This consultation closed 2013-12-20
Table of Contents
- 1 Purpose
- 2 Background
- 3 Current Framework - Feed Ingredient Assessment and Authorization
- 4 Classification of Feed Ingredients
- 5 Proposed Framework for Assessment and Authorization of Feed Ingredients
- 6 Other Areas of Potential Regulatory Change
- 7 Summary
- Annex
1 Purpose
This document outlines the principles, objectives and proposed direction of a modernized regulatory framework to assess and authorize livestock feed ingredients in Canada and seeks broad stakeholder comments and input on the regulatory proposal. This is one of four proposals prepared by the Canadian Food Inspection Agency (CFIA) regarding the modernization of Canada's regulatory framework for livestock feed.
2 Background
2.1 Change Management at the CFIA
The Canadian Food Inspection Agency (CFIA) has embarked on a comprehensive change agenda to strengthen its foundation of legislation, regulatory programs, and inspection delivery. These directions set the context for renewal of the Feeds Regulations (Regulations).
In December 2011, the CFIA initiated a systematic, multi-year review of its regulatory frameworks for food safety, plant health and animal health. Through this structured and comprehensive review, the Agency intends to update its regulatory frameworks to:
- reduce overlap and redundancy;
- increase responsiveness to industry changes;
- address gaps, weaknesses and inconsistencies; and
- provide clarity and flexibility to affected regulated parties.
The modernization of the Regulations has been identified as a short-term priority in the first three years of the modernization initiative.
The Agency will take opportunities to explore opportunities for modernizing its legislative base as regulation modernization takes place. In the case of feeds this could mean seeking to update the Feeds Act (Act) with modern powers such as incorporation by reference (the inclusion of external documents into regulations) and novel approaches to feed regulation such as licensing facilities.
The Agency is engaged in developing a new inspection delivery model that is based on common inspection activities and standard processes for all of its inspection programs and is more focused on risk as a basis for prioritizing inspection activities. This new model, supported by a renewed training regime, is being pioneered for food inspection, but will be adapted as appropriate to help streamline and create efficiencies for other inspection activities including feed.
2.2 Feed Regulatory Modernization
Modernization of the Regulations strives to reduce compliance burden and support innovation while maintaining animal and human health, as well as environmental and economic sustainability.
The current feed regulatory framework is product-based including pre-market assessment and approval or registration of single feed ingredients and any mixed feeds (comprised of one or more ingredients) that do not meet exemption from registration criteria. In addition, the Regulations include requirements for labelling of feeds to compel accurate information on:
- the purpose of the ingredient or feed;
- the physical properties of the feed;
- the nutritional components;
- truthful claims; and
- information about its:
- nature;
- proper use; and
- any necessary details to ensure safe and effective use.
The Agency maintains post-market oversight of ingredients and feeds to further assure they are compliant with the composition, labelling and registration standards of these Regulations.
Modernization of the Regulations benefits the collective Canadian feed industry which includes commercial feed manufacturers, retailers, importers, exporters, ingredient manufacturers, and farmers. As well as aligning with other international feed regulatory regimes, modernization also maintains the objective of enhancing animal health and food safety for the Canadian public.
2.3 Principles of CFIA Regulatory Modernization
The following principles guide the overall CFIA regulatory modernization process:
- protecting public safety and confidence in the inspection framework;
- enabling an environment of improved business opportunity and consumer choice by facilitating innovation and competitiveness;
- establishing clear policy objectives through consultation with partners - including industry, consumers, small business, other departments and provinces;
- finding the appropriate balance between administrative costs and benefits derived from regulatory intervention;
- developing consistent, transparent and, where appropriate, outcome-based regulations with performance measurements;
- recognizing the vital importance of science and risk management approaches in decision making; and
- aligning, to the extent possible, with international trading partner and provincial standards.
The ultimate objective of the review of the Regulations is to develop a modernized risk- and outcome-based regulatory framework for feeds that supports fair and competitive trade in the market, minimizes regulatory burden, and protects animal health, the environment, and the food production continuum.
2.4 Consultation Process and the Role of this Proposal
Stakeholder consultation is an important component of regulatory modernization. The CFIA conducted several pre-consultation activities with stakeholders during 2012 to stimulate interest in a renewed feed regulatory framework, as well as to identify current and future trends, opportunities, and challenges facing stakeholders as they might relate to feed production and use.
The Agency took a three-tiered approach to this pre-consultation. First, informal bilateral meetings were held with stakeholder groups. Following this, a discussion paper and survey were made available on-line to provide all stakeholders with the opportunity to provide feedback. Finally, a two-day multi-stakeholder workshop was held (September 2012).
Following the September 2012 workshop there was recognition that further stakeholder consultation was needed. An industry and government "Feed Regulatory Steering Group" (FRSG) was formed in November 2012 with members representing the commercial feed manufacturers, major national livestock producer associations, the CFIA, and Health Canada's Veterinary Drugs Directorate. Early on the FRSG determined that feed regulatory modernization was sufficiently complex that it should be broken down into the following components ("modules") for further discussion:
- Feed ingredient assessment and authorization;
- Feed hazard identification and preventive controls (possibly including facility licensing);
- Feed labelling; and
- Compliance and enforcement.
A January 2013 workshop was held to discuss the topic of feed ingredient regulation with interested stakeholders. The CFIA prepared and distributed to workshop participants a background discussion paper on the current approach to feed ingredient regulation in Canada. The Animal Nutrition Association of Canada (ANAC), Canada's principal domestic feed industry trade association, also prepared and distributed a proposal for a risk-based framework for feed ingredient approvals. ANAC submitted a supplementary paper to further clarify elements of their proposal in March 2013.
The information provided and gathered from all of these activities has formed the foundation of this proposal on the first module (feed ingredient assessment and authorization) which outlines what changes the Agency proposes to the pre-market assessment and approval (registration) of single feed ingredients, what could remain the same, and what the benefits and implications are for the feed industry.
3 Current Framework - Feed Ingredient Assessment and Authorization
The Agency's current role in feed ingredient regulation is pursuant to the Feeds Act (Act) and the Feeds Regulations (Regulations) which prescribe the requirements for composition, labelling, registration and standards for ingredients that are manufactured, sold in and imported into Canada. Safe and effective feeds contribute to the production and maintenance of healthy livestock and safe foods of animal origin.
Within the scope of the current authorization framework, all ingredients require pre-market assessment and authorization on the basis of safety and efficacy.
Ingredients are assessed for safety in order to ensure there will not be any negative impacts on human health (food safety and occupational health), animal health, or the environment.
Efficacy is the ability to produce a desired or intended result. Ingredients are currently required to have scientific evidence in support of:
- intended or purported purpose (have a feed-related purpose, which can include supplying nutrients, aiding or improving the manufacture of feeds, preserving the feed, or adding flavour or colour);
- fitness for purpose (do they perform their intended purpose, also referred to as efficacy or utility); and,
- any specific claims.
The efficacy data supports the classification of a product as a feed (not a drug, veterinary biologic, etc.) and supports the safety assessment of the ingredient. Safe and effective feeds contribute to the production and maintenance of healthy livestock and safe foods of animal origin.
All currently approved feed ingredients are listed in Schedules IV or V of the Regulations.
Schedule IV is comprised of eight classes:
- Dry Forages and Roughages;
- Pasture, Range Plants and Forages Fed Green;
- Silages;
- Energy Feeds;
- Protein Feeds;
- Mineral Products;
- Vitamin Products and Yeast Products; and
- Miscellaneous Products.
Schedule V comprises a list of extracts, oils and chemicals which are approved for use as flavouring ingredients in feeds.
Ingredients listed in Schedules IV and V are further delineated as Part I or Part II ingredients based on whether the specific ingredient source poses safety or efficacy concerns and whether these concerns can be mitigated by restrictions within the definition (e.g. use restrictions, warning or caution statements, maximum levels of contaminants). Part I of Schedules IV and V lists ingredients for which any new source of the ingredient does not require further assessment provided it meets the definition. Part II of Schedules IV and V lists ingredients for which there are safety or efficacy concerns with the source. Each new source of any ingredient listed in Part II of either Schedule IV or V must supply information to support its approval.
Any ingredient that is not currently listed in, or that does not meet the definition provided in Schedule IV or V, must be approved by the CFIA prior to use for feeding livestock. Ingredients which are found to be unsafe or unsuitable, or where the information submitted is insufficient to substantiate safety and intended purpose are not approved. Ingredients found to be safe and efficacious are approved and incorporated into Schedules IV or V either as a new ingredient or by modifying an existing ingredient definition.
4 Classification of Feed Ingredients
In order for an ingredient to be assessed and authorized as a feed ingredient by the CFIA, it must first be classified as feed. There are other substances that can be orally consumed by livestock which are not feed and are regulated differently. These include therapeutic (drug) products (regulated under the Food and Drugs Act), pest control products (Pest Control Products Act) and veterinary biologics (Health of Animals Act). This can lead to confusion, especially for foreign companies as products may be regulated differently in foreign jurisdictions.
Products such as coccidiostats and histomonostats are regulated as veterinary drugs in Canada, but are considered feed additives in the European Union (EU). Other types of products such as microbial products may have a different classification depending on the purpose or mode of action associated with them. Additionally, the level a product is used at, the method of administration and types of claims associated with it can all have an impact on how the product is classified in Canada. In the past, difficulties associated with the interpretation of the drug and feed definitions have resulted in significant industry confusion, which led to inefficiencies regarding product evaluation and approval, as well as compliance and enforcement activities. Therefore, there is a need for a clear and transparent classification process for some products intended for use in livestock species.
The CFIA recognizes the importance of this issue and is working collaboratively with Health Canada to clarify the policy for how feeds and veterinary drugs are classified in Canada.
In February 2013, Health Canada posted for consultation a draft guidance document developed in conjunction with the CFIA which aims to clarify some of the overlap between the feed and drug legislative definitions. See Consultation - Guidance Document on Classification of Veterinary Drugs and Animals Feeds. It outlines the present approach to the classification of feeds and veterinary drugs, and provides interpretations and definitions. This guidance document is intended to assist proponents seeking to have new products assessed and authorized for marketing in Canada with determining what constitutes a feed versus a veterinary drug and where to direct their applications accordingly. The consultation period on the draft guidance ended in early June 2013; the CFIA and Health Canada recognize the importance of this guidance and are presently working together to review the feedback received from the consultation towards finalizing the guidance this year.
In addition, Health Canada is working on the modernization of Canada's regulatory system for veterinary drugs. Key features of the proposed regulatory framework include enabling proportional oversight on veterinary drugs based on benefits, harms and uncertainties.
Given the finalized guidance will form the policy basis for how feeds and veterinary drugs are defined and classified in Canada, it is not included in the scope of this proposal and is not discussed in further detail. The guidance will, however, be referred to in further consultations with the industry in the context of a modernized approach to management of applications for the assessment and authorization of new or modified feed ingredients.
5 Proposed Framework for Assessment and Authorization of Feed Ingredients
5.1 Scope
This proposed modernized authorization framework describes how ingredients for use in livestock feeds and feeding will be regulated and will apply to:
- Ingredients - defined as a "single ingredient feed" in the current regulations i.e. "single ingredient feed" means any substance or mixture of substances that is assessed or evaluated as being acceptable for use in feeds and that is described in an item of Schedule IV or V.
- Speciality Feed Additives - which are composed of one or more single ingredient feeds, such as flavouring agents, colouring agents, enzyme supplements, rumen buffers and mould inhibitors which may be considered ingredients in the manufacture of feed products by the feed industry.
- Processing Additives - defined as additives used at low levels to improve the manufacturing process of an ingredient, or its production process.
Once a product has been classified as a feed, as with the current feed regulatory framework, any feed ingredient that is new (i.e. not on a positive list such as currently served by Schedules IV and V), has been modified such that it differs significantly from a conventional ingredient or is represented for a purpose other than the one(s) it has already been authorized for must undergo a mandatory pre-market assessment before it can be sold, manufactured or imported into Canada as livestock feed.
Figure 1 illustrates the process flow envisioned for the modernized ingredient assessment and authorization framework discussed in detail below.
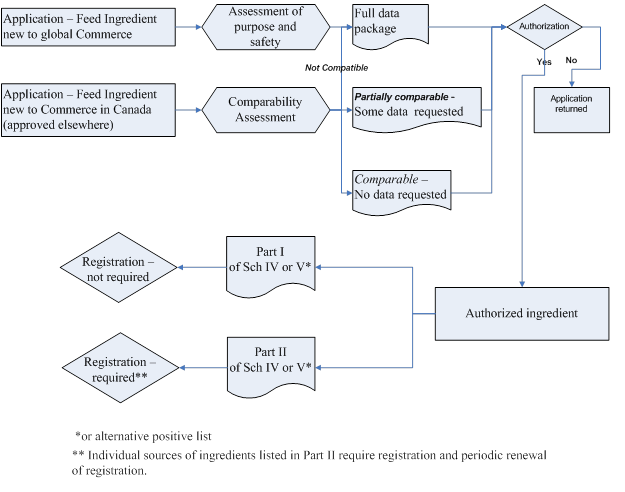
Description for flowchart envisioned for the modernized ingredient assessment and authorization framework
Figure 1 shows the route a new feed ingredient takes to market in Canada.
There are two possible routes an application can take. The first route is when the application is for a feed ingredient new to global commerce. The ingredient is submitted to the CFIA and must undergo an assessment of purpose and safety. A full data package must be submitted with this application. Once the data package has been reviewed, the ingredient moves to the authorization stage.
Concurrently underneath this path is another route for a feed ingredient that is new to commerce in Canada but has been approved for use elsewhere. After submitting this type of application, a Comparability Assessment will be performed. This assessment examines the methods used by the foreign authority to assess the new ingredient. There are three possible outcomes of the comparability assessment:
- If the foreign methodology is not comparable to the standards used by the CFIA, the application will have to go through the full assessment process.
- If the foreign methodology is partially comparable, the applicant will have to provide additional data as required.
- If the foreign methodology is deemed comparable to the standards used by the CFIA, no further data will be required. The ingredient would then go to the Authorization stage.
At the authorization stage the ingredient may receive approval. If approval is not received, the application is returned to the applicant. If approved, the ingredient is authorized.
Once authorized, an ingredient would be added to either Part I of Schedule IV or V or Part II of Schedule IV or V.
If the ingredient is added to Part I of either Schedule IV or V, it does not need to be registered. However, if the ingredient is added to Part II of Schedule IV or V, the ingredient must be registered and periodically reviewed.
Organization of this Proposal
This proposal is organized based on the following components:
- Application for Ingredient Authorization
- Notification of Applications
- Assessments
- Purpose
- Safety
- Claims
- Assessment Outcomes
- Consultation on Proposed Outcomes
- Positive Lists of Authorized Ingredients
- Post-authorization Conditions
For each component, this proposal provides a brief discussion as to:
- the current situation;
- areas of proposed change;
- a rationale for the CFIA proposal; and
- anticipated benefits.
In addition, other areas of potential regulatory change being examined by the CFIA as part of the feed ingredient authorization framework are identified and discussed at the end of the proposal. These areas include:
- processing additives;
- definitions;
- mechanism to provide exclusivity of proprietary data;
- incorporation by reference; and
- role of product registration.
It is anticipated that modernizing the feed ingredient authorization framework will have positive impacts on other aspects of the regulatory framework. Section 6.5 of this proposal (Additional Anticipated Benefits of the Proposed Ingredient Authorization Framework) provides a discussion on the nature of some projected benefits.
5.2 Applications for Ingredient Authorization (Unchanged)
As in the current framework, it is proposed that before it is imported, manufactured or sold in Canada, proponents continue to apply for authorization for any feed ingredient that:
- is new (i.e. not on a positive list);
- has been modified such that it differs significantly from a conventional ingredient; or
- is represented for a purpose other than the one(s) for which it has already been authorized.
5.2.1 Application Format
Proponents will have to submit all of the required information in their application package; however, to promote flexibility the format and presentation of that application package will not be dictated by a rigid format. This will help to minimize the applicant's regulatory burden by allowing them the option to either use or reformat an existing data package (such as one submitted to another country) or submit a package prepared specifically for Canada. In either case, the application must address Canadian submission requirements. Information provided as part of a data package must be of a quality that would be acceptable for publication in a peer-reviewed scientific journal. The CFIA will continue to provide policy guidance on the application package to facilitate submission of applications.
5.3 Notification of Applications (New)
At the present time, the CFIA does not disclose whether applications for new or modified ingredients have been received or the status of the application in the evaluation queue. It is proposed that the CFIA adopt a "notice of submission" (NoS) mechanism for all feed ingredient applications. A NoS is presently applied on a voluntary basis for novel feeds from plant sources. See: Background.
The NoS mechanism would be a formal notice communicated by the CFIA (e.g. posted on the CFIA website) which would indicate the receipt of an application for ingredient authorization by the CFIA and include information such as:
- the proposed name of the ingredient;
- a brief description of the ingredient;
- the name of the proponent; and
- the date that the application was accepted for review.
The information for the notice would be required from the proponent as part of the application package, and confidential business information will continue to be respected.
In circumstances where an ingredient is determined unacceptable for approval following evaluation, or where the proponent has withdrawn the application, this information would be reflected on the notification page, but no further information would be published. The proponent would be notified prior to posting if their ingredient has not been approved.
During the CFIA's January 2013 feed ingredient classification and authorization workshop, some feed industry comments were heard that such a notification mechanism would be useful as it is helpful to know what products are or were in the queue for assessment and prevent duplication of effort to have the same ingredient submitted for authorization through multiple applications. This mechanism supports the Government of Canada's Open Government initiative and will increase the transparency of the CFIA's ingredient authorization process by providing stakeholders with information regarding ingredients which are in the assessment process. This concept is also consistent with both national and international practices such as:
- Health Canada's Pesticide Public Registry where information on all pesticide applications, their status and outcomes is available on Health Canada's website;
- the U.S. Food and Drug Administration's (FDA) GRAS Inventory Notification where information on GRAS notices are placed on the FDA's website when they are received and the FDA's response is posted when it is available;
- The U.S. FDA's Food and Color Additive Petitions Under Review where a listing of food and color additive petitions filed by the FDA's Office of Food Additive Safety (OFAS) and under active review are available on their website; and
- The European Food Safety Authorities' (EFSA) Register of Questions, which contains information on questions asked to EFSA by the European Union (EU) regulatory authorities on food and feed safety issues within EFSA's remit. The Register of Questions provides information on the progress of a question as it moves through the risk assessment process. A question normally results in the publication of an opinion by one of EFSA's ten scientific panels or its scientific committee.
Have your say:
The CFIA is seeking comments on the proposal to implement a notification of submission mechanism.
5.4 Assessments
The pre-market assessment of ingredients will continue to include safety for animals, people and the environment as well as purpose and any claims that a proponent would wish to use in association with the marketing and use of the ingredient. However, a number of measures to provide flexibility are also proposed.
Definitions
Purpose – each ingredient presented for assessment must have a purpose. Purpose is the objective, aim, goal or end use for which an ingredient is intended. Ingredients must have a purpose that is:
- within the scope of the definition of a "feed" set out in the Feeds Act; and
- suitable for listing in one or more of the classes of ingredients set out in Schedule IV of the Feeds Regulations (for example, an energy feed, a protein feed, a vitamin, a mineral etc.); or
- as a flavouring ingredient in Schedule V.
For example: Zinc oxide has a purpose of supplying zinc in feed rations and is listed as an item (6.85) in Class 6, Mineral Feeds, Part I of Schedule IV of the current Regulations; Brilliant Blue FCF Lake has the purpose of a dye to colour the feed, (item 8.8, Class 8, Miscellaneous Feeds, Part I of Schedule IV.
Claims - Any representation which states, suggests, or implies that a feed ingredient has particular characteristics relating to its origin, nutritional properties, nature, purpose, production, processing, or composition, or any characteristic that relates to animal productivity, animal health, food safety, food characteristics (for example, the feeding of a pigment to colour egg yolks) or the environment.
5.4.1 International Data (Modernized)
The framework will continue to require that a proponent provide satisfactory scientific evidence for the assessment of ingredients. Rather than having only one authorization pathway as is the case in the current framework, it is proposed that the type and amount of information required in support of an ingredient assessment will be subject to one of three authorization pathways:
- For a feed ingredient that is new to the global market – If Canada is the first country in which approval is sought and where it will be marketed (i.e. it has not been approved as a feed anywhere else in the world), a full assessment process and data package will be required to support the application to have the ingredient authorized.
- For a feed ingredient that is new to the Canadian market – If an ingredient is already authorized as a feed in another country and the following criteria are met, a modified application package would be allowed:
- From a recognized country – if the ingredient has already been authorized in a country which the CFIA has recognized as having an authorization process that is equivalent to that used by Canada, a modified application package would be allowed. An applicant would need to submit evidence of the foreign authorization and a summary characterizing the ingredient and indicating it is the same ingredient, its intended purpose and directions via the application process.
- If the ingredient has already been authorized in a country which the CFIA does not recognize as having an equivalent process, the full data package will be required.
- Voluntary submission of data – Proponents that have ingredients which are eligible to have a modified application package may still choose to submit the full package on a voluntary basis.
Comments received from some stakeholders suggested that the CFIA consider adding ingredients approved by other jurisdictions (for example, the U.S. or EU) directly to its positive lists. While the approach of adding a foreign-approved ingredient without any assessment is not possible, the above approach offers a provision to allow the addition of ingredients authorized by foreign jurisdictions to the list of approved feed ingredients with reduced data requirements. Acceptance of foreign evaluation procedures would need to be based on an assessment process equivalent to that used by Canada.
Maintaining the current framework that all ingredients are assessed for purpose and safety prior to authorization is seen as appropriate with the systems used by major trading partners like the U.S. and the EU. The proposed framework would not be an impediment for the CFIA to participate in international convergence efforts and may aid such initiatives by providing more flexibility in what is allowed as part of the information supplied to support an ingredient assessment. Further details on the three aspects of the assessment (safety, purpose and claims) are provided below.
Have your say:
The CFIA is seeking comments on the proposal to accept foreign authorization based on CFIA recognition of the foreign country having an authorization process that equivalent to that used by Canada.
5.4.2 Intended Purpose Assessment (Modernized)
The Agency currently assesses ingredient purpose and performance by requiring results of "scientific investigations respecting the conditions under which the feed would be efficacious for its intended purposes" to be included as part of an application for authorization. As a general principle, the Agency requires that proponents substantiate an ingredient's purpose and performance. Once authorized, ingredients may then be used in mixed feeds in accordance with their purpose and any of the other registration, compositional and labelling standards in the Regulations that apply to mixed feeds.
Coupled with the pre-market assessment, authorization and, in some cases, registration of feed ingredients in respect of intended purpose, another current CFIA approach is to assess "nutritional efficacy" with respect to mixed feeds. For example, this is done by way of requiring assessment and registration of mixed feeds having nutrient guarantees outside the ranges of nutrients identified for different species and classes of livestock set out in Table 4, Schedule I.
Consultations to date with stakeholders have re-affirmed that ingredients must have a purpose. However, stakeholders have questioned the value of assessing the "nutritional efficacy" of mixed feeds and the mandatory registration of certain products in this regard.
Within the scope of a modernized regulatory framework, the CFIA is proposing to continue requiring that feed ingredients have an intended purpose or purposes identified that correspond to the definition of a "feed" listed above and allow for proper categorization of the product (protein feed, energy feed, vitamin etc.) For products whose purpose would not correspond to those listed below, for example, ones which would supply a nutrient that is not required by livestock, evidence to support a feed purpose would be required. This approach is consistent with other international feed regulatory jurisdictions where ingredients are classified and regulated on the basis of purpose and scientific evidence in support of intended purpose is required.
Suitable purposes for feed ingredients would include:
- providing nutritional requirements of livestock necessary for maintenance, growth and production (e.g. protein, fat, vitamins, minerals);
- preventing or correcting nutritional disorders of livestock;
- aiding or improving the manufacture, handling or storage properties of feeds (e.g. anticaking, pelleting);
- improving or conserving the nutrient content of feeds (antioxidant, mould inhibitor); or
- flavouring or colouring feeds (e.g. colouring agents, flavouring agents).
If a feed purpose is not demonstrated, the product would be better classified as something other than a feed, such as a drug or pesticide. As outlined in Section 4 above, the CFIA is working with Health Canada to provide clear guidance on this issue.
However, the CFIA proposes to redefine its role with respect to ingredient performance assessment by restricting the submission and pre-market assessment of scientific evidence supporting ingredient performance to only those products whose failure to perform an identified purpose would have significant health or safety implications to animals or humans. The scope of this approach would include the submission of evidence to demonstrate nutritional essentiality. Other examples of products for which evidence of performance would have to be submitted for assessment would include information about the bioavailability of a nutrient or the ability of an ingredient to fulfill an intended effect (e.g. prevent feed oxidation, adjust feed pH). Categories of ingredients or criteria for which purpose evidence would not be required would be established by policy. The nature of evidence where it would be required to support the assessment of ingredients would be tiered based on ingredient type, purpose, and safety. Under this modernized approach, the Agency would:
- add flexibility to its assessment process by including the acceptance (when appropriate, as outlined above) of purpose assessments for ingredients performed and authorized internationally by other feed regulatory authorities;
- reduce the kind and number of feed ingredients requiring mandatory registration and expand the number of classes of purposes of feed ingredients in the Regulations to facilitate increased options and flexibility; and
- develop a list of pre-approved claims that will facilitate the marketing of new ingredients and mixed feeds without further oversight (as discussed in the Claims Assessment section of this proposal).
Where non-health and safety implications associated with ingredient purpose failure would apply(for example, an anti-caking agent), the ingredient manufacturer or other proponent would still be required to have determined and have evidence on hand of a product's capacity to achieve its intended purpose. This evidence would not be required to be submitted as part of the ingredient authorization process. This redefined role would, however, no longer make Canada's approach to assessing ingredient performance consistent with how other regulatory jurisdictions (for example, the U.S. and EU) as these jurisdictions require that ingredient performance be demonstrated in all cases.
By taking this approach, the CFIA would reduce the pre-market assessment burden associated with the authorization of some ingredients and many mixed feeds. Regulated industry would no longer have to register multiple products to support quality issues or claims and realize major savings in access time to market and registration fees. In a certain sense, maintaining pre-market assessment of feed ingredients provides assurance that a decrease in oversight over mixed feeds is counter-balanced, or underpinned, by robust oversight of ingredients.
Further details on the specific requirements for the purpose assessment will be contained in policy via regulatory guidance documents to provide proponents with more clarity. Required information does not need to be company generated data; publically available data sources may be used to support fitness for purpose as well as information from foreign jurisdictions. The CFIA will continue to offer support and advice to stakeholders through these guidance documents, pre-submission consultations, and training workshops.
Have your say:
The CFIA is seeking comments on the proposal to:
- maintain a role in assessing the purpose of feed ingredients;
- modernize the approach taken to assess and authorize new or modified feed ingredients as to their purpose by restricting the requirement to submit evidence to substantiate the performance of an ingredient to ones having potential animal or human health or safety implications only; and
- reduce the scope of pre-market assessment and registration of mixed feeds with respect to nutritional efficacy.
5.4.3 Safety Assessment (Unchanged)
Feed ingredients must be safe. Consultations to date with stakeholders have confirmed that the government has a central role to play in the assessment and authorization of ingredients within the scope of safety in respect of animal health, human health (via food residues and worker/by-stander exposure), and the environment.
The Regulations will be designed to continue to provide objectives and general criteria which proponents will need to consider when preparing the safety component of an application for an ingredient assessment, while still providing flexibility for applications to be tailored based on history of use and complexity of the product. The general criteria would include aspects pertaining to the identity and characterization of an ingredient (e.g. what it is, what it will be used for, how it was developed, etc.), intended purpose of the ingredient, information requirements to be used to identify risks, and manufacturing process.
The CFIA does and would continue to provide guidance to stakeholders on how to meet the data requirements for the assessment process by:
- publishing guidance documents;
- staging workshops; and
- holding one-on-one consultations with prospective applicants.
Information requirements outlined in these documents will allow data to be addressed by either empirical data or valid scientific rationale. The use of peer-reviewed scientific literature or foreign data, where appropriate, would also be permitted. Assessment of these new ingredients will follow internationally established standards and guidance such as those established by Codex and the Organization for Economic Cooperation and Development (OECD).
5.4.4 Claims Assessment (Modernized)
Much like the pre-market assessment of ingredient intended purpose, the role that the CFIA currently plays in pre-market assessment and authorization of claims associated with feed ingredients is among the foremost subjects discussed in consultations with stakeholders to date.
Recommendations related to claims featured prominently in the ANAC's January 2013 proposal and were discussed in detail at the CFIA's stakeholder workshop on ingredients in January 2013. Additional recommendations to the CFIA were provided in ANAC's March 2013 supplement and discussed bilaterally with CFIA officials in April 2013.
Among their January 2013 recommendations, a definition for a "claim" was proposed by the ANAC (See Recommendation 5 in Annex I).
For the purposes of this proposal and discussion, it has been further refined by the CFIA to be defined as follows:
"Any representation which states, suggests, or implies that a feed ingredient has particular characteristics relating to its origin, nutritional properties, nature, purpose, production, processing, or composition, or any characteristic that relates to animal productivity, animal health, food characteristics, food quality or the environment".
In their January 2013 proposal, the ANAC recommended the following approach be taken to the oversight of claims:
"Recommendation 8: It is recommended that a positive list of permissible claims be developed and pre-approved for use with feed ingredients. If an ingredient carries an approved claim, then that claim may also be used in mixed [feeds] containing that ingredient without further efficacy assessment. Examples of such claims are provided in Figure 3: Permissible Claims."
In ANAC's view, this approach would be applied to ingredients that have already been authorized and for claims related to livestock production, nutritional requirements and nutritional disorders. Examples of possible permissible production claims cited by ANAC included "average daily gain" and "feed conversion efficiency". Examples of nutritional disorder claims cited by ANAC included "mitigation of subclinical acidosis (ruminants)" and "mitigation of subclinical ketosis (ruminants)". For new claims associated with new ingredient applications, the CFIA agrees with the January ANAC proposal to continue pre-market assessment and authorization.
In its March 2013 supplement, ANAC expanded further on the oversight of claims as follows:
"ANAC proposes that regulatory language reflect the general parameters for claims, however the regulations do not need to be prescriptive in defining their format. For example, guidance documents could provide the necessary information to allow sponsors to make appropriate claims.
Given the provisions and safeguards described above, there should be no CFIA pre-market assessment of claims. Instead, the use of a post-market monitoring and complaint-driven process will address non-substantiated or inappropriate claims. This approach should incorporate measures to provide appropriate sanctions for complaints upheld, while enabling protection against frivolous complaints."
On the basis of these recommendations, the CFIA:
- agrees to include the concept of a positive list of pre-approved permissible claims for use with feed ingredients within the scope of a modernized framework; and
- proposes, with respect to its role regarding oversight of claims for feed ingredients, to redefine its role by restricting the pre-market assessment and authorization of claims which would only have health or safety implications; and
- proposes to allow the use of claims pre-approved for all or specific ingredients in accordance with the list without requiring additional pre-market approval by the CFIA; and
- subject to CFIA and Health Canada acknowledgement that new or modified claims do not have health and safety implications requiring government oversight, proposes to allow the addition of new or modified claims to the list and allow their use with all or specific ingredients in accordance with the list without requiring pre-market CFIA approval.
Claims with health and safety implications would be those that, if the feed ingredient fails to perform as claimed, the failure would result in an adverse animal health effect or food safety risk or both. An example of such a claim would be for an ingredient which claims that it binds a mycotoxin.
The CFIA would continue to require pre-market assessment and authorization of health and safety claims associated with new ingredients to any market (including Canada's even if authorized elsewhere) or for authorized ingredients for which a new intended purpose and claim(s) may have health and safety implications. Once authorized, health and safety claims associated with specific ingredients could be added to the positive list of permissible claims. For general production, nutrition, nutritional disorder and other non-health and safety feed claims, the list would be prepared and updated collaboratively by the CFIA, Health Canada and regulated industry.
For claims which do not appear on the list, they would require a review by the CFIA and Health Canada to determine whether there are health and safety implications and whether they constitute a claim consistent with a feed purpose. For claims which are determined to be feed claims and for which there are no health and safety concerns, the claim would be added to the list of permissible claims without further verification of the claim. Claims would need to be truthful and the proponent would need to have the evidence on hand of a product's capacity to perform as claimed.
To accommodate the concept of a positive list of permissible claims that could be managed in a flexible, timely manner, the Regulations would be designed to set out broad criteria for permissible claims so that a list could be included in guidance and administered by:
- the CFIA;
- industry (such as a code of practice); or
- a third party (for example, claim guidance modelled on the Low Risk Veterinary Health Products (LRVHP) Interim Notification Program for products for sale in Canada that are for use in cats, dogs, and horses that are not intended for food.)
A positive list of authorized feed ingredients might allow for the inclusion of categories for Permissible Performance Claims and Permissible Nutritional Disorder Claims. A pre-approved list of allowable claims would be established collaboratively with Health Canada. A proponent would provide supporting data to establish the utility of the ingredient for the specific claim. Once the ingredient was listed, it would not require further pre-market authorization for the claim and the claim may be permitted to carry forward onto a mixed feed product without triggering an additional pre-market assessment and authorization.
As suggested by commercial feed industry stakeholders, the CFIA would work collaboratively with Health Canada and stakeholders to identify and assess regulatory and non-regulatory options regarding the establishment of a permissible claims list and proceed accordingly with the outcome of the assessment in a manner that will respect both regulatory authorities. Pre-market assessment and registration of non-health and safety claims will still be considered by the CFIA if requested by an applicant for the purpose of marketing advantages or access.
In lieu of pre-market authorization by the CFIA of claims, ingredient manufacturers or distributors could choose to use permissible claims on their products from the pre-approved list. They would, however, be obliged to make information available upon request by customers or the CFIA to demonstrate their claims are truthful in a post-market oversight context.
Expected benefits of a liberalized approach to allowing non-health and safety claims to be used on ingredient labels would be that ingredient manufacturers and distributors could more easily and quickly put products on the market with permissible claims.
Have your say:
The CFIA is seeking comments on the proposals to:
- design the Regulations to set out broad ingredient claims criteria;
- reduce the scope of pre-market assessment requirements on claims to apply only to ingredients whose failure to perform as claimed may result in adverse effects on health and safety;
- collaborate with stakeholders to develop a permissible claims list for feed ingredients;
- oblige ingredient manufacturers or distributors to make information available upon request by customers or the CFIA to substantiate their product claims in a post-market assessment context.
5.5 Assessment Outcomes
Within the current framework, once an ingredient goes through the assessment process, there are three possible outcomes with respect to its authorization:
- Not authorized - If the applicant has failed to provide or satisfy the assessment requirements or if the assessment determined that the ingredient cannot safely be used for its intended purpose, or that the evidence does not support the intended purpose, the application is returned and the ingredient is not authorized.
- Authorized – no registration requirement. If ingredient safety and purpose are demonstrated, and no post-authorization restrictions are necessary due to source and specificity of the ingredient, it may be granted an authorization as a new ingredient with no further registration requirements. The ingredient definition may include restrictions such as maximum use rates, required guarantees, cautions or warning statements, or maximum contaminate levels.
- Authorized – requiring registration. If the safety and the intended purpose of an ingredient is demonstrated but it is determined that there are potentially significant variations between sources that have direct impacts on either the safety or performance of the ingredient, the ingredient is authorized and available for use, but each new source requires pre-market assessment and registration. In addition, the ingredient definition may include restrictions such as species restrictions, maximum use rates, required guarantees, cautions or warning statements, or maximum contaminant levels.
As a modernized approach, it is proposed that the CFIA maintains the above three assessment outcomes for pre-market authorization and registration; however, flexibility will be added to the framework for the following situations:
- The outcome "Authorized – requiring registration" would require consideration for registration of each source based on safety only where it has been determined that variation that could impact on the introduction or control of hazards. The efficacy determinants for this category will be removed;
- If either the CFIA or the applicant/manufacturer request that:
- the data submitted for the new ingredient application be kept in a proprietary nature (see discussion Section 6.3 below); or
- voluntary registration is desirable to meet proponent or trade needs; and
- Enable the timely marketing and use of new ingredients upon authorization and prior to listing in the Schedules.
As outlined in Section 3 above, authorized feed ingredients are eligible for listing in Schedule IV (general ingredients) or Schedule V (flavouring ingredients) in the Regulations upon authorization. Both Schedules are divided into two Parts which serve to differentiate the degree of post-authorization oversight applied by the CFIA for ingredients listed in each part:
- For ingredients whose source does not pose a safety concern or when risk can be mitigated by restrictions found in the definition (e.g. use restrictions, warning/caution statements, maximum levels of contaminants), they will be added to Part I of Schedules IV or V. Consequently, any new source of such an authorized ingredient does not require further assessment provided it meets the definition.
- For ingredients where there may be potential safety risks dependent on the source of the ingredient, they will be added to Part II of Schedules IV or V. The determination of variation for a source includes such factors as method of manufacturing, location of mine site and variation in microbial strains. Proponents must register their specific ingredient source. Ingredients that are registered and are listed in Part II of Schedules IV or V can then be used in the manufacture of mixed feeds.
To the extent that is possible and reasonable to do so in consideration of product purpose and safety, the objective of this modernized approach would be to authorize and list ingredients as non-proprietary in order to expedite their access to the Canadian market and minimize the burdens of registration and renewal of registration over time. Proprietary authorizations would be used only in situations where safety or other considerations warranted oversight of each source.
Have your say:
The CFIA is seeking comments on the proposals to:
- maintain three assessment outcomes for pre-market authorization and registration of feed ingredients; and
- add flexibility to the outcomes for specific situations.
5.6 Consultation on Proposed Outcome (New)
To enhance the transparency of the ingredient authorization process and meet the CFIA's requirements for consultation, it is proposed that the CFIA consult with stakeholders on the results of its ingredient assessments and a recommended outcome prior to final authorization. The CFIA proposes that a post-assessment notification and consultation approach be taken prior to final authorization for ingredients which have been assessed and found to be acceptable for inclusion in the list of authorized ingredients by the CFIA. The applicant would be notified and asked to supply a description of the product and relevant information that was presented to the CFIA for assessment. This information would then be posted on the CFIA website, and notified via listserv, for a short period (2-4 weeks) of public review and comment. The CFIA will accept input from the public on the proposed decision and will consider scientific or economic rationale submitted in favour of or opposed to the decision. The CFIA would establish a listserv when posting a post-assessment notification.
This approach is being proposed in order to increase the transparency of decision-making and provide regulated parties, the public and trading partners with an opportunity to comment on what ingredients are being authorized for feeding to food-producing animals. Such an approach may provide preliminary indications of potential consumer or domestic/international market access issues related to the acceptability of feed or food products for reasons other than safety or quality. For example, at one time, poultry manure was an authorized ingredient for feeding to beef cattle in Canada. Before the authorization was repealed in 2000 due to risks associated with transmitting Bovine Spongiform Encephalopathy (BSE) to cattle, the authorization gleaned some public attention and objections in the late 1990s.
Following the public consultation, the CFIA will consider any comments received and either finalize the authorization of the ingredient or go back to the applicant for more information to address any issues raised during the consultation period. Any comments received would need to be valid in order to be considered. Once given final authorization, CFIA would begin the process to add the ingredient to the appropriate list of authorized ingredients.
In the current ingredient authorization framework, there is no consultation on the outcomes of Agency assessments. The CFIA authorizes the feed ingredient and notifies the applicant. The CFIA then begins the process to add the ingredient to the appropriate schedule of authorized ingredients.
This proposal supports the Government of Canada's Open Government initiative and will increase the transparency of the CFIA's ingredient authorization process. Engagement, communication and timely updates of positive lists of authorized ingredients are important steps in providing transparency of decisions taken. During the consultation process to date, stakeholders identified a lack of timely access to current information as an issue. To enhance the transparency of the ingredient authorization process and meet the CFIA's requirements for consultation, it is proposed that the CFIA consult with stakeholders on the results of its ingredient assessments and are commended outcome prior to final authorization.
For ingredients which are not found acceptable or which have their application withdrawn by the applicant, if a "Notification of Submission" system is used (as per the above proposal) this information will be reflected on that notification page, but no further information will be published. If a Notification of Submission system is not used, then just the proponent will be notified if their ingredient is not approved.
The anticipated benefit of post-assessment consultation would be to improve the transparency of decision-making regarding the assessment and authorization of feed ingredients, contribute to the CFIA's consultation requirements, provide additional rigor in the science-based systems and provide stakeholders with an opportunity to input on the decision to be taken.
Have your say:
The CFIA is seeking comments on the proposal to increase the transparency of decision making on the ingredient authorization process.
5.7 Positive List of Authorized Feed Ingredients (Modernized)
Canada has benefitted from employing a positive list of authorized feed ingredients in Schedules IV and V, and this use of a positive list approach continues to be supported by stakeholders in consultations to date. The current list already contains over 1200 ingredients that are categorized according to intended purpose, are described, and contain the appropriate guarantees as necessary. Only those ingredients listed in these schedules may be used in the manufacture and sale of feeds in Canada, or in feeds imported into Canada.
The following proposal is made with regards to modernizing the positive list approach for authorized feed ingredients:
- Use the current version of the consolidated Schedules IV and V as a starting point in a phased approach for developing an updated positive list. Discussions with ANAC representatives in April 2013 have confirmed the feed industry's interest in collaborating with the CFIA to review and update the list, ideally in advance of the modernized regulatory framework coming into effect. This may be a longer term project and will:
- review existing ingredients for alignment with their descriptions, compositional criteria, current science, intended use and safety to animals, humans and the environment;
- establish the continued agronomic relevance and market demand as defined by livestock feeding practices, manufacturing practices, and modern marketing and trade implications;
- Create new categories or groupings of categories for ingredient types based on intended purpose, especially for the Class 8 Miscellaneous Products listed in Schedule IV. The current list has a number of general ingredient categories such as protein feeds, energy feeds, vitamins and minerals as well as a miscellaneous category. These existing categories no longer reflect the types of feed ingredients that are being used and the creation of new categories will allow ingredients to be better classified based on their intended purpose. At the same time, merge Schedule V (flavouring ingredients) into Schedule IV towards the creation of one, comprehensive positive ingredient list.
- Consider merging Parts I and II of the schedules into a single list. The CFIA would review the ingredients currently listed in Part II and re-assess the risks associated with having them included in Part II. Certain categories of ingredients, for example, fat-soluble vitamins, could be moved into Part I but it may still be advisable to maintain Part II to accommodate ingredients where each source are variable and can impact safety (for example, bacterial cultures, manufacturing process etc.) The addition of use and other restrictions to the ingredient descriptions may mitigate the risk and allow for greater inclusion of ingredients in Part I from the current Part II list.
- The regulatory process will allow for making amendments to the purpose of ingredients currently on the positive list. Such amendments would require the submission to the CFIA of data to support the proposed amendments. The required information would be the same as that required with a new ingredient application.
A well-defined list of authorized ingredients not only allows for a positive listing of ingredients, but is a useful risk management tool that includes categorization of ingredients which can make clear which ingredients require more stringent controls. Positive ingredient lists are employed by other major trading partners (e.g. U.S., EU) so continuation of using this approach is consistent with international practices.
Have your say:
The CFIA is seeking comments on the proposals to:
- expand the number of classes of ingredients in the positive lists to more clearly identify their intended purpose;
- merge the existing positive lists of authorized ingredients into a single, comprehensive list.
5.8 Post Authorization Conditions (New)
Under section 4.4 of the current Regulations, post-authorization conditions are required with respect to the release of novel feeds only. To some extent, taking action on the registration of certain authorized ingredients at present offers some measure of post-authorization control but there are otherwise no explicit regulations at present that could be applied in this regard to ingredients.
It is proposed that the scope of post-authorization conditions be expanded to apply to ingredients with respect to:
- Requirement to notify if manufacturing changes - Following the authorization and inclusion of ingredients in the positive list of authorized ingredients it is proposed that the Regulations include a requirement for proponents to notify the CFIA and provide supporting information in the event that there are changes in how the ingredient is manufactured or any risks associated with it. Changes to the manufacturing process for an ingredient may result in ingredients which are non-compliant with their definition. Changes in the manufacturing process would include:
- any changes to the raw materials or additives being used to manufacture the ingredient that may introduce different or additional hazards;
- any changes to the manufacturing process that may introduce different or additional hazards; or
- any change to the characteristics of the ingredient.
The ingredient manufacturer would be expected to initiate a hazard identification process of their own and communicate adjustments to their preventive controls as part of this process.
- Enable the CFIA to reassess an authorized ingredient if there are risks raised, including in other jurisdictions. To ensure that the CFIA is able to maintain its strong commitment to feed safety, it is also proposed that the Regulations include a mechanism to allow the CFIA to reassess any authorized ingredients should risk regarding them be raised. This can include results of CFIA sampling and monitoring activities, new scientific data, or information concerning the ingredient in other jurisdictions, generated by the applicant or in peer-reviewed scientific literature.
Expanding the scope of post-authorization conditions to authorized ingredients would provide the Agency with authorities to re-assess and take action on ingredients whose safety comes into question following authorization, especially if there is a reduction in the number of ingredients that are required to be registered in future.
6 Other Areas of Potential Regulatory Change
6.1 Processing Additives
Processing additives are defined as additives used at low levels to improve the manufacturing process of an ingredient, or its production process. These additives must be considered during the description of the feed ingredient manufacturing process, and are assessed during the new ingredient safety assessment. These additives are in themselves not considered to be "feed ingredients", and as such they will not result in individual ingredient listings in the approved list of feed ingredients. Their safety must, however, be demonstrated within the constraints of the new feed ingredient submission for approval.
Part of the consideration could simply be a description of the processing additive used, its use rate in the manufacturing process and a reference to an existing approval in feed or food use, either domestically or internationally.
For processing additives which have no prior authorization in this context, and that are not contained on any of the acceptable lists, scientific data will need to be submitted (e.g. could be by the additive supplier) for a feed safety assessment. If the additive contributes a significant concentration in the final ingredient, a more extensive safety assessment may be required.
Acceptable reference lists could include:
- The Feeds Regulations (Schedule IV, Part II), which is available upon request from the Animal Feed Division, CFIA;
- The Food and Drug Regulations, Division 16, lists additives considered to be a food additive;
- The Food and Drug Regulations, Division 28, lists additives considered to be a food, but not considered to be a novel food; and
- The Canadian Environmental Protection Act lists enzymes and Saccharomyces cerevisiae strains in the Domestic Substances List (DSL) and is in compliance with the New Substance Notification Regulations (Chemicals and Polymers, and Organisms).
6.2 Definitions
Well-defined terms are important for clear understanding of regulations and policies. A number of definitions are included in both the Act and the Regulations, as well as in policy. Consultation with stakeholders demonstrated a need to clarify some of the terms that are currently used.
In their submissions, ANAC has made several recommendations related to the interpretation and amendment of current definitions as well as on the adoption of new ones. The recommendations related to definitions were discussed in plenary with stakeholder groups that participated in the CFIA's Feed Ingredient Classification and Authorization Workshop in January 2013.Subsequent to the workshop, ANAC provided the CFIA with a supplementary paper in March 2013 that also included other definitions-related recommendations. The collective of ANAC recommendations regarding definitions is provided in Annex I.
In particular there is an overlap between the legislative definitions of "feed" and "drug" which creates a challenge whereby products may be classified as a feed or a drug, or both. The definition for "feed" is found in the Feeds Act and the definition for "drug" is found in the Food and Drugs Act. It is not as easy to make changes to these definitions and opportunities for amending either of these acts in the near future are not anticipated. To address this need, stakeholders have proposed, and the CFIA agrees, that a clearer interpretation for feed is needed. This includes defining:
- "nutrient";
- "nutritional disorder"; and
- "feed ingredient".
In its own analysis to date, the CFIA has also concluded that other current definitions related to feed ingredients require review and that modifications and additional terms may be warranted to correspond with modification to the framework itself and other specific needs. The CFIA will investigate the best tool – using policy or regulation – to address the need for defined terms.
6.3 Mechanism for Proprietary Approvals: per ANAC Recommendations
Under the current system, there is no specific mechanism which provides for exclusivity of data that is used during the approval process. Especially for ingredients which are authorized and listed in the schedules, once the initial assessment and approval is complete, anyone may market the ingredient from their own source. During consultations, stakeholders proposed that a mechanism for data exclusivity be considered. This data exclusivity mechanism would:
- recognize and protect feed ingredient innovations;
- encourage new ingredient development; and
- allow sponsors to recoup their research and development costs.
Under the proposed mechanism, new ingredient sponsors conducting the required development activities would be eligible for five years of data exclusivity from the time the ingredient is approved for use in Canada. To qualify, ingredient sponsors would make a formal request to the CFIA for exclusivity in advance. If another company wishes to market the same ingredient (assuming no patent protection) they would be allowed to do so as long as they generate their own data set. At the end of the five-year data exclusivity period, the ingredient would be added to the positive list and will thereby be made generally available. This would work in conjunction with the above proposal for tiered outcomes of assessments, but would introduce a five-year gap between the time the ingredient is approved and the time it is added to a list for general use. In that time period, a list of these proprietary approvals could be published administratively but anyone wishing to market a new source would need to submit an application and a full data package in order to have their product approved.
Have your say:
The CFIA is seeking comments on including a mechanism to provide data exclusivity.
6.4 Incorporation by Reference
Timely amendments to schedules that form part of the Regulations can be problematic. Based on the experience of the CFIA and feedback received from stakeholders, it is important to continue to have a positive list of authorized feed ingredients, but the list needs to have flexibility so that it can be maintained and updated in a timely fashion. Delays to the update of this list are frequently cited as an irritant to stakeholders.
One of the regulatory tools the CFIA may pursue in respect of increased flexibility and improved timely updating is the ability to incorporate by reference documents into regulations under an act that are externally or internally generated, and that may change over time.
Incorporation by reference is a term used to describe a means to allow a document, not in the body of the regulation, to be made part of the regulation. Such documents could be technical or non-technical standards, methods and guidelines.
The legal effect of incorporation by reference is to write the words of the incorporated document into the regulation as if it had actually been reproduced word for word. Documents incorporated by reference have the force of law.
Incorporation by reference helps to reduce delays in making regulatory changes. In terms of the responsibilities of the CFIA, the use of appropriately incorporated documents could make the food safety regulatory system more responsive. For example, instead of needing to initiate a regulatory change to revise a list set out in the regulations, a list developed by the CFIA and able to be incorporated by reference on an ambulatory basis could be revised by the CFIA, usually following consultations with stakeholders and the public, without amending the regulation itself. In addition, the CFIA would be able to incorporate international or third-party standards into the regulations, where appropriate, rather than reproduce the standard in the regulation itself.
6.5 Additional Anticipated Benefits of the Proposed Ingredient Authorization framework
The proposed framework for assessment and authorization of new or modified feed ingredients will result in additional benefits, including for some mixed feeds. It is proposed that some feeds which currently require pre-market registration will no longer require it. The following are examples that are directly related to this feed ingredient assessment and authorization framework:
6.5.1 Expanded Definitions
Ingredients that are included on the list of approved ingredients may be allowed to have flow agents, pellet binders, preservatives, antioxidants, etc. added provided there are no restrictions on the ingredient to prevent the inclusion of these items and the added items are also on the list of approved ingredients. Specific combinations of these ingredients may be restricted due to health and safety reasons if applicable.
6.5.2 Diluted Ingredients
Ingredients which are listed on the list of approved ingredients may be diluted to create a less potent ingredient without the requirement for reassessment or registration provided the item used as a diluent is also an approved ingredient and the initial ingredient doesn't have any restrictions on mixing or diluting. Any required guarantees would need to reflect the diluted product and any directions for use must reflect the lower concentration of the ingredient.
6.5.3 Specialty Additive Feeds
There are a number of products which under the current system are considered mixed feeds that require registration, although they are used in a manner more similar to ingredients. This would include things such as pelleting agents, anticaking agents, enzyme supplements, and fermentation product supplements. These types of feeds will need to be reviewed with the intent to assess whether any would be more appropriately considered an ingredient or if they may be exempted from the registration requirements.
6.5.4 Mixed Feeds (More Generally)
The greatest impact will be offered "downstream" to commercial feed manufacturers with the elimination of many mandatory mixed feed registration requirements where authorized ingredients are used to make premixes, supplements, complete and other types of mixed feeds. Given that authorized ingredients are safe and fit for their intended purpose, the need for pre-market assessment and registration of mixed feeds containing authorized ingredients in many cases may no longer be pertinent. For example, Table 4 of Schedule I in the current Regulations (prescribed ranges of nutrients for regulated livestock species as a basis for exempting mixed feeds from mandatory registration) will likely be eliminated from the modernized regulatory framework.
Further positive impacts on the registration system for mixed feeds will be explored in a future proposal.
7 Summary
7.1 Proposal Synopsis
The proposed changes to the feed ingredient regulatory framework are one part of the overall approach to modernizing Canada's Feed Regulations. Comments and feedback on the proposal, from stakeholders and other interested parties will contribute to the development of regulations that are flexible, effective and in step with current technologies, feed and livestock production systems and markets. Stakeholders are encouraged to provide written feedback on both the components that they support and those which they are not in agreement with.
7.2 Next Steps
Feedback obtained during this consultation will be combined with feedback from the other three modules. A summary document, which will integrate the information from the four separate modules, will be published following the individual consultations. This summary document will outline the overall proposed feed framework and will provide an additional opportunity for stakeholder input. Following the development and publication of the proposed overall framework, the CFIA will review its service delivery costs and fees within the Feed Program as part of an Agency-wide User Fee modernization initiative.
Objectives for the User Fee initiative include the promotion of effective and responsive service delivery; establishing a consistent and robust approach to user fee development; and fostering an understanding that user fees may be charged for services, products, rights and privileges provided by the Agency. For more details, see the Cost Recovery Policy and Framework.
The Agency will hold public consultations on user fees related to services provided by the Feed Program. Service recipients will have an opportunity to comment on the detailed user fee proposal once it is made public.
7.3 Contact
Your input and feedback is critically important to the success of the regulatory modernization initiative and we strongly encourage you to provide your input. Written comments can be sent to:
Sergio Tolusso
Canadian Food Inspection Agency
Animal Feed Division
59 Camelot Drive
Ottawa, ON K1A 0Y9
Email: Sergio Tolusso
Fax: 613-773-7565
Annex
Summary of Recommendations submitted by the Animal Nutrition Association of Canada related to Definitions
Modernizing the Feeds Regulations - A Proposal by the Animal Nutrition Association of Canada for a Risk-Based Framework for Feed Ingredients (January 2013)
Recommendation 1 - Maintain the definition of "feed" as contained in the Feeds Act.
Recommendation 2 - Using the current feed definition as cited in Recommendation 1, it is recommended that additional emphasis and a revised interpretation be applied as follows:
1 - For part "b" of the feed definition, which indicates "providing the nutritional requirements of livestock", it is recommended that this definition allow for the use of feed ingredients for the optimization of growth and production efficiency of animals.
2 - In the case of part "c" which states "for the purpose of preventing or correcting nutritional disorders of livestock", within this definition the role of feeds in correcting and mitigating various metabolic and sub clinical nutritional disorders e.g. sub-acute ruminal acidosis, ketosis, and subclinical necrotic enteritis should be permitted given the nutritional etiologies of these syndromes.
Recommendation 3: For the purposes of the modernized regulations, and the Canadian definition of a nutrient, it is recommended that the USDA (Animal diets and Feed Management, Nutrient Management Technical Note No. 8, January 2012) definition for nutrient be adopted and refined to state:
A nutrient is "Any ingredient or compound in the diet that supports reproduction, growth, lactation, or maintenance of life processes".
Recommendation 4: In terms of a definition for nutritional disease, it is proposed that the following definition be adopted:
A nutritional disorder is "Any disorder in animals that is directly or indirectly caused by a lack of an essential nutrient or conditionally essential nutrient (those that must be supplied only under special conditions, such as stress, illness, etc.) in the diet, or a nutritional imbalance."
Recommendation 5: Adopt a simplified overarching feed claim definition. Combining and modifying a number of Codex definitions, the following is recommended:
Claim: "Any representation which states, suggests or implies that a feed has particular characteristics relating to its origin, nutritional properties, nature, production, processing, composition or any other quality that impacts an animal's productivity and health."
Supplement to the January 2013 Paper on Feed Ingredient Regulatory Modernization (March 2013)
Feed Ingredient Definition
Clear definitions will be required in the modernized regulations. To complement the definitions proposed in our January 2013 report (Recommendations 3, 4, and 5) ANAC proposes the following feed ingredient definition:
"any substance or mixture of substances for use in water or feed other than drugs, micro-premixes, macro-premixes, mineral feeds, supplements and complete feeds."