This page has been archived
Information identified as archived is provided for reference, research or record-keeping purposes. It is not subject to the Government of Canada Web Standards and has not been altered or updated since it was archived. Please contact us to request a format other than those available.
This consultation closed 2014-07-25
Table of Contents
- 1 Purpose
- 2 Background
- 3 Current Situation
- 4 Proposed Regulatory Framework
- 5 Implementing Proposed Regulatory Framework
- 6 Summary
- Annex 1 - FRSG Recommendations
- Annex 2 - Proposed Outcome-based Regulatory Requirements
1 Purpose
This document outlines the principles, objectives, and proposed direction of a modernized regulatory framework that would apply to the identification of hazards in livestock feeds, the management of risks they may pose, and the establishment of preventive controls. The Canadian Food Inspection Agency (CFIA) seeks broad stakeholder comments and input on the regulatory proposal.
This proposal is one of a series of modules that have been prepared as part of the Feed Regulatory Renewal project.
2 Background
2.1 Change Management at the CFIA
The CFIA has embarked on a comprehensive change agenda to strengthen its foundation of legislation, regulatory programs, and inspection delivery. These directions set the context for renewal of the Feed Regulations (Regulations).
In December 2011, the CFIA initiated a systematic, multi-year review of its regulatory frameworks for food safety, plant health, and animal health. Through this structured and comprehensive review, the Agency intends to update its regulatory frameworks to
- reduce overlap and redundancy,
- increase responsiveness to industry changes,
- address gaps, weaknesses, and inconsistencies, and
- provide clarity and flexibility to affected regulated parties.
The modernization of the Regulations has been identified as a short-term priority in the first three years of the modernization initiative.
The Agency will continue to explore opportunities for modernizing its legislative base as regulatory modernization takes place. For feeds, Canada's Minister of Agriculture and Agri-Food tabled the Agriculture Growth Act (Bill C-18) in Parliament, proposing to amend several federal agricultural statutes, including the Feeds Act (Act). Among the suite of proposed amendments, a new authority to make regulations "respecting quality management programs, quality control programs, safety programs, preventive control plans (PCPs) or any other similar programs or plans to be implemented by persons who conduct any activity regulated under this Act" would be provided to the CFIA.
The Agency is also engaged in developing a new inspection delivery model, the Integrated Agency Inspection Model (iAIM), which is based on common inspection activities and standard processes for all its inspection programs, and is more focused on risk as a basis for prioritizing inspection activities. This new model, supported by a renewed training regime, is being pioneered for food inspection, but will be adapted, as appropriate, to help streamline and create efficiencies for other inspection activities, including feed.
2.2 Feed Regulatory Modernization
Modernization of the Regulations strives to reduce compliance burden and to support innovation while maintaining animal and human health, as well as environmental and economic sustainability.
The current feed regulatory framework is product-based, including pre-market assessment and approval, or registration of single feed ingredients and any mixed feeds (comprised of one or more ingredients) that do not meet exemption from registration criteria. In addition, the Regulations include requirements for labelling of feeds to provide accurate information.
The Agency maintains marketplace oversight of ingredients and feeds to verify they are compliant with the composition, labelling, and registration standards of these Regulations.
The modernization of the Regulations is being designed to benefit the collective Canadian feed industry, which includes commercial feed manufacturers, retailers, importers, exporters, ingredient manufacturers, livestock producers, and consumers. As well as aligning, where possible, with other international feed regulatory regimes, modernization also maintains the objective of safeguarding animal health, plant health, food safety, and environmental safety for the Canadian public.
The ultimate objective of the Regulations' review is to develop a modernized risk- and outcome-based regulatory framework for feeds that
- supports fair and competitive trade in the market;
- minimizes regulatory burden; and
- protects public, animal and plant health, and the environment.
2.3 Principles of CFIA Regulatory Modernization
The following principles guide the overall CFIA regulatory modernization process:
- protecting public safety and confidence in the inspection framework;
- enabling an environment of improved business opportunity and consumer choice by facilitating innovation and competitiveness;
- establishing clear policy objectives through consultation with partners – including industry, consumers, small business, other departments, and provinces;
- finding the appropriate balance between administrative costs and benefits derived from regulatory intervention;
- developing consistent, transparent and, where appropriate, outcome-based regulations with performance measurements;
- recognizing the vital importance of science and risk management approaches in decision making; and
- aligning, to the extent possible, with international trading partners and provincial standards.
2.4 Consultation Process and the Role of this Proposal
Stakeholder consultation is an important component of regulatory modernization.
During 2012, the Agency took a three-tiered approach to initiate pre-consultation on this comprehensive project. First, informal bilateral meetings were held with stakeholder groups. A subsequent discussion paper and survey were made available online to provide all stakeholders with the opportunity to provide feedback. Finally, a two-day multi-stakeholder workshop was held in September 2012.
Following the September 2012 workshop, there was a need for further stakeholder consultation. An industry and government Feed Regulatory Steering Group (FRSG) was formed in November 2012, with members representing the commercial feed manufacturers, major national livestock producer associations, the CFIA, and Health Canada's Veterinary Drugs Directorate (VDD). Early on, the FRSG determined that feed regulatory modernization was sufficiently complex, and thus should be broken down into the following three components ("modules") for further discussion:
- Feed Ingredient Assessment and Authorization
- Feed Hazard Identification and Preventive Controls (possibly including facility licensing)
- Feed Labelling
Modules 1 and 3 have been posted for public review and comment; consultation closed on December 5, 2013.
A March 2013 workshop was held with interested stakeholders to discuss the topic of feed hazard identification and preventive controls.
The information provided and gathered from all these activities forms the foundation of this proposal, which outlines what direction the Agency envisions for the identification and management of hazards, associated with feeds, on public health, animal and plant health, and the environment.
3 Current Situation
The scope of the Feeds Act applies to the manufacture, sale, and importation of feeds in accordance with regulations that set out registration and labelling requirements and standards for feeds.
Whether manufactured domestically or imported, certain feed ingredients such as vitamin A, D, and E sources currently require mandatory CFIA registration prior to manufacture, sale, or entry into Canada. If not required to be registered, imported feed ingredients are otherwise permitted entry into Canada, provided they have been authorized for use as an ingredient in livestock feed in Canada and comply with standards established for the authorized ingredient.
Domestic mixed feeds are largely exempt from mandatory pre-market assessment and registration by the CFIA. Domestic feed manufacturers, retailers, and some farms are inspected by the CFIA on a risk-based frequency to verify compliance with current regulatory requirements, including compliance with requirements set out in the Health of Animals Regulations, respecting the restrictions ("feed ban") related to preventing the transmission of bovine spongiform encephalopathy (BSE). All imported mixed feeds must be registered by the CFIA prior to entry into Canada.
Livestock producers who manufacture feeds are exempt from the Act and any of its associated regulations, provided the feeds they manufacture are neither sold nor contain any drug or other substance that may adversely affect human health or the environment.
Currently, the requirements in the Feeds Regulations regarding feed safety, composition, and quality focus on the feed products themselves and not on processes employed in feed manufacturing. A number of standards are set in regulation regarding the form and composition of the feed.
3.1 Feed Hazards
The CFIA takes a three-tiered approach to identifying hazards and providing guidance on maximum acceptable levels in feed:
- In regulation – In some cases, hazards are identified in the Regulations. This includes a list of deleterious substances that are mainly prohibited pesticides, a list of prohibited weed seeds, maximum levels for substances that are likely to be deleterious to livestock such as fluorine and aflatoxin and other extraneous material (e.g. mould, heat damage, or sawdust). Further, feed ingredient definitions may contain additional standards such as a standard for glucosinolates or a maximum use rate to allow safe use of the ingredient.
- In guidance – The CFIA provides administrative guidance on a number of other hazards in feeds such as mycotoxins, metals, dioxins, furans, and PCBs. In addition, the guidance sets out action levels for many of the individual contaminants. A feed Action Level is defined as the level at which, if exceeded, a contaminant may present a health risk to the animal or result in unacceptable residues in foods of animal origin.
- Ad hoc assessments – The CFIA also undertakes ad hoc assessments of hazards in feeds for which maximum or action levels are not specified in the Regulations or guidance. Often, these assessments are undertaken in response to accidental contamination of food and feed products that is reported by regulated parties or identified by sampling.
3.2 Feed Preventive Controls
With respect to preventive controls for hazards that may be present in feeds, requirements for such controls are not included in the current Regulations, nor do they contain specific process or other control provisions, or identify who is responsible for their administration.
Alternatively, the CFIA has implemented a number of control measures since 1997, using authorities provided by the Health of Animals Act, to address risks associated with the transmission of BSE via feeds along the feed and livestock production chain. The hazards are identified and defined in the Health of Animals Regulations as "prohibited material" (mammalian proteins with some exceptions) and "specified risk material" (certain cattle tissues such as brain and spinal cord from animals over 30 months in age). To manage risks posed by these materials in the production system, preventive control measures are present in the Regulations, requiring that
- operators of rendering plants use dedicated production lines, equipment, and conveyances to manufacture and distribute prohibited and non-prohibited animal protein products to prevent cross-contamination;
- operators of establishments have procedures to prevent the mixing or contamination of food for ruminants with prohibited material; and
- manufacturers, importers, distributers, and vendors of animal food establish and maintain procedures to facilitate effective recall.
Even though more comprehensive regulatory requirements are not currently in effect, industry group programs have developed and implemented a number of sector-specific food-safety-oriented preventive control programs nationally in recent years.
Here are some examples:
- Animal Nutrition Association of Canada (ANAC) – FeedAssure
- Livestock sector programs:
- Canadian Pork Council – Canadian Quality Assurance
- Dairy Farmers of Canada – Canadian Quality Milk
- Grain and oilseed sector – Hazard Analysis Critical Control Points (HACCP)-based programs:
- Canada Grains Council – Grains, Oilseeds, Pulses and Special Crops Canadian On-Farm Food Safety Program
For these livestock and grain and oilseed programs, the CFIA provides government recognition via the Agency's Food Safety Recognition Program, which acknowledges that a food safety program has been developed in line with HACCP principles, as defined by the Codex Alimentarius Commission; conforms to federal, provincial, and territorial legislation, policy and protocols; and recognizes that a Food Safety Management System has been implemented in an effective and consistent manner.
Along with these domestic private industry programs, dozens of other international feed and food safety programs exist; specifically, GMP+, FAMI-QS , GLOBALG.A.P., and the American Feed Industry Association's (AFIA) Safe Feed/Safe Food Certification Program. Consequently, many commercial and farm-based feed manufacturers are already familiar with the concepts of preventive controls, have voluntarily enrolled in one of these programs, and are subject to audits to confirm they are conforming to program requirements. The commitment to improving feed safety, shown by those parties that are choosing to participate in one of these programs, is commendable and, in fact, demonstrates the progressive role many in the feed industry are already playing.
3.3 Record Keeping
Under the current Feeds Regulations, record-keeping requirements are limited to the following:
- Mixing formulae for customer formula feeds, consultant formula feeds, feeds described in paragraph 5(2)(d) (any complete feed, supplement or macro-premix manufactured in Canada that is designed to be fed to beef cattle, dairy cattle, sheep, swine, chickens, turkeys, horses, goats, ducks, geese, salmonid fish, mink, or rabbits) and veterinary prescription feeds; and
- Copies of customer formulae or veterinary prescriptions under which such feeds are manufactured, together with a list of each date on which the feeds were manufactured.
The Health of Animals Regulations require manufacturers of animal food for ruminants, equines, porcines, chickens, turkeys, ducks, geese, ratites, or game birds to
- keep records that contain
- the formula for the animal food, including the name and weight of each ingredient used for each lot of the animal food;
- a mixing sheet that shows that each lot of the animal food has been produced in accordance with the animal food's formula;
- information on whether the animal food contains any prohibited material;
- the date of preparation of the animal food;
- the lot number and any other information used to identify each lot of animal food; and
- the name and address of any person to whom any animal food is distributed or sold, along with a description of the food, including the name and quantity;
- ensure that persons who import, package, store, distribute, sell, or advertise animal food for sale retain the following:
- the name, the lot number, and any other information used to identify the animal food;
- the name and address of any person to whom the animal food is distributed or sold; and
- a description of the animal food, including the name and quantity; and information on whether the animal food contains any prohibited material;
- require that persons who own or have the possession, care, or custody of a ruminant keep copies of all invoices for animal food that contains prohibited material.
3.4 International Situation
The Codex Code of Practice on Good Animal Feeding (2004) (Code of Practice) provides guidance to help ensure the safety of food for human consumption by adhering to good animal feeding practice at the farm level and good manufacturing practices (GMPs) during the procurement, handling, storage, processing, and distribution of animal feed and feed ingredients for food-producing animals.
Other jurisdictions already have hazard identification and preventive control requirements in place or are proposing to further strengthen current measures:
- The European Union (EU) sets out a range of undesirable substances for animal feed by way of Directive 2002/32/EC Annex I of the Directive identifies undesirable substances such as heavy metals, aflatoxin, and certain pesticides. It also sets maximum levels for the identified undesirable substances:
- in some cases, for all feeds (ingredients, premixes, complete feeds); or
- specific undesirable substances in specific feeds (e.g. dioxin in citrus pulp).
The EU also has had comprehensive legislation and regulations regarding requirements for feed hygiene since 2005 (Regulation [EC] 183/2005), where "feed hygiene" is defined as "the measures and conditions necessary to control hazards and to ensure fitness for animal consumption of a feed, taking into account its intended use." Commercial feed operators are required to put in place, implement, and maintain a permanent written procedure, or procedures, based on HACCP principles that are set out in the regulations. At a farm level, livestock producers are required to comply with a set of good animal feeding practices also set out in the regulations.
- In the U.S., the federal Food, Drug, and Cosmetic Act, administered by the U.S. Food and Drug Administration (FDA), requires animal feed to be pure and wholesome, be produced under sanitary conditions, and not contain harmful substances. Animal feed manufacturers are responsible for ensuring that feed is truthfully labelled, that it does not contain unsafe additives or contaminants and, if the feed contains drugs, that the drugs are approved by the FDA for use in animal feeds.Footnote 1
The FDA considers feed contaminants to be toxic or deleterious substances, grouping into two classes:
- inherent, naturally occurring constituents of an animal food that are not the result of environmental, agricultural, industrial, or other contaminations – for example, some mycotoxins, such as aflatoxin and fumonisin, glucosinolates, and heavy metals such as lead and cadmium; and
- industrial substances, which are not naturally occurring and are amplified to abnormal levels in the animal food through mishandling or other acts. Examples include polychlorinated biphenyls (PCBs) and certain pesticides such as DDT.
The FDA may prohibit any detectable amount of a contaminant or establish a regulatory limit for the contaminant. Action is based on the protection of public health, the extent to which the presence of the contaminant cannot be avoided, and other ways in which the consumer may be affected by the contaminant.Footnote 2
With respect to preventive controls, the FDA has required compliance with a Current Good Manufacturing Practices (CGMP) rule for medicated feed manufacturing (21CFR225) since the 1970s. The rule comprises two tiers: 1) includes a more comprehensive set of CGMPs that apply to facilities using drug sources that require the facility to be licensed and registered by the FDA to manufacture medicated feeds and 2) applies to facilities that use drug sources not requiring facility licensing by the FDA. Examples of CGMPs requirements for facilities in either tier are those specified for facilities and equipment, product quality control, and packaging and labelling. Regardless of the drug source used, one or the other tier of the rule applies to medicated feeds made either in commercial feed mills or on farms, even if manufactured for the feeding of a livestock producer's own animals.
In late October 2013, to improve the safety of these products, the FDA published a proposed rule to establish more comprehensive CGMPs and Hazard Analysis and Risk-Based Preventive Controls for Food for Animals. The preventive controls provisions of the proposed rule, which are required by the FDA Food Safety Modernization Act, would apply to domestic and imported animal food, including pet food, animal feed, and raw materials and ingredients. Facilities producing animal food would be required to have written plans that identify hazards, specify the steps that will be put in place to minimize or prevent those hazards, identify monitoring procedures and record monitoring results, and specify what actions would be taken to correct problems that arise. The comprehensive hazard analysis rule is intended to address hazards that
- pose a risk to animal health (including appropriate nutritional levels, contaminants, and species specific hazard analysis); and
- pose a risk to human health, either through consumption of the animal products or direct handling of the animal food in the home.
The proposed rule would also establish certain CGMPs that specifically address animal food. The medicated feed CGMPs rule will remain in effect.
Written comments in response to this regulatory proposal were due to the FDA by March 26, 2014.
3.5 Consultation Outcomes to Date
In the 1990s, the CFIA developed and consulted extensively with the feed industry and livestock producer organizations on a proposed set of Medicated Feed Regulations. These regulations were designed to introduce process controls for the manufacture of feeds containing medications. Results from these consultations have been used to shape the development of this proposal.
To date, stakeholders have generally supported a process-based approach to feed regulation. During the pre-consultation stage, the following objectives were articulated:
- a need for process controls
- a need for tiered levels of control
- recognition of existing industry programs
Hazard identification and preventive controls were also included as part of the CFIA's 2012 discussion paper and stakeholder survey.
The survey indicated
- support for the inclusion of hazard identification and PCPs for feed manufacturing.
- support for higher levels of control or oversight for medicated feeds than that for non-medicated feeds.
- a majority of feed and livestock producers are already following a GMP or HACCP system.
- most were in favour of including risk assessment and identification of undesirable substances in regulation.
At the September 2012 multi-stakeholder workshop, the Code of Practice was used as a framework to guide discussions with stakeholders. The Code of Practice outlines a number of good manufacturing practices that should be followed, both on-farm and in commercial feed and ingredient production. In general, stakeholders were in agreement that the Code of Practice provided good starting principles. However, they also identified a need for the following:
- all sectors of the industry to undertake complete hazard identification
- a tiered risk assessment approach
- adequate information on acceptable levels of contaminants for some ingredients
- requirements that should align internationally
- additional guidance for smaller industry that does not have experience with PCPs
Building further on the results from pre-consultation activities in 2012 and in response to guidance provided by the FRSG in early 2013, the Agency staged a multi-stakeholder technical workshop on feed hazard identification and preventive controls in Ottawa in March 2013. These were the key points provided to the CFIA:
- Participants agreed upon three categories of relevant hazards (biological, chemical, and physical) and that animal health, food safety, and the environment are appropriately addressed by current identification practices (e.g. HACCP and good quality assurance and quality control systems).
- Participants expressed concerns that the Agency is avoiding the use of HACCP terminology as the accepted way to characterize approaches to proactively identify and prevent hazards.
- All agreed that, if there are preventive control programs (such as HACCP) in place to monitor hazards, and the government has some confidence in such programs, the appropriate role for the government should be to inspect that a facility or regulated party adheres to the program.
- There was consensus about making preventive controls a requirement as long as they are not prescriptive.
In addition to CFIA consultation activities, ANAC presented the Agency with a proposal in June 2013 for a Risk-Based Approach to Hazard Identification and Control for commercial feed manufacturers. ANAC proposed that the process be founded on six principles:
- mandatory tier-based registration/licensing of facilities
- the use of process systems and controls to mitigate risks
- risk-based regulatory oversight
- increased recognition of industry-led HACCP programs, particularly FeedAssure
- collaboration and transparency between industry and government
- modeled, where appropriate, on demonstrably successful international examples
In October 2013, the CFIA shared its current thinking regarding a proposed regulatory framework for feed hazard identification and preventive controls with the FRSG. Following this briefing, the industry association members of the group collaborated to provide the CFIA with feedback and six recommendations (Annex 1). A working draft of this proposal was provided to members of the group for their review in February 2014, prior to broader stakeholder distribution, and feedback from the group was incorporated into this proposal.
This proposal aligns with feedback obtained from the collective consultation activities that have been undertaken by the CFIA to date. The feedback from these consultations has been closely considered in the development of this proposal.
4 Proposed Regulatory Framework
4.1 Objectives of Hazard Identification and Preventive Control Plans
As the agriculture and food sectors in Canada and around the world continue to become more complex, the CFIA's relationship with industry needs to evolve. The CFIA will continue to work toward a common goal of effectively managing risk. While the Agency can play a greater role in promoting industry understanding and compliance with requirements, industry is ultimately accountable for ensuring the safety of its products and processes. Regulated parties and consumers have an equally important role in keeping Canada's food supply and plant and animal resource base safe.
Appropriate hazard identification and preventive control procedures for feeds are an internationally recognized method of preventing or mitigating the risks associated with foods and feeds.
Risks associated with feeds and ingredients can be prevented outright or mitigated, either through controls on the finished product or via a systems approach. A systems approach means that the products are produced, transported, and stored in a safe and consistent manner, thus ensuring that each batch of feed that is manufactured will meet the same standards. Product controls only verify that individual products meet standards. Inspecting and enforcing a process-based system provides the regulator and the consumer with greater confidence in the feeds that are produced under that system.
4.2 Scope
This proposal addresses the introduction of requirements in a modernized regulatory framework for hazard identification and preventive controls and includes discussions on the following:
- hazard identification and analysis
- preventive control plans
- record keeping
- roles and responsibilities
The mandate of the CFIA and the Feeds Act support the protection of public health, as well as animal health, plant health, and the environment. With this in mind, the Agency proposes the following scope for a risk-based feed hazard identification and preventive controls framework that would apply to
- the manufacture and distribution of feed ingredients and mixed feeds ("feeds") alike, representing an "end-to-end" approach throughout the feed supply chain.
- feeds for food-producing animals, including species not currently defined as "livestock" in the Act and Regulations (e.g. deer, elk, bison, and other mammals and birds that produce food products [milk, eggs] or are slaughtered and processed as a meat product for human consumption and for which an inspection system has been established [according to current "food animal" definition in the Meat Inspection Regulations]).
- domestically produced and imported feed (a discussion on possible approaches to the application of the framework on imported products is provided in section 5 of this proposal).
- hazards with human health, animal health, plant health, and environmental implications.
The proposed framework would require all those throughout the feed supply chain to whom the regulatory framework would apply to
- identify hazards that may be associated with their products and processes.
- design and implement a PCP to address the identified hazards that are appropriate for their specific system.
A risk-based framework would acknowledge reducing requirements for lower-risk operations and feeds.
Figure 1 illustrates the feed supply chain and how an integrated hazard identification and preventive control framework would apply throughout the continuum. All parties to which the regulatory framework applies would share in the responsibility of identifying hazards and putting in place effective preventive controls in their specific part of the chain.
Figure 1 - Hazard ID / Preventive Control
Click on image for larger view
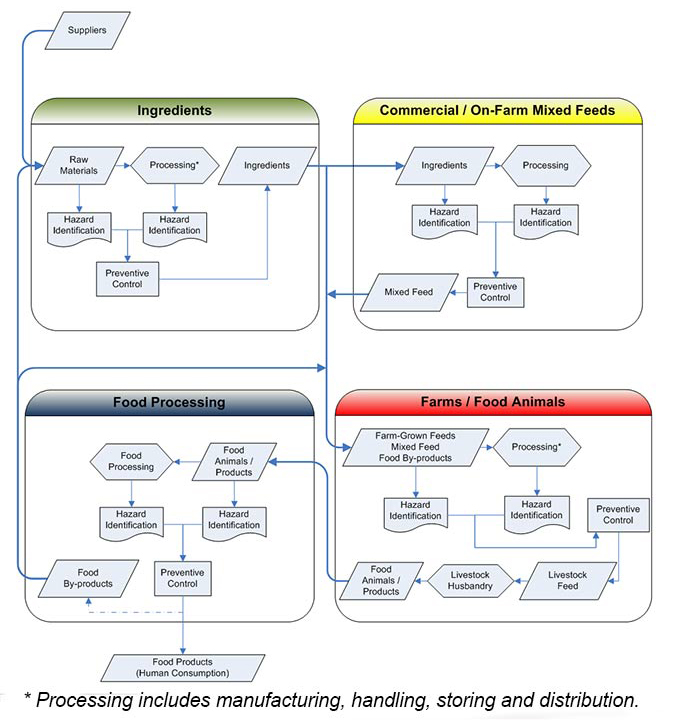
Description for Feed Supply Chain Continuum – Hazard Identification and Preventive Control image
This image is a flowchart that illustrates how the proposed hazard identification/preventive controls framework would apply to regulated parties involved in the supply chain linking the production of feed ingredients, mixed animal feeds and livestock prior to animals and animal products being delivered for food processing. The flowchart includes four frames depicting the sectors involved in feed, animal and food production that would be impacted by the modernized regulatory framework.
In each frame, the identified sector would be responsible for identifying any hazards in the incoming materials or products they receive as well as any that might be introduced by the processing practices that are used. Once identified, the sector would also have to develop and implement preventive controls in order to prevent or reduce any risks posed by the hazards prior to distributing their own products or animals to other frames in the supply chain. The sectors identified in the four frames of the flowchart are as follows:
- Commencing with a feed ingredient frame at the top left of the flowchart, raw materials are received from suppliers, including by-products from food processing, for the production of feed ingredients;
- In the second frame of the flowchart to the right, feed ingredients are received by commercial feed manufacturers or livestock producers on farms for the production of mixed animal feeds;
- In the third frame at the bottom right of the flowchart, mixed animal feeds are received from the second frame at farms and fed to food animals as received or in combination with other farm-grown ingredients (for example, hay or other forages, grains), ingredients received directly from ingredient producers in the first frame or even food byproducts received directly from food processors.
- In the fourth and final frame at the bottom left of the flowchart, animals and animal products (for example, milk and eggs) are received by food processors for the production of food for human consumption. This sector also generates by-products (for example, whey from milk processing, egg shells) that can be re-processed into ingredients for animal feeding. Consequently, this frame is connected to the other three frames as food by-products are received by feed ingredient producers, mixed animal feed producers and livestock producers on farm.
* Processing includes manufacturing, handling, storing and distribution.
Exemptions
Even with the proposed amendments to the Feeds Act, contained in the Agriculture Growth Act, referred to in sections 2.1 and 5 of the proposal, livestock producers who manufacture feeds will remain exempt from the scope of the Act and any of its associated regulations, provided the feeds they manufacture are neither sold nor have any drug or other substance incorporated into them that presents a risk of harm to human or animal health or the environment. Table 1 shows the application of the proposed framework to livestock producers.
| Feeds manufactured by Livestock Producers Criteria |
Will the proposed regulatory framework apply? Feed is not sold |
Will the proposed regulatory framework apply? Feed is sold |
|---|---|---|
| Feed does have a drug or other substanceTable Note 1 that presents a risk of harm to human or animal health or the environment incorporated into it. | Yes | Yes |
| Feed does not have a drug or other substanceTable Note 1 that presents a risk of harm to human or animal health or the environment incorporated into it. | No | Yes |
Table Notes
- Table note 1
-
For example, specified risk material, as defined in the Feeds Regulations and Health of Animals Regulations, which is prohibited from being fed to any animals, as it poses risk of transmitting BSE.
Sections 4.3 and 4.4 of this proposal detail the feed-specific hazards, their identification, and PCP requirements.
This proposal will not address the following:
- facility licensing by the CFIA;
- inspection and/or enforcement approaches that are relative to the oversight of compliance with the requirements of regulations; or
- the role and potential impacts of sector-specific hazard identification and/or preventive control programs (either recognized or not recognized by the CFIA) on CFIA oversight activities.
These compliance and enforcement topics are addressed in the CFIA's proposed Integrated Agency Inspection Model (iAIM). This model integrates all three of the Agency's business lines of food safety, and plant and animal health into a single inspection model based on the Improved Food Inspection Model that was posted on the CFIA website in June of 2013. Consultation on the iAIM is currently underway.
4.2.1 The Agency's Approach to Regulation
[adapted from CFIA Foundations of an Outcome-based Approach paper]
Traditionally, the Agency has relied on a combination of three basic approaches to regulation: prescriptive, systems-based, and outcome-based. However, fundamental changes to the production, processing, packaging, distribution, and sale of agricultural products, coupled with changing consumer demands, industry consolidation, and rapidly evolving science and technology have prompted the Agency to re-examine the mix of approaches used to verify the integrity and safety of the agriculture and agri-food supply chain.
In committing to modernize its regulations, the Agency intends to systematically evaluate how best to use outcome-based and systems-based regulatory approaches.
An outcome-based approach specifies what is to be achieved and how compliance will be measured, but does not prescribe how to achieve an outcome. It affords regulated parties the opportunity for innovation and provides flexibility to introduce new technologies, processes, and procedures that enhance safety and/or reduce costs. It is rooted in the need to adapt to changing scientific, technological, and economic conditions.
With systems-based regulation, regulated parties are obliged to develop internal risk management plans that include procedures, training, documentation, and internal risk analyses to identify and mitigate risks in the context of a corporate compliance system.
The flexibility of an outcome-based approach will help the Agency to regulate, in an environment of change through innovation, and to enhance its capability to incorporate scientific and technological advancement without compromising safety. For the Agency, the outcome-based approach may also facilitate the assessment of comparability with safety regimes in other jurisdictions that are based on similar outcomes. Ultimately, it is a more progressive way to achieve safety and health outcomes for consumers, regulated parties, and the Agency alike. Under systems-based regulation, regulated parties must commit time and resources to understand and mitigate risk, rather than simply follow a set of specification and process requirements. Systems-based regulation lays the groundwork for enhanced accountability on the part of regulated parties.
4.3 Hazard Identification (Modernized)
It is proposed that all those involved throughout the feed supply chain complete a hazard identification and assessment with respect to the feed-related activities in which they are involved. The hazard identification and assessment must be carried out on incoming materials, materials sourced in-house, feed formulation, processes, and transportation by the feed manufacturer.
Animal feed and feed ingredients can act as a route of entry for hazards into human food and for hazards that pose a risk to animal health, plant health (e.g. pests and diseases of domestic or international quarantine significance), or the environment (e.g. invasive plant species not known to be present in a local ecosystem). To mitigate the associated risks, hazards in feeds must be identified and assessed prior to their manufacture, distribution, and feeding to animals. Multifaceted assessments are often necessary to consider both the safety to animals as the primary consumers of the feed and safety to humans as the consumers of resulting food of animal origin (e.g. meat, milk, fish, and eggs). In some cases, the risk to persons who work with and mix feed, as well as the risk to plant health or the environment, also requires assessments.
The Code of Practice offers the following general guidance, respecting the identification of hazards:
The presence in feed and feed ingredients of undesirable substances [hazards] such as industrial and environmental contaminants, pesticides, radionuclides, persistent organic pollutants, pathogenic agents and toxins such as mycotoxins should be identified, controlled and minimised.
The risks of each undesirable substance to consumers' health should be assessed and such assessment may lead to the setting of maximum limits for feed and feed ingredients or the prohibition of certain materials from animal feeding.
In some cases, hazards may be naturally occurring, accumulated from the environment or introduced into feed ingredient during their manufacture. Their concentrations in individual ingredients and their cumulative amounts in finished feeds contribute to the overall safety of feeds. The most common contaminants of concern include heavy metals, dioxins, furans and PCBs, veterinary drug residues, pesticides residues, processing aids, mycotoxins, and pathogenic microbial contaminants.
The feed supply chain has traditionally made use of materials that are by-products from food processing. Over the past two decades, a number of by-products from the fuel, forestry, mining, and other industrial sectors (e.g. kiln dust, ball clay), previously used for other purposes or disposed of, are being used in feed. Such by-products may also contain hazards such as processing additives and contaminants, other than those identified in Table 2, that are not known or routinely expected to be present in feed ingredients.
From a mixed feed perspective, hazards in ingredients may carry forward into mixed feeds. While feed ingredient manufacturers would be required to identify hazards and apply preventive controls to their products in the framework being proposed, feed ingredient manufacturing processes are not always capable of removing or reducing certain hazards if present in raw materials or introduced into the ingredient (e.g. metals, pesticides). As a result, they must be identified and accounted for in the formulation and production of mixed feed products. In addition, hazards can be introduced within feed mills from production practices (e.g. additives used to process grains and other ingredients, added medications and residues of medications remaining in cross-utilized equipment, manufacturing errors resulting in the unintentional addition of medications or ingredients that could be harmful to certain species).
Hazards may also be introduced into feeds and feed ingredients through contamination during transportation, handling, packaging, storage, and distribution. In some cases, they may be the types of contaminants already discussed, but in others, they could be substances such as fertilizers or other bulk materials remaining in rail cars, trailers, silos, or other conveyances and storage equipment that are not dedicated to the transportation or storage of feed.
With the Codex guidance in mind and with a view to providing guidance to those involved in the feed supply chain, it is further proposed that the hazards set out in Table 2, considered as known or reasonably foreseeable in feeds and feed ingredients, are, at a minimum, those that should be considered as part of the hazard identification and assessment process. Note Table 2 is provided as a non-exhaustive list of potential hazards by feed category combinations and must be considered in relation to one's individual manufacturing processes. Authorized ingredients, as listed in the Regulations, may contain specific standards for hazards of relevance to the ingredient.
| Feed/Ingredient Category | Known or Reasonably Foreseeable Hazards |
|---|---|
| Grains, oilseeds, forages, fruit and vegetables, and their by-products |
|
| Minerals |
|
| Animal products and by-products (including fish) |
|
| Fermentation products Viable microbes |
|
| Mixed feeds |
|
TSE = transmissible spongiform encephalopathy
To modernize the current regulatory framework regarding the identification of hazards, the Agency proposes to
- review and update the list of prescribed deleterious substances currently contained in section 4 of the Regulations.
- revise the list of specific hazards and standards identified for feeds, as appropriate.
- identify specific hazards and maximum limits in feeds in guidance (build on current RG-8).
- include known hazards and limits, where appropriate, in ingredient descriptions set out in Schedule IV.
As noted previously in section 2.1, the proposed Bill C-18 sets out several new regulation-making authorities for the Act, including
[5] (e.1), prescribing standards for the manufacturing or the safety of feeds.
To this end, the CFIA also proposes to use this provision, should this legislation be adopted, to require all parties involved in the manufacturing, sale, importation, and distribution of feeds and feed ingredients to complete a hazard identification and assessment for their own system.
As discussed in section 3.2, a number of industry sector feed and food safety programs are in place, and familiarity already exists with having to identify hazards in feeds and feed ingredients as a component of participating in such programs. To provide clarity and consistency, the CFIA would support such a regulation by providing guidance on feed-related hazards, their sources, and strategies to prevent or mitigate their risks.
Have your say
The CFIA is seeking feedback on the proposal to
- require all parties involved in the manufacturing, sale, and distribution of feeds and feed ingredients to complete a hazard identification and assessment for their own system.
- require that the hazards set out in Table 1 of this proposal, known or reasonably foreseeable in feeds and feed ingredients, are those that, at a minimum, should be considered as part of the hazard identification and assessment process for each system.
4.3.1 Feeds Regulations, Range of Nutrient Guarantees for Complete Feeds for Use in the Exemption of Feeds from Registration (Schedule I, Table 4)
Through negotiations and consultations among the federal government, academia, and commercial feed industry representatives, Table 4 of Schedule I was created and incorporated into the Feeds Regulations in the 1980s, as a mechanism to exempt certain groups of feeds from registration. This table contains ranges of common essential minerals and vitamins for complete feeds or those manufactured from more concentrated products (e.g. supplements and premixes) that support livestock nutritional requirements while maintaining a margin of safety for human and animal health, and the environment. The original Table 4 established exemption ranges for chickens, turkeys, swine, beef and dairy cattle, and sheep. In 1990, via two amendments, the table was expanded to include nutrient ranges as exemption criteria for complete feeds for horses, goats, ducks, and geese; and then for rabbits, mink, and salmonid fish. Since that time, there have been no other substantive changes to the table or to any of the nutrient ranges. Feeds with nutrient guarantees that fall outside the listed ranges have been subject to assessment and registration by the CFIA prior to manufacture and sale.
Although the table has never intended to establish absolute nutritional limits for livestock feeds or feeding programs, the domestic feed industry considers its nutrient ranges out of date, and therefore its use as a mechanism to exempt feeds from registration is no longer an appropriate regulatory tool.
Higher levels of certain vitamins in livestock rations (e.g. Vitamins A, D, and E) can be harmful to livestock or can be concentrated into tissues that are used for human consumption, thus posing potential risk to human health. Similarly, certain minerals (e.g. copper and zinc) or, as in the case of phosphorus, contain levels in rations that can also contribute to risks of harming the environment. To this end, the CFIA proposes to remove Table 4 from the modernized regulatory framework and to replace this mechanism with more outcome-based regulations and guidance regarding appropriate levels of specific vitamins and minerals for livestock rations.
Have your say
The CFIA is seeking feedback on the proposal to modernize the regulatory framework regarding hazards, including the replacement of Table 4 of Schedule I with a more outcome-based objective and guidance, respecting maximum levels of vitamins and minerals used in feeds.
4.3.2 Toxic Substances regulations
The federal Health of Animals Act provides authority to control not only animal diseases but also toxic substances. The control of "toxic substances" within the scope of the Act serves to enable the response to risks that are posed when animals that ingest contaminated feed are exposed to unapproved pest control agents or veterinary drugs, or other accidental or unintended exposures to hazards. The Act provides the authority to prescribe "toxic substances" by way of a separate regulatory amendment.
Current federal regulations enable action to be taken for chemical contaminations of livestock production inputs (e.g. feed and veterinary biologics) and food products derived from livestock (i.e. meat, milk, and eggs), but not on live animals. This represents a gap in the CFIA's regulatory control of the food continuum. CFIA inspectors can go on-farm to control animals for disease outbreaks, but not for exposure to chemical contaminations.
Although the Agency developed and consulted on a proposed list of toxic substances and associated amendments to the Regulations during the mid- to late 2000s, there have been no amendments to the Health of Animals Regulations to date in this regard.
The Agency proposes to move forward with the implementation of amendments to the Health of Animals Regulations regarding toxic substances, enhancing Agency response to situations wherein animals are accidently or unknowingly exposed to hazards that may have health implications to animals or humans. To prescribe a list of toxic substances in the Health of Animals Regulations allows the Agency to respond to toxic contamination events in the same way it responds to animal disease outbreak situations. Given the exemption provided to livestock producers by the Feeds Act where farm-manufactured feeds are not for sale or contain drugs or other harmful substances, as discussed in section 4.3, it is reasonable to enable the regulatory capacity to respond to accidental or unintentional situations involving feed-related hazards that could pose risks to human or animal health or the environment.
Examples of key response measures that such amendments would provide are as follows:
- mandatory notification and reporting upon suspicion of contamination with a toxic substance;
- prohibitions on the sale of a contaminated animal without a licence;
- prohibitions on the sale, the offer for sale, the exposure for sale or transfer ownership of an animal, animal product or by-product that is contaminated with a toxic substance;
- restriction on the import of animals or their products contaminated with a toxic substance;
- the ability to declare an "infected place" in order to limit the movement of animals or food produced from those animals;
- the declaration of an area as a control area until measures to mitigate the situation may be taken;
- enhanced sampling measures; and
- the ability to order means of disposal or treatment.
The principal change involves listing the toxic substances in a new schedule to the Health of Animals Regulations. In addition, minor amendments would be required to add the term "toxic substances" to a number of existing provisions in the Health of Animals Regulations. Both Governor-in-Council and Ministerial amendments would be needed, and all proposed amendments would be provided for public consultation.
The amendments that form part of this proposal would specifically address the regulations that are necessary to complement the addition of a toxic substance list. Such amendments would further strengthen the CFIA's capacity for regulatory control along the feed and food production continuum.
Have your say
The CFIA is seeking feedback on the proposal to move forward with the implementation of amendments to the Health of Animals Regulations regarding toxic substances to better enable Agency response to situations where animals are accidently or unknowingly exposed to hazards that may have health implications.
4.4 Preventive Controls (New)
To maintain a robust approach to human health, animal health, plant health, the environment, and consumer protection, the CFIA is modernizing its approach to inspection. The move toward a more prevention-focused and systems-based approach under the integrated Agency inspection model allows both the CFIA and regulated parties to more readily adapt to emerging global and scientific trends. Canadians will continue to be protected by an effective system that is both science- and risk-based. A key element of the model includes applying preventive controls by parties that are subject to CFIA-regulated products and activities.
In keeping with the direction set out in the integrated Agency inspection model, it is proposed that, following the identification and assessment of hazards within an individual system, regulated parties must develop, implement, and maintain outcome- and performance-based PCPs in accordance with the requirements proposed for each preventive control element set out in Annex 2. This requirement would apply to all in the feed supply chain; however, the extent of which will depend on the hazards present and the risk of the specific systems.
Preventive controls represent a combination of measures developed, implemented, documented, validated, and maintained by an establishment to prevent or reduce hazards to an acceptable level that, when taken as a whole, provide a science-based, objective, and auditable system, supporting the manufacture, importation, and distribution of feed and feed ingredients that
- are safe for their intended uses;
- meet all regulatory requirements; and
- do not impact negatively on human health, animal health, plant health, or the environment.
All potential hazards, as they pertain to human health, animal health, plant health or the environment, are to be identified and assessed with respect to an individual manufacturing system or situation. This approach is consistent with that in the CFIA's A New Regulatory Framework for Federal Food Inspection: Discussion Document. This type approach is already in place in Europe and being developed in the United States. The Code of Practice is an internationally developed and recognized framework that also supports a preventive controls approach to feed safety. Many of these other systems only address aspects of feed safety that affect human health and human food safety. Mitigating risks to food safety is the CFIA's highest priority, and the health and safety of Canadians is the driving force behind the design and development of CFIA programs. In addition, the current and future economic prosperity of the Canadian agriculture and forestry sectors relies on a healthy and sustainable animal and plant resource base. As such, to minimize and manage risks, the CFIA is continually improving its program design and delivery in the animal health and plant resource areas. In an effort to protect the natural environment from animal and plant diseases, invasive plant species, and plant pests, the CFIA also performs extensive work related to the protection of environmental biodiversity. For this reason, the scope of feed preventive controls must also address related risks to animal health, plant health, and the environment.
The CFIA proposes that all feed manufacturers – including ingredient manufacturers, commercial feed mills, on-farm feed manufacturers (not subject to exemptions), retailers that repackage feeds, importers, and anyone handling feeds in bulk – be required to develop, implement, and maintain a written PCP.
As noted in section 2.1, the proposed Bill C-18 sets out several new regulation-making authorities for the Act, including
[5] (h.2) respecting quality management programs, quality control programs, safety programs, preventive control plans, or any other similar programs or plans to be implemented by persons who conduct any activity regulated under this Act.
The CFIA proposes to use this provision, should this legislation be adopted, to develop regulations in this regard.
For on-farm feed manufacturers that are not subject to exemption, the scope, scale, and level of detail expected in these plans is anticipated to be minimized (and, consequently, the level of oversight by CFIA) where, for example
- operations are only producing one species or similar species (e.g. poultry), as opposed to multi-species farms that have ruminants and non-ruminants; or
- if medicated feeds are being manufactured that do not require a withdrawal prior to marketing of livestock for food processing.
To elaborate further on its expectations regarding preventive controls, the following subsections to 4.4 provide additional information and guidance in this regard.
4.4.1 Preventive Control Plans
A PCP is a written document that sets out how safety and other regulatory requirements (e.g. labelling, product composition) will be achieved for an individual system. PCPs are recognized internationally as the best way to demonstrate that risks and hazards are controlled or eliminated, because they focus on prevention through a systems-based examination of operations by regulated parties. PCPs will address not only food safety requirements but also related requirements such as animal, plant, and environmental risks, and labelling.
4.4.2 Why is a Preventive Control Plan required?
The key components of a preventive control system-based approach that contribute to the production of safe and compliant feeds
- are consistent with current and future international approaches.
- facilitate market opportunities.
- allow for more flexible and efficient compliance verification processes.
- allow for continuous improvement.
- contribute to a more effective and consistent approach to follow-up and correction of deviations.
4.4.3 Elements of a Preventive Control Plan
Based on the elements set out in the CFIA's proposed modernized food regulatory framework, the integrated Agency inspection model and the Codex Code of Practice on Good Animal Feeding, seven elements are proposed for PCPs for feed:
- product and process control;
- sanitation, biosecurity, and pest control;
- hygiene, biosecurity, and employee training;
- equipment design and maintenance;
- physical structure and maintenance;
- receiving, transportation, and storage; and
- recalls, complaints, and record keeping.
Domestic Feeds and Feed Ingredients
In developing a PCP, operators of domestic production and distribution establishments should identify
- and describe all hazards in their system, and consider regulatory requirements and their related validated controls by indicating
- the planned sequence of measurements and observations to determine whether a control measure is effective (monitoring procedures).
- the methods, procedures, tests, and other evaluations, in addition to the monitoring that is used to determine whether a control measure is, or has been operating as intended (verification procedures).
- the planned corrective actions when validated controls are ineffective at controlling the hazard and/or when regulatory requirements are not met (corrective action).
- who will be responsible to ensure that there is monitoring, verification, and corrective action by specifying
- who the responsible party is for each step and the required training and competencies.
- the frequency of delivering each activity.
- where, how, and by whom activities will be documented:
- Identify appropriate records that demonstrate they are delivering their system.
- Identify where the records will be generated, who is responsible for their completion, how frequently and how long they will be retained.
Imported Feeds and Feed Ingredients
Importers have no direct control over production in other countries, and thus need to develop other strategies to address risks, including records demonstrating how these strategies are effective. Ultimately, such strategies would need to demonstrate that foreign manufacturers have designed, developed, implemented, and verified PCPs to assure the safety of feed and feed ingredients and compliance with other Canadian regulatory requirements that are comparable with those required by domestic manufacturers.
Importers should have PCPs that include elements which apply to their operation. Importers without facilities, for example, would not be expected to address physical structure and maintenance. However, all importers would, at a minimum, include the following elements in their PCPs:
- Element 1: Process and Product Controls
- Element 7: Traceability, recall, and complaints
Importers who are involved in further handling or repackaging of commodities would address all seven elements of the PCP.
Feeds and feed ingredients for export
An exporter's PCP would require that export controls address any foreign regulatory requirements (e.g. product identification and labelling requirements).
4.4.4 Requirements for each element of a preventive control plan
As additional more substantive guidance regarding feed-related PCPs, a set of proposed outcome-based regulatory requirements and performance criteria are identified and described for all preventive control elements in Annex 2.
4.4.5 Preventive Control Plans versus HACCP Systems
During the CFIA's March 2013 Hazard Identification and Preventive Controls stakeholder workshop, in particular, participants questioned the CFIA's use of "preventive control plans" as current terminology in lieu of such other terms as "HACCP," "HACCP-based," "prerequisite programs" that were commonly used in the context of both the CFIA's Food Safety Enhancement Program and Quality Management Program (QMP) for fish and seafood in recent years.
While PCPs (also referred to as GMPs or prerequisite programs) make up a portion of a HACCP system, the intent of a HACCP system is solely to address potential food safety risks and focuses control at critical control points (CCPs). HACCP is a well-recognized system for controlling food safety hazards. A number of existing private sector feed programs use the principles of HACCP in their systems.
The requirements for a feed PCP extend beyond those that are typically met by a HACCP system. Addressed in addition to food safety risk, PCPs mandated within the scope of the proposed regulatory framework need to address hazards that pose a risk of harm to animal health, plant health, and the environment. This proposal does not preclude the development and implementation of voluntarily HACCP systems by regulated parties in order to achieve food safety objectives and compliance with regulatory requirements. If a HACCP program does not meet all of the proposed regulatory requirements, additional preventive controls may need to be implemented to address these gaps.
Have your say
The CFIA is seeking feedback on the proposals to
- require feed manufacturers – including ingredient manufacturers, commercial feed mills, on-farm feed manufacturers, retailers that repackage feeds, importers, exporters and anyone handling feeds in bulk – to develop, implement, and maintain a written PCP.
- Indicate whether there should be exceptions (other than those provided by the Feeds Act to livestock producers) for specific persons or feed activities. Why?
- require feed manufacturers, including ingredient manufacturers, distributors, importers and exporters, to include all the elements of a PCP that apply to their operations proposed in section 4.4.3.
- use the requirements for each element of a PCP proposed in section 4.4.4 and Annex 2 of the proposal as a framework from which outcome-based regulations, model systems, industry guidance, and guidance to CFIA inspection staff (how to determine whether compliance is being achieved) will be developed.
4.5 Record Keeping
Record keeping is a critical part of the system. A system will only be as strong as the records that support it. It is proposed that record keeping be included as a component of the PCP.
The CFIA proposes to expand the scope of the currently required records to include those that pertain to the hazard identification and the PCP. The PCP would need to identify all records associated with each element of the PCP. It would also indicate the location and retention period for the records, how frequently they will be updated, and responsibility for their completion. Examples of record-keeping activities include, but are not limited to, the following:
- validation of control measures (e.g. critical limits or tolerances), monitoring (demonstrating control of a hazard or regulatory requirement; for example, shipping, purchase orders, training, equipment calibration and maintenance, pest/disease management records);
- verification;
- complaints and how they were resolved;
- deviations and corrective actions taken; and
- management review by regulated party.
In addition, records pertaining to incoming materials and production should be kept to facilitate feed recalls and trace-backs.
Have your say
The CFIA is seeking feedback on the proposal to
- expand the scope of the required records to include those pertaining to the hazard identification and the PCP.
- require a retention period for records.
4.6 Roles and Responsibilities
There are many different participants in the livestock feed and production system, each with an equally important role to play. From government to producer, each participant shares responsibility in addressing and maintaining the safety of Canada's animal and plant resource bases, contributing to the production of safe food for all Canadians and our international customers and to the safety of the environment.
4.6.1 Government
The Government has a role in feed safety through the development and enforcement of regulations, and their associated guidance documents, for feeds. The CFIA will be responsible for
- setting standards in regulations and guidance for known hazards.
- conducting risk analysis of known and emerging hazards in feed and feed ingredients according to the Codex Risk Analysis Framework to include:
- risk assessment comprised of hazard identification, hazard characterization, exposure assessment, and risk characterization.
- risk management, including the evaluation and implementation of risk management options, monitoring, and review.
- the provision of risk communication advice by documenting the risk assessment and risk management options in a transparent manner;
- verifying initial compliance and conducting compliance inspections based on the level of risk associated with a facility through inspection activities;
- focusing efforts on health and safety aspects of the feed industry along a continuum with higher risk manufacturers receiving greater oversight;
- developing education programs to promote understanding by feed manufacturers and producers of their role and the ways in which they can lower their risk factors;
- employing compliance management activities; and
- responding to instances of non-compliance through a wide variety of measures that are available to the CFIA.
4.6.2 Industry Organizations
Industry involvement is essential to the effective management of a feed safety system. Industry groups are in an excellent position to provide leadership and direction within their sector by playing a role in
- producing safe feed, designing systems and processes (i.e. manufacturing and preventive control) to ensure that they meet regulatory requirements;
- promoting best practices and providing peer support and guidance or knowledge sharing and technical expertise within their sector;
- knowing and complying with relevant legislative requirements to their business;
- providing complete, accurate, and timely information to the CFIA;
- understanding the role Agency employees perform;
- being aware of the ethical obligations that govern the actions of CFIA officials who are part of the broader obligations of regulated parties;
- educating the government about the needs of the industry and what can help industry succeed; and
- working with government to establish standards, and identify key preventive controls and maximum levels of hazards.
4.6.3 Individual Regulated Parties
Individual regulated parties, such as feed establishment operators and livestock producers, are the purchasers and end-users of feed products. Their role in the system includes
- being knowledgeable of, understanding, and complying with relevant legislative requirements and stewardship practices related to their business.
- obtaining third-party certification where appropriate.
- monitoring of feeds and feed manufacturing conditions to verify that requirements are being met.
- providing complete, accurate, and timely information to the CFIA.
- understanding the role Agency employees perform.
4.6.4 Existing Private Certification Programs
A number of external feed preventive control programs already exist. This includes the feed component of Canadian On Farm Food Safety Programs, ANAC's FeedAssure program, private industry programs, and a number of international programs such as GMP+, FAMI-QS, GLOBALG.A.P., and AFIA's Safe Feed/Safe Food program. The commitment to improving feed safety, shown by a number of parties that are choosing to participate voluntarily in one of these programs, is commendable and demonstrates the progressive role many in the feed industry are already playing. In addition to these existing programs, third parties – namely, industry-related non-government organizations, academia, or other service providers – have expertise and knowledge in their fields and may offer guidance, programs, training, self-assessments, and peer support to equip their members or sectors with the necessary tools to achieve compliance with the proposed changes to the Feeds Regulations. There are significant opportunities to leverage their leadership capacity and expertise through partnerships (e.g. private sector or public-private). Likewise, other government departments at the federal and provincial, and territorial levels may play an advisory role by providing expertise and tools.
5 Implementing Proposed Regulatory Framework
On December 9, 2013, Canada's Minister of Agriculture and Agri-Food tabled the Bill C-18 in Parliament, proposing to amend several federal agricultural statutes, including the Act. Among the suite of proposed amendments, a new authority to make regulations "respecting quality management programs, quality control programs, safety programs, PCPs or any other similar programs or plans to be implemented by persons who conduct any activity regulated under this Act" would be provided to the CFIA. This strengthens the regulation-making authorities in the Act and would allow the development of modernized regulations, requiring the preparation, implementation, and maintenance of PCPs relative to livestock feeds. For the purposes of the measures set out in this regulatory framework proposal, the CFIA will move forward with regulatory development on the assumption that the proposed Act amendments in Bill C-18 will come into force in the near term.
The Parliament of Canada website provides a copy of Bill C-18.
Should Bill C-18 not pass into law in the near term, the CFIA would explore other options for implementing regulations for process-based feed controls.
6 Summary
6.1 Proposal Synopsis
- In aligning with the mandate of the CFIA and the Act, the Agency is proposing a feed preventive control framework that addresses hazards to animal health, plant health and the environment, in addition to mitigating public health risks.
- With respect to identifying hazards, the Agency proposes to review the current Regulations, the ingredient Schedules attached to the Regulations, and regulatory guidance publications with respect to hazards and any maximum limits or action levels for feeds, and subsequently revise the regulations and policies, as appropriate.
- As a complementary measure, the Agency proposes to move forward with the implementation of amendments to the Health of Animals Regulations in respect of toxic substances to enhance Agency response in situations where animals are accidently or unknowingly exposed to hazards that may have health implications.
- Other than livestock producers who are specifically exempted from the application of the Feeds Act and Regulations, all regulated parties in the feed supply chain must develop, implement, and maintain outcome- and performance-based PCPs.
- The CFIA will provide guidance and/or model systems to assist smaller operations with developing PCPs and in guiding inspection staff.
6.2 Next Steps
Feedback obtained during this consultation will be combined with feedback from the other two modules. A final consolidated proposal for feed regulatory modernization will be developed to
- integrate the feedback received on the three individual proposals (feed ingredient authorization and assessment, feed labelling, and feed hazard identification and preventive controls).
- include information on compliance and enforcement approaches, including the role of private sector programs in risk-based inspection approaches.
- outline the modernized inspection approach the CFIA will administer, and incorporate the input received from consultations on the integrated Agency inspection model.
Following the development and publication of the proposed overall framework, the CFIA will review its service delivery costs and fees within the Feed Program as part of an Agency-wide User Fee modernization initiative. Objectives for the User Fee initiative include promoting effective and responsive service delivery; establishing a consistent and robust approach to user fee development; and fostering an understanding that user fees may be charged for services, products, and rights and privileges provided by the Agency. For detailed information, refer to the Cost Recovery Policy and Framework.
The Agency will hold public consultations on user fees related to services provided by the Feed Program. Service recipients will have an opportunity to comment on the detailed user fee proposal once it is made public.
6.3 Contact
We strongly encourage you to provide your input and feedback, which is critically important to the success of the regulatory modernization initiative. Written comments may be forwarded to
Sergio Tolusso
Canadian Food Inspection Agency
Animal Feed Division
59 Camelot Drive
Ottawa, ON K1A 0Y9
Email: Sergio.tolusso@inspection.gc.ca
Fax: 613-773-7565
Annex 1 - FRSG Recommendations
In October 2013, the CFIA shared its current thinking regarding a proposed regulatory framework for feed hazard identification and preventive controls with its industry/government Feed Regulatory Steering Group (FRSG). Following this briefing, the industry association members of the group collaborated to provide the CFIA with the following feedback and six recommendations:
The associations took particular account of the comments made by participants at the stakeholder workshop held in March 2013, where a clear majority supported the inclusion of hazard identification and preventive controls in a new Canadian regulation. The associations also considered the current EU and proposed U.S. rules, both of which require that hazard identification and control plans be documented and implemented in animal feed production establishments. Finally, the exchange of views was guided by the overriding goal—advanced by CFIA and supported by the agri-food stakeholder community—of implementing outcome-based regulations focused on the mitigation of risk.
At the conclusion of discussions, the six associations unanimously agreed to make the following recommendations to the co-chairs of the FRSG:
- A modernized feed regulation—in concert with regulatory guidance and administrative procedures—should include measures to promote the implementation of hazard identification and preventive control plans in feed manufacturing, storage and distribution facilities. Such measures should focus exclusively on feed safety, considering that nutritional requirements are based on commercial arrangements between suppliers and their customers.
- While such plans would need to be appropriately documented, monitored and verified, the form of delivery would be outcome-based. For example, while most commercial facilities will favour ANAC's FeedAssure program, there are other HACCP-based approaches that might be acceptable, provided they satisfy the intended feed safety outcome.
- It is recognized that the EU and U.S. feed safety statutes authorize the inclusion of mandatory hazard identification and preventive controls in regulation. However, there appears to be some doubt as to whether the Feeds Act would enable such a requirement in a new Canadian regulation. The associations agreed that a mandatory approach along the lines of the EU and U.S. frameworks, coupled with a risk-based facility licensing system, would be the preferred route for the commercial feed industry in Canada.
- If the statutory authority for mandatory hazard identification and preventive control programs is indeed found to be lacking in Canada, the associations agreed that CFIA should put in place appropriate incentives to encourage the widespread use of recognized hazard identification and control plans for the commercial feed industry.
- In its December 2010 policy paper and again in its June 2013 submission to the regulatory steering group, ANAC proposed that CFIA refocus its inspection resources to concentrate on establishments without hazard identification and control plans in place, and to adopt a modernized, system-based review of facilities with formal preventive controls and independent verification programs. In 2010, CFIA officially acknowledged that ANAC's FeedAssure program meets all the requirements of the Agency's own Food Safety Enhancement Program. However, FeedAssure-certified facilities are given only token recognition, by receiving one less partial inspection per year. The six associations recommend that CFIA provide an incentive for commercial feed facilities to participate in the FeedAssure program by lessening the inspection burden on certified facilities and reallocating scarce enforcement resources to commercial establishments without preventive control plans and thus representing the highest risk.
- Finally, the associations recommend to the FRSG co-chairs that CFIA staff expedite any policy or legal inquiries needed to ascertain whether a modernized feeds regulation can, within existing statutory authorities, incorporate provisions for risk-based licensing of commercial facilities, and mandatory hazard identification and control plans. In the absence of such authorities, the associations further recommend an examination—in consultation with the full FRSG—of methods of adapting the facility inspection system to fit an outcome-based regulatory environment. The intent would be to provide appropriate incentives which would best encourage the expansion of FeedAssure or other hazard identification and control programs of equivalent rigour.
Annex 2 - Proposed Outcome-based Regulatory Requirements
Introduction
As identified in section 4.4.3 of the proposal, the following elements are proposed for PCPs for feed:
- Product and process control
- Sanitation, biosecurity and pest control
- Hygiene, biosecurity and employee training
- Equipment design and maintenance
- Physical structure and maintenance
- Receiving, transportation and storage
- Recalls, complaints and record-keeping
This Annex is intended to provide a framework from which outcome-based regulations, model systems, industry guidance and guidance to CFIA inspection staff (as to how to determine if compliance is being achieved) will be developed. The balance between requirements being established in regulations versus provided in guidance will be determined once the legal drafting of the regulations begins in earnest following consultation. The performance criteria outlined for each PCP element in this Annex represent examples of measures that may be employed to address potential risks posed from hazards identified in materials and processes used to manufacture, store and distribute feeds and feed ingredients. Flexibility would be provided for regulated parties to use other measures not outlined here, provided they are effective in addressing potential risks.
1. Product and Process Control
Outcome
Process and product controls ensure the production of safe feed that is in compliance with applicable regulations.
Standard
The regulated party must develop, implement, monitor and review for their specific system the effectiveness of a PCP, relative to product and process controls, to assure that:
- a hazard identification exercise that considers all potential biological, chemical and physical hazards is completedFootnote 3;
- written plans identifying preventive control measures to eliminate, prevent, mitigate, or reduce hazards to an acceptable level as well as meeting other regulatory requirements are developed; and
- effective control systems relative to incoming ingredients, process controls, product flow and people flow are implemented, monitored and reviewed periodically and when operations change.
Process control outcome: performance criteria
- Incoming inputs (for example, ingredients, raw materials, plants and animals)
- identification of incoming inputs materials/ingredients and their source
- identification of incoming inputs, with written specifications, if:
- there is a potential hazard
- an incoming input is critical to biosecurity, product composition, health status and/or nutrition profile
- verification from source suppliers regarding the potential hazard
- documented handling procedures for incoming inputs to maintain integrity and prevent hazard introduction, degradation or contamination
- Materials originating in the facility (for example, returns, rework, flush, dust collector materials, recycled solids from wastewater treatments)
- identification of incoming inputs materials/ingredients and their source
- identification of incoming inputs, with written specifications, if:
- there is a potential hazard
- an incoming input is critical to biosecurity, product composition, health status and/or nutrition profile
- verification from source suppliers regarding the potential hazard
- documented handling procedures for incoming inputs to maintain integrity and prevent hazard introduction, degradation or contamination
- Product formulation and specifications
- written formula for each feed or feed ingredient manufactured in the facility
- written specifications for each feed or feed ingredient manufactured in the facility that addresses animal health, including appropriate nutrient levels, contaminants that present a risk to animal health, and species specific hazard analysis, in addition to hazards that pose a risk to human health
- feed additives, processing aids and chemicals (including medicating ingredients) used are permitted for use in and on the feed or feed ingredient commodity
- Processing
- written description of:
- processing steps (for example, mixing records), associated control measures and critical limits
- movement of the feed or feed ingredient in the facility (for example, process flow diagram)
- procedures for production sequencing where multiple feed and feed ingredients are manufactured in a facility
- critical limits are validated using scientific data
- written description of:
Product control outcome: performance criteria
- Packaging
- All packaging materials are suitable for the intended use.
- Labelling, Product identification
- The information on the label is complete, truthful and not misleading.
- It accurately represents the content of the feed or feed ingredients and its content.
- It meets regulatory requirements.
- Finished Product
- The finished feed or feed ingredient is evaluated for compliance with regulatory requirements.
- Outgoing feed and feed ingredients and their destination are identified, including any feed and feed ingredients destined for export (for example, the immediate purchaser is identified).
2. Sanitation, biosecurity and pest control
Outcome
Prevent contamination, control hazards and facilitate hygienic production of feed or feed ingredient through effective sanitation and pest control.
Standard
The regulated party must develop, implement, monitor and review the effectiveness of a PCP, relative to sanitation and pest control, to assure that:
- the sanitation of the premises and equipment is maintained at an appropriate level so as not to become a source of contamination for feed and feed ingredients before, during and after production; and
- the entry of pests (rodents, insects, birds, etc.) inside and outside the facility is controlled (including detection and elimination of pests).
Sanitation and biosecurity outcome: performance criteria
- There is a schedule for cleaning and/or sanitizing, which includes the concentration of chemicals to be used, and temperature and pressure requirements (if applicable), for:
- the facility, including preparation and storage areas;
- equipment; and
- feed and feed ingredient contact surfaces, including containers and utensils.
- Equipment used for cleaning and/or sanitizing is capable of delivering the requirements of the sanitation program.
- Cleaning and/or sanitizing is carried out in a manner that does not contaminate feed, feed ingredients or packaging materials commodities (through contact with these) or spread pests or pathogens, during or after cleaning and/or sanitizing.
- Effectiveness of cleaning, sanitizing is assessed visually or through sampling (for example, environmental, product) using accepted methods or procedures, as applicable.
- Feed and feed ingredient contact surfaces are free from contamination (for example, the accumulation of dust, dirt, feed or feed ingredient residue and other debris).
Pest control outcome: performance criteria
- There is a program for control and removal of pests, including vectors.
- There is a schedule for monitoring for evidence of pest activity, including vectors.
Chemicals outcome: performance criteria
- A listing of chemicals or specification sheets for chemicals used in biosecurity, sanitation, treatment, and pest control is maintained.
- The use of chemicals does not contaminate feed or feed ingredients, equipment, utensils or feed and feed ingredients contact surfaces.
- Chemicals are suitable for their intended use.
- Chemicals are properly stored, controlled, and labelled.
- Chemicals are dispensed, handled, used and applied by trained personnel.
3. Hygiene, biosecurity and employee training
Outcome
All persons in the feed manufacturing facility adhere to sound hygiene practices so as to not contaminate feed. All staff involved in the manufacture, storage and handling of feed and feed ingredients are adequately trained and supervised and aware of their role and responsibility in ensuring safe and compliant feed.
Standard
The regulated party must develop, implement, monitor and review the effectiveness of a PCP that includes measures to assure that people who come directly or indirectly into contact with feed (employees and visitors) do not become a source of contamination of feed, including requirements that people:
- maintain an appropriate degree of personal cleanliness;
- behave and operate in an appropriate manner (includes appropriate protective clothing, health program);
- have training to a level appropriate to the operations they are to perform;
- are following effective bio-security practices; and
- are provided with proper hygiene and bio-security instruction (staff and visitors).
Hygiene and biosecurity outcome: performance criteria
- Effective biosecurity practices are in place, including:
- controlled access
- employee facilities, hand-washing and sanitizing stations
- use of sanitary clothing and footwear
- Hygienic practices are in place, including:
- proper hand-washing or use of sanitizers/gloves or boot wash
- rules for employee conduct
- precautions for personnel with open wounds or a communicable disease
Employee training outcome: performance criteria
- A written description of training requirements and training records for employees responsible for activities under the PCP are maintained.
- Employees are trained to carry out the range of tasks and duties they are required to perform.
- Effectiveness of training is verified by the regulated party.
4. Equipment design and maintenance
Outcome
Equipment, utensils and containers are effective for their intended purpose and are designed, constructed, installed and maintained in a manner that prevents feed contamination.
Standard
The regulated party must develop, implement, monitor and review the effectiveness of a PCP to assure that:
- equipment, utensils and containers are:
- of adequate and appropriate design, constructed, installed and maintained (including calibration) such that they do not become sources of contamination for feed and are effective for their intended purpose;
- easily cleaned, accessible for servicing and inspection or easily disassembled for those purposes, as required;
- constructed of materials with no toxic effect in intended use
- scales and metering devices used in the manufacture of feed and feed ingredients are appropriate for the range of weights and volumes to be measured, and tested regularly for accuracy
- mixers used in the manufacture of feed and feed ingredients are appropriate for the range of weights or volumes being mixed and are capable of manufacturing suitable homogeneous mixtures and homogeneous dilutions, and tested regularly to verify their performance; and
- other equipment used in the manufacture of feed and feed ingredients is appropriate for the range of weights or volumes being processed and monitored regularly.
Equipment design and maintenance - performance criteria
- All equipment, utensils, containers, and controlling and measuring devices used in the preparation and storage of feed and feed ingredients are:
- used only for the facility's intended purpose,
- functioning as intended,
- maintained in a good state of repair,
- cleanable, and
- properly stored.
- Controlling or measuring devices used in the preparation of feed and feed ingredients are calibrated and maintained to ensure measurement accuracy and effectiveness.
5. Outside premises, surroundings and maintenance
Outcome
The design and layout of feed facilities prevents cross-contamination between and during operations and facilitates effective maintenance and sanitation.
Standard
The regulated party must develop, implement, monitor and review the effectiveness of a PCP to assure that physical structures are:
- constructed in a manner that permits ease of operation, maintenance and cleaning and minimizes feed contamination;
- maintained in a manner to assure the facility does not become a source of contamination for feeds, including:
- appropriate product and people flow;
- water and steam meet potability standards and be of suitable quality for animals and the food chain; and
- sewage, waste feed and feed ingredients, wastewater, and other material containing unsafe levels of hazards are disposed of in a manner which avoids contamination of equipment, feed and feed ingredients.
Buildings outcome: performance criteria
- Exterior building structures
- The exterior of the building is maintained to prevent entry of pests and contaminants.
- Interior building structures are appropriate for intended use
- Floors, walls and ceilings are:
- cleanable
- made of materials that will not contaminate regulated commodities
- in good repair
- In feed and feed ingredient preparation areas:
- floors are constructed and maintained to permit adequate drainage
- glass, where breakage may contaminate feed and feed ingredients, is properly protected
- Floors, walls and ceilings are:
- Hygienic flow and separation
- Where there is potential for cross-contamination, incompatible operations are controlled by physical separation or other effective means.
- Lighting
- There is sufficient light to allow the intended activity to be conducted effectively.
- Lighting equipment (bulbs and fixtures) that is located where breakage could contaminate feed and feed ingredients is protected, as needed.
- Ventilation
- Ventilation provides sufficient air exchange to control moisture and minimize condensation.
- Ventilation systems are designed and constructed to prevent contamination (for example, air flow into clean areas is not contaminated).
- Ventilation systems are adequately maintained and cleaned.
- Employee facilities
- When present, the location and design of employee facilities such as washrooms, lunch rooms and change rooms do not contribute to or cause contamination of feed and feed ingredients or production areas.
- An adequate means of hygienically washing and drying hands and personal protective equipment (for example, boots) is provided (including wash basins and a supply of safe water):
- The location of hand-washing and sanitizing stations does not contribute to or cause contamination of feed and feed ingredients.
Water, ice and steam outcome: performance criteria
- Water, ice and steam safety
- Water, ice and steam that come in contact with feed and feed ingredients or feed and feed ingredients contact surfaces meet safety requirements as defined in Health Canada's Guidelines for Canadian Drinking Water Quality – Summary Table or are safe for intended use.
- There is no cross-contamination between safe and unsafe water supplies.
- Safety of water must be confirmed using recognized methods at a frequency adequate to confirm its safety:
- Where municipal sources are used, municipal test results may be accepted.
- Water and steam handling equipment
- Equipment is designed, installed and maintained in a manner that will not jeopardize the safety of water.
Waste disposal outcome: performance criteria
- Waste storage areas and containers are identified, of suitable capacity, secured and cleaned to avoid attracting pests.
- Waste disposal, including effluent lines, does not contaminate commodities, feed and feed ingredient preparation areas or the environment.
- Drainage and sewage systems are adequate for the volume and type of effluent being produced during normal processing and cleaning operations, and backflow is prevented.
6. Receiving, transportation and storage
Outcome
Feed is protected from potential sources of contamination and any damage that is likely to render the feed unsuitable for consumption or use during receiving, transportation and storage. Growth of pathogenic micro-organisms is controlled.
Standard
The regulated party must develop, implement, monitor and review the effectiveness of a PCP that assures:
- measures to eliminate, prevent, mitigate, or reduce hazards to an acceptable level during receiving, transportation and storage (including incoming and outgoing materials) are implemented, monitored and periodically reviewed, including:
- feed and feed ingredients are received, transported and stored in a manner that maintains their identity and integrity, minimises deterioration, contamination and cross-contamination and enables the correct feed to be sent to the right animal group;
- chemical fertilizers, pesticides and other materials not intended for use in feed and feed ingredients are identified, transported, handled and stored in a manner to avoid the potential for manufacturing errors and contamination and cross-contamination of feed and feed ingredients
The regulated party needs to consider both finished products and by-products which are intended for other uses that are regulated by the CFIA (e.g. fertilizer).
Receiving, transportation and storage: performance criteria
- Conveyances, when used for the transport of feed and feed ingredients:
- are clean and protect feed and feed ingredients from contamination, damage, pest propagation and deterioration, (including temperature and humidity controls) (for example, refrigeration); and
- are not being used to transport any material or substance that might cross-contaminate or adulterate feed and feed ingredients.
- Feed and feed ingredients in need of temperature and humidity controls (for example, refrigeration or freezing) are maintained (for example, not left out at ambient temperatures for prolonged periods).
- Conveyances are loaded, arranged and unloaded in a manner that prevents damage and contamination of the incoming materials and ingredients and/or finished feed and feed ingredients.
- Storage areas allow the:
- separation of feed and feed ingredients and other materials, including returned feed and feed ingredients;
- control of temperature and humidity to prevent deterioration and spoilage; and
- stock to be rotated to maintain suitability, quality and safety.
7. Recalls, complaints and record-keeping
Standard
The regulated party must develop, implement, monitor and review the effectiveness of a PCP for recalls, complaints and record-keeping, including controls relative to:
- traceability of feed and feed ingredients and by-products (one step forward, one step back) is supported by proper records regarding the production, distribution and use of feed and feed ingredients;
- notification of the CFIA of any unsafe or non-compliant products in Canada or abroad;
- identification, investigation, correction and reporting of product complaints; and
- procedures to effectively and rapidly identify, locate, control distribution and/or remove feed and feed ingredients from the marketplace if known or probable adverse effects on human health, animal health, plant health or the environment health are identified.
Traceability and recall outcomes: performance criteria
The traceability and recall system must be able to:
- identify sources of inputs incoming materials (ingredients) for the implicated feed and feed ingredients;
- stop any further distribution and sale;
- trace feed and feed ingredients (including by-products, if implicated) to the next point of distribution;
- contact customers and the CFIA;
- retrieve the implicated feed and feed ingredients; and
- maintain accurate traceability records in accordance with certain applicable standards.
Complaints outcome: performance criteria
- Complaints are documented and investigated by the regulated party are investigated to determine if there is non-compliance with legislative requirements.
- Corrective action is taken for non-compliance, to address the implicated feed and feed ingredient, and any changes required to processes or procedures to prevent recurrence