies to sulphites that are present in the prepackaged product as a result of being added [B.01.010.2(1) and (2), FDR].
- Sweetening agent
- Sweetening agent includes any food for which a standard is provided in Division 18, FDR but does not include those food additives included in Health Canada's List of permitted sweeteners [B.01.001(1), FDR].
- Date modified: